© Borgis - Postępy Nauk Medycznych 9/2009, s. 696-705
Tomasz Dębski, Lubomir Lembas, *Józef Jethon
Basal cell carcinoma. Current views (Part I). Epidemiology, pathogenesis, clinical features
Rak podstawnokomórkowy skóry. Współczesne poglądy (Część I). Epidemiologia, patogeneza, obraz kliniczny
Department of Plastic Surgery, The Medical Centre of Postgraduate Education in Warsaw
Head of the Department: Józef Jethon, MD, PhD, Professor of Plastic Surgery
Streszczenie
Basal Cell Carcinoma (BCC) is one of the most common human malignancies. Although it is not directly life-threatening and rarely metastasises, its local malignant features cause significant functional and aesthetic disturbances, what has a profound effect on the quality of life of patients.
The constantly increasing incidence of BCC observed within the last couple of years and the fact that this malignancy tends to occur in younger and younger people prove that BCC is now and will be in the future an important clinical problem. However, this problem is undernoticed and underestimated mainly to its low mortality and the fact that this malignancy is not listed in incidence reports.
Although currently several factors are suspected to be responsible for BCC the most important roles in cancerogenesis are played by UV radiation and advanced age of patients. They account for more than 90% of BCC.
Diagnosis of BCC basing only on the clinical appearance is difficult and depends on physician´s experience to a large extent. Traditionally, several clinical forms of BCC can be distinguished; however, the awareness of their existence may only help distinguish oncologically suspicious lesions, and not diagnose them. A histopathological examination is the only test that can verify and complete the BCC diagnosis.
The clinical course of BCC is not characteristic and cannot be predicted: a lesion may not change for years, it may grow slowly or extremely fast, the infiltration area may enlarge or recede, it may also ulcerate or tend to heal. BCC metastases occur extremely rarely (<0.1% of cases).
Summary
Rak podstawnokomórkowy skóry (Basal Cell Carcinoma – BCC) jest najczęściej występującym nowotworem złośliwym człowieka. Chociaż nie zagraża on bezpośrednio życiu i rzadko daje przerzuty, jego miejscowa złośliwość jest przyczyną znacznych zaburzeń funkcjonalnych i estetycznych, co istotnie wpływa na jakość życia pacjentów.
Stały wzrost zachorowań na BCC obserwowany w ciągu ostatnich lat oraz tendencja do występowania tego nowotworu u osób coraz młodszych, świadczą o tym, że BCC stanowi i będzie stanowił istotny problem kliniczny w przyszłości. Problem ten jest jednak niedostrzegany i niedoceniany głównie ze względu na niewielką śmiertelność oraz nieuwzględnianie tego nowotworu w zestawieniach zachorowalności.
Chociaż w chwili obecnej postuluje się kilka czynników odpowiedzialnych za powstawanie BCC, najistotniejszą rolę w kancerogenezie odgrywają promieniowanie UV oraz zaawansowany wiek chorych. Odpowiadają one za ponad 90% zmian nowotworowych.
Rozpoznanie BCC na podstawie samego obrazu klinicznego jest trudne i w dużej mierze zależy od doświadczenia badającego. Tradycyjnie wyróżnia się kilka form klinicznych BCC, których znajomość może pomóc jedynie w wyodrębnieniu zmian podejrzanych onkologicznie, a nie w rozpoznaniu. Jedynym badaniem weryfikującym rozpoznanie wstępne i decydującym o rozpoznaniu BCC jest badanie histopatologiczne.
Przebieg kliniczny BCC jest niecharakterystyczny i nieprzewidywalny: zmiana może nie zmieniać się przez lata, rosnąć powoli lub bardzo szybko, zwiększać obszar nacieku lub ustępować, ulegać owrzodzeniu lub nawet goić się. Przerzuty BCC zdarzają się niezwykle rzadko (<0,1% przypadków).

„A surgeon treating neoplasms has to acquire the whole extensive knowledge on neoplasms, their distinctive pathology and natural history as well as the knowledge on rules, methods, techniques, diagnostic and therapeutic possibilities as well as possibilities to work in an oncological team the part of which a surgeon is.”
prof. Tadeusz Koszarowski, the creator of Polish oncology
Neoplastic lesions of the skin are among the most common human malignancies. They originate from different skin tissues and structures such as:
? epidermis,
? skin appendages (hair follicles, sebaceous glands, eccrine sweat glands, apocrine sweat glands),
? pigment cells (melanocytes),
? mesenchymal structures (fibrous tissue, fatty tissue, blood and lymphatic vessels, muscles),
? nerves and APUD cells of the neuroendocrine system,
? lymphatic system cells.
The vast majority of neoplasms originating from the above skin structures are benign neoplasms. They are characterised by slow local growth and lack of intensive tissue damage.
The remaining part of neoplastic lesions consists of skin malignancies including carcinomas (originating from the epidermis and skin appendages), melanomas (originating from pigment cells), lymphomas (originating from the lymphatic system cells) and sarcomas (originating from other skin cells) (tab. 1).
Table 1. Classification of skin cancers according to the WHO classification (14).
| Skin structures |
Benign neoplasms (examples) |
Malignancies |
| Non-melanoma skin cancers (97%) |
Melanoma malignum (3%) |
| Cancers (96%) |
Sarcomas |
Lymphomas |
| 1. Epidermis |
seborrhoic wart |
BCC (80%)
SCC (20%) |
|
|
|
| 2. Skin appendages: |
|
|
|
|
|
| • hair follicles |
perifollicular fibroma |
trichoblastic carcinoma |
|
|
|
| • sebaceous glands |
sebaceous adenoma |
sebaceous carcinoma |
|
|
|
| • eccrine sweat glands |
eccrine poroma |
primary eccrine carcinomas (e.g. mucous carcinoma) secondary eccrine carcinomas (e.g. malignant cylindroma) |
|
|
|
| • apocrine sweat glands |
papillary apocrine adenoma |
apocrine carcinoma Paget´s disease |
|
|
|
| 3. Pigment cells |
|
|
|
|
melanoma malignum |
| 4. Mesenchymal structures: |
|
|
|
|
|
| • fibrous tissue |
fibroma |
|
dermatofibrosarcoma protuberans malignant fibrous histiocytoma fibroxanthoma fibrosarcoma |
|
|
| • vascular tissue |
angioma |
|
angiosarcoma Kaposi´s sarcoma |
|
|
| • fat tissue |
lipoma |
|
liposarcoma |
|
|
| • muscle tissue |
leiomyoma |
|
leiomyoma |
|
|
| 5. Nervous tissue |
neurofibroma |
|
neurosarcoma Merkel cell carcinoma |
|
|
| 6. Lymphatic tissue |
|
|
|
granuloma sarcomatodes Sezary´s disease T-cell lymphoma B-cell lymphoma |
|
Other classification of malignancies is as follows:
Non-Melanoma Skin Cancer (NMSC) and Melanoma Malignum (MM.) NMSC include skin cancers (96% of skin malignancies), lymphomas and sarcomas (1% of skin malignancies). The remaining 3% is MM which is characterised by high malignancy and accounts for 75% of all deaths due to skin neoplasms (1, 2).
Despite the fact that there are different classifications available in literature, one thing that does not change is that cancer is the most common histopathological form of malignancies. Almost all skin cancers originate in the epidermis. Cancers arising in the skin appendages constitute a very low per cent. From a histological point of view the epidermis is stratified epithelium with several layers of cells. Depending on an epidermal layer (the basal cell or squamous cell layer) skin cancers are divided into:
? Basal Cell Carcinoma (BCC) originating from the basal cell layer of the epidermis and sheaths of hair follicles. It constitutes 80% of all skin cancers.
? Squamous Cell Carcinoma (SCC) originating from cells in the Malpighi layer and it accounts for 20% of skin cancers (1).
Although both BCC and SCC originate from the epidermis, their biology is completely different and therefore they cannot be discussed together. For that reason the authors of this review have decided to present problems associated only with one of them, namely BCC.
BCC is of the most common human malignancies, and its incidence has been rising within the last decade (3). Although it is not life-threatening, its local malignant features, especially in the area of the face may cause significant functional and aesthetic disturbances what has a profound effect on the quality of life of patients. If BCC is left untreated, it can infiltrate not only adjacent tissues but also bones and even deeper structures like brain (4). Moreover, extremely rare distant metastases of this neoplasm have been described (5).
BCC diagnostics and treatment is managed by physicians of different specialities (dermatologists, plastic surgeons, general surgeons, oncologists, ophthalmologists, ENT specialists, and even general medicine specialists) who promote therapeutic options which are closely related to their specialities and are often controversial. Available literature reports different therapeutic methods including non-invasive techniques such as local application of Imiquimod-containing ointments (6) photodynamic therapy (7), radiation therapy (8), CO2 laser ablation (9),cryosurgery (10), cautery (11) and curettage (12) or surgical excision of a lesion with a margin of clinically normal surrounding tissues (13).
Functional and aesthetic results, treatment efficacy, side effects and effects on the quality of life are different and depend on the method that has been used.
Method selection depends on lesion morphological features and patient´s condition and preferences. The majority of methods to treat BCC described so far may be used only in some, highly selected cases. The only universal method that can be used to treat all cases of BCC is surgical excision with a margin of clinically normal surrounding tissues.
Due to high efficacy of this method, its versatility, good functional and aesthetic results, low risk of complications, availability, low costs, and what is the most important, the ability of postoperative histological assessment of excision completeness, surgical treatment is currently the most common method to treat BCC.
As there is no unanimous algorithm how to manage this condition and as publications casting a new light on the problem of BCC treatment have been published within the last several years the authors of this review have decided to make an attempt to systematise current views on epidemiology, etiopathogenesis, diagnostics and treatment of this neoplasm, with special attention drawn to surgical treatment.
Epidemiology
Among all human malignancies skin cancer occurs the most frequently and it accounts for almost 1/3 of all detectable neoplasms (1). Despite the fact that since the early 1990s the global incidence of neoplasms has been decreasing the rate of incidence of skin cancer has been rising and it is estimated to be 10-15% annually, what is almost ten times higher than the population growth rate (1, 15, 16). It has to be emphasised that there are no precise records especially with regard to BCC and therefore epidemiological data are often understated and not included in global lists of incidence rates of neoplasms (1).
Only in the USA, more than one million cases of skin cancers are detected every year (17). It is the number almost equal to the number of all other cancers detected annually in this country (18). It is estimated that one out of five Americans will develop skin cancer (17), and in almost half of 65-year-olds this cancer will occur at least once in their lives (19).
Although the data presented above regard all cases of skin cancers it can be assumed that they reflect BCC epidemiology to a large extent, as BCC accounts for almost 80% of all cases of skin cancers (1).
Global statistics unanimously indicate that BCC is one of the most common neoplasms in Europe, Australia (20) and the USA (21), and the number of new cases is increasing every year (tab. 2).
Table 2. Incidence rate for selected neoplasms based on Globocan 2002 (number of detected cases/year/100 000 citizens) (22).
| Country |
Non-melanoma skin cancers (w/m) |
Lung cancer (w/m) |
Colon cancer (w/m) |
Breast cancer (w) |
| Finland |
399/416 |
20/55 |
43/41 |
137 |
| Switzerland |
433/560 |
26/78 |
54/75 |
137 |
| Netherlands |
402/470 |
29/92 |
57/62 |
130 |
| United Kingdom |
458/471 |
51/83 |
55/66 |
135 |
| USA |
459/534 |
59/84 |
55/60 |
144 |
| Australia |
403/483 |
27/57 |
58/67 |
114 |
| Poland |
320/382 |
23/104 |
40/41 |
73 |
Polish epidemiological data are also alarming despite the fact that they are not complete due to lack of precise records (fig. 1).
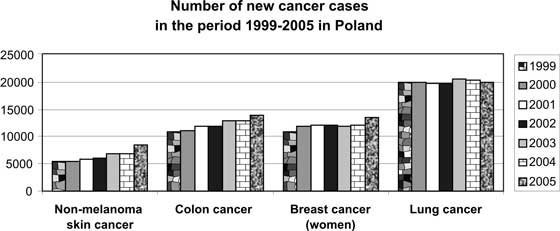
Fig. 1. Number of new cancer cases in the period 1999-2005 in Poland (source: Krajowy Rejestr Onkologiczny) (23).
Epidemiology of skin cancer unanimously indicates that it is a significant global problem. However, this problem is not noticed and is underestimated especially because of the fact that mortality related with these neoplasms is low (in the USA 1000-2000 patients/year) (24) and that this neoplasm is not listed in incidence records (data are not complete.) The importance of this problem is mainly affected by the number and dynamics of new cases, a high recurrence rate (even 18%) (25) and generally high costs of treatment (in the USA the costs of treating skin cancers were more than one billion dollars in 2004) (26).
Etiopathogenesis
Although currently several factors are suspected to be responsible for BCC the most important roles in cancerogenesis are played by UV radiation and advanced age of patients. They account for more than 90% of neoplastic lesions (27).
Aetiological factors
Solar radiation (UV) can be divided into three parts depending on the wavelength: UVA (wavelength of 320-400 nm), UVB (wavelength of 280-320 nm) and UVC (wavelength of 200-280 nm) (1).
The majority of radiation emitted by the Sun is absorbed by the ozone layer of the atmosphere, and consequently, only a low amount reaches the Earth. As due to atmosphere pollution the thickness of the ozone layer is gradually reduced, more and more UV radiation reaches the Earth, therefore the incidence of BCCs can increase (28). This phenomenon may also explain the fact that this neoplasm occurs in younger and younger patients as they earlier achieve a cancerogenesis threshold of Average Accumulated Exposure (29).
Exposure to UVB radiation contributes to the BCC development the most (30).
Contrary to common opinions short-term but intensive and long-term but less-intensive exposure are equal (31). The dose of UV absorbed in the childhood does not contribute significantly to neoplasm pathogenesis according to the latest reports (32).
Long-term UVB actions lead to the formation of mutagenic photoproducts that damage DNA chains in skin cells. DNA damaged in this way is repaired within 24 hours as a result of the effective repair system called NER (nucleotide excision repair system) (33). Impairment of this system present in patients with xeroderma pigmentosum inevitably leads to multifocal skin cancer and death at a young age.
Apart from damage to DNA of skin cells UV radiation also causes mutations in a suppressor gene of the p-53 protein. The protein coded by this gene has anti-oncogenic properties as it induces apoptosis in the cells with damaged DNA. As a result of a mutation in this gene the anti-oncogenic properties of the p-53 protein are turned off, therefore the cells with damaged DNA proliferate without control. The presence of a mutation in the p-53 gene is found in 60-100% of cases of skin cancer (34).
The skin inflammatory response induced during the exposure to UV also participates in the process of damaging DNA (35) inducing disturbances of division and mutations in newly produced cells (36).
The sources of UV radiation include not only solar radiation but also PUVA lamps used to treat psoriasis and albinism as well as tanning lamps.
Due to the fact that tanning lamps are widely accessible and due to fashion trends they have become especially important in BCC etiopathogenesis in the last years. Some of these modern tanning lamps may emit radiation doses which are even 12 times higher than the ones emitted by the Sun (37). The risk of BCC in subjects using tanning lamps regularly is twice the risk observed in the general population (38). The significance of solar radiation in BCC pathogenesis is emphasised by the fact that this neoplasm is found on the skin areas with the most exposure to sunlight, such as the head and neck (85% of all lesions, including 30% within the nose (39, 40).
Exposure to some chemicals
It is claimed that such substances as arsenic, wood tar, gas pitch, synthetic antimalarial agents or psoralens participate in BCC cancerogenesis (1).
Risk groups
Age. A peak in the incidence is between 60 and 80 years of age. More than 95% of patients are patients above 65 years old (1) although recently it has been observed that the incidence in the population below 40 years old is growing (29). With age the total period of UV radiation exposure (Average Accumulated Exposure) increases, therefore when a given threshold is exceeded cancerogenesis processes are initiated. Moreover, the reduced immunity and reduced DNA repair and regeneration properties occurring in the elderly also contribute to the increasing incidence of BCC in this age group (41).
Sex. Men slightly more frequently suffer from BCC (M/F ratio 1.2) (42). It is probably associated with higher exposure to UV radiation what is a result of different working conditions (usually outdoors) and rarer use of sun screens in the case of men (43). It should be noted that during the last 30 years the rate of women below 40 years of age suffering from BCC has tripled (2).
Race. People with fair skin type, light blue and grey eyes and with light red and fair hair suffer from BCC more frequently than people with dark skin (43, 44, 45).
Fair hair, frequent sunburn and freckles in the childhood are features which are especially associated with BCC development (46).
Previous BCC treatment. After the first case of BCC in one´s life the probability of the second one increases ten times (47). In 50% of patients with previously diagnosed BCC other foci will form within 5 years since the first manifestation (42). It is estimated that the likelihood of BCC in such patients is almost 140 times higher that the one in the general population (48).
Post-organ transplant patients. In post-organ transplant patients BCC is the most frequent neoplasm and accounts for 35% of all neoplasms occurring in this group of patients. It is estimated that post-organ transplant patients suffer from BCC 65-250 times more frequently than the general population. BCC most frequently occurs in patients after heart and renal transplantation (49).
Higher incidence of BCC in this group is associated with long-term immunosuppressive therapy that reduces the number of CD4 + cells (50).
The pharmacological immunosuppression combined with UVB radiation that additionally reduces the number of Langerhans cells (immune properties) leads to increased immunosuppression in the skin and increases the risk of neoplasm (51).
Leukaemic patients due to immune system dysfunctions are the next group at a risk. BCC most frequently occurs in patients with chronic lymphocytic leukaemia (8-13 times more frequently than in the general population) (52).
Other risk groups. According to recent opinions risk factors that have not been so widely studied include genetic predisposition (53, 54) freckles (55) and rich-fat diet (56) which is low in antioxidants and vitamins (58).
Precancerous lesions
Precancerous lesions are morphologically changed tissues (a pathological process) on the basis of which neoplasm develops more frequently than in other unchanged tissues (1).
BCC most frequently develops in the skin without previous lesions. It can significantly more rarely (contrary to SCC) develop on the basis of precancerous lesions. Precancerous lesions leading to BCC development include:
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
24 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
59 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
119 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 28 zł
Piśmiennictwo
1. Kordek R et al.: Onkologia. Podręcznik dla studentów i lekarzy. Wyd. II: Via Medica; 2004. p. 191-196.
2. The Burden of Skin Cancer. National Center for Chronic Disease Prevention and Health Promotion. 13 May 2008.
3. Preston DS, Stern RS: Nonmelanoma cancers of the skin. N Engl J Med 1992; 327: 1649-62.
4. Franchimont C: Episodic progression and regression of basal cell carcinomas. Br J Dermatol 1982; 106: 305-10.
5. Lo JS et al.: Metastatic basal cell carcinoma: report of twelve cases with a review of the literature. J Am Acad Dermatol 1991; 24: 715-19.
6. Marks R et al.: Imiquimod 5% cream in the treatment of superficial basal cell carcinoma: results of a multicenter 6-week dose-response trial. J Am Acad Dermatol 2001; 44: 807-13.
7. Clark C et al.: Topical 5-aminolaevulinic acid photodynamic therapy for cutaneous lesions: outcome and comparison of light sources. Photodermatol Photoimmunol Photomed 2003; 19: 134-41.
8. Kwan W, Wilson D, Moravan V: Radiotherapy for locally advanced basal cell and squamous cell carcinomas of the skin. Int J Radiat Oncol Biol Phys 2004; 60: 406-11.
9. Nouri K et al.: Ultrapulse CO2 used for the successful treatment of basal cell carcinomas found in patients with basal cell nevus syndrome. Dermatol Surg 2002; 28: 287-90.
10. Giuffrida TJ, Jimenez G, Nouri K: Histologic cure of basal cell carcinoma treated with cryosurgery. J Am Acad Dermatol 2003; 49: 483-6.
11. Spiller WF, Spiller RF: Treatment of basal cell epithelioma by curettage and electrodesiccation. J Am Acad Dermatol 1984; 11: 808-14.
12. Reymann F: 15 years´ experience with treatment of basal cell carcinomas of the skin with curettage. Acta Derm Venereol (Stockh) 1985; 120 (Suppl.): 56-9.
13. Walker P, Hill D: Surgical treatment of basal cell carcinomas using standard postoperative histological assessment. Australas J Dermatol 2006; 47:1-12.
14. Kossard S et al.: Basal cell carcinoma. In: LeBoit P, Burg G, Weedon D, Sarasin A. The WHO classification of tumours. Pathology and genetics of skin tumours. IARC Press, Lyon 2006; 13-19.
15. Cole P, Rodu B: Declining cancer mortality in the United States. Cancer 1996; 78: 2045.
16. Parkin DM, Pisani P, Ferley J: Global cancer statistics. CA Cancer J Clin 1999; 49: 33.
17. American Cancer Society. Cancer Facts & Figures 2008. Atlanta: American Cancer Society 2008.
18. American Cancer Society: http:www.cancer.org (2006).
19. Robinson JK: Sun Exposure, Sun Protection, and Vitamin D. JAMA 2005; 294: 1541-43
20. Gilbody JS, Aitken J, Green A: What causes basal cell carcinoma to be the commonest cancer? Aust J Public Health 1994; 18: 218-21.
21. Miller DL, Weinstock MA: Nonmelanoma skin cancer in the United States: incidence. J Am Acad Dermatol 1994; 30: 774-8.
22. Global Cancer Statistics Globocan 2002: http://www-dep.iarc.fr/
23. Krajowy Rejestr Onkologiczny: http://epid.coi.waw.pl/krn/
24. Jemal A et al.: Cancer statistics, 2003, CA Cancer J Clin 2003; 53: 5.
25. Silverman MK et al.: Recurrence rates of treated basal cell carcinomas. Part 2: curettage-electrodesiccation. J Dermatol Surg Oncol 1991; 17: 720-6.
26. Bickers DR et al.: The burden of skin diseases: 2004. J Am Acad Dermatol 2006; 55: 490-500.
27. Taylor CR et al.: „Photoaging/Photodamage and Photoprotection” J Am Acad Dermatol 1990; 22.
28. Goldsmith LA: Skin effects of fair pollution. Otolaryn. Head and Neck Surg 1996; 114: 217.
29. The skin Cancer Foundation: Skin Cancer Facts 2008: www.skincancer.org
30. Boukamp P: Non-melanoma skin cancer: what drives tumor development and progression? Carcinogenesis 2005; 26: 1657-1667.
31. Marks R et al.: The role of childhood exposure to sunlight in the development of solar keratoses and non-melanocytic skin cancer. Med. J. Aust. 152 (1990): 62-66.
32. Godar DE, Urbach F, Gasparro FP, van der Leun JC. UV Doses of Young Adults. Photochemistry and Photobiology 2003; 77(4): 453-457.
33. Szepietowski J et al.: Mnogie raki kolczystokomórkowe u chorego z łuszczycą. Przegl. Dermatol 1996; 83: 127.
34. Marks R: The epidemiology of non-melanoma skin cancer: who, why and what can we do about it. J Dermatol 1995; 22: 853.
35. Maeda H, Akaike T: Nitric oxide and oxygen radicals in infection, inflammation, and cancer, Biochemistry (Mosc) 1998; 63: 854-865.
36. Hendrix JD, Harry L, Parlette HL: Micronodular basal cell carcinoma. A deceptive histologic subtype with frequent clinically undetected tumor extension. Arch Dermatol 1996; 132: 295.
37. „11th ROC: Ultraviolet Radiation Related Exposures.” 27 January 2005. U.S. Department of Health & Human Services. 15 April 2008.
38. Karagas MR et al.: Use of Tanning Devices and Risk of Basal Cell and Squamous Cell Skin Cancers. J Natl Cancer Inst 2002; 94: 224.
39. McCormack CJ, Kelly JW, Dorevitch AP: Differences in age and body site distribution of the histological subtypes of basal cell carcinoma: a possible indicator of differing causes. Arch Dermatol 1997; 133: 593-6.
40. DeVita VT Jr, Hellman S, Rosenberg SA: Cancer: Principles and Practice of Oncology. 6th Edition, Lippincot 2001: Chapter 41.
41. Pietrzykowska-Chorążak A: Odczyny skórne na promieniowanie UV w zależności od wieku. Przegl Dermatolog 1978; 61: 3.
42. Brodowski R, Lewandowski B: Rak skóry twarzy. Przegląd piśmiennictwa. Przegląd Medyczny Uniwersytetu Rzeszowskiego 2004; 2-3: 224-230.
43. McCarthy EM, Ethridge KP, Wagner RF: Beach holiday sunburn: the sunscreen paradox and gender differences. Cutis 1999; 64: 37-42.
44. Jabłońska S, Chorzelski T: Choroby skóry. Dla studentów medycyny i lekarzy. Wyd. V PZWL 2002; 387-394.
45. Gloster Jr HM, Neal K: Skin cancer in skin of color. J Am Acad Dermatol 2006; 55: 741-760.
46. Bouwes Bavinck JN et al.: The risk of skin cancer in renal transplant recipients in Queensland, Australia. A follow-up study. Transplantation 1996; 61: 715-721.
47. Marcil J, Stern RS: Risk of developing a subsequent nonmelanoma skin cancer in patients with a history of nonmelanoma skin cancer. A critical review of the literature and meta-analysis. Arch. Dermatol 2000; 136(12): 1524-1530.
48. Aston SJ, Beasley RW: Thorne CH. Grabb and Smith´s Plastic Surgery. 5th Edition Philadelphia: Lippincott-Raven; 1997.
49. Jensen P et al.: Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens, J Am Acad Dermatol 1999; 40: 177-186.
50. Viac J et al.: Langerhans cells, inflammation markers and human papillomavirus infections in benign and malignant epithelial tumors from transplant recipients, J Dermatol 1992; 19: 67-77.
51. Parrish JA. Ultraviolet radiation affects the immune system, Pediatrics 1983; 71: 129-133.
52. Manusow D, Weinerman BH: Subsequent neoplasia in chronic lymphocytic leukemia. JAMA 1975; 232: 267-269.
53. Schreiber MM et al.: The risk of developing subsequent nonmelanoma skin cancers. J Am Acad Dermatol 1990; 23: 1114-18.
54. Gailani MR et al.: Relationship between sunlight exposure and a key genetic alteration in basal cell carcinoma. J Natl Cancer Inst 1996; 88: 349-54.
55. Gilbody JS, Aitken J, Green A: What causes basal cell carcinoma to be the commonest cancer? Australian Journal of Public Health 1994; 18: 218-21.
56. Zak-Prelich M, Narbutt J, Sysa-Jedrzejowska A: Environmental risk factors predisposing to the development of basal cell carcinoma. Dermatol Surg 2004; 30: 248-52.
57. Wei Q et al.: Vitamin supplementation and reduced risk of basal cell carcinoma. J Clin Epidemiol 1994; 47: 829-836.
58. Jeacock DA: Dietary fats and antioxidants and BCC incidence. 1998; Uni Qld. MSc.
59. Chicheł A, Skowronek J: Współczesne leczenie raka skóry – dermatologia, chirurgia czy radioterapia? Współczesna Onkologia; 2005; 9-10: 429-435.
60. Marks R, Staples M, Giles GG: Trends in non-melanocytic skin cancer treated in Australia: the second national sutvey. Int J Cancer 1993; 53: 585-590.
61. Braun RP et al.: Three-dimensional reconstruction of basal cell carcinomas. Dermatol Surg 2005; 31: 562-6.
62. Raasch BA, Buettner PG: Multiple nonmelanoma skin cancer in an exposed Australian population. International journal of Dermatology 2002; 41(10): 652-8.
63. Ionesco DN, Arida M, Jukic DM: Metastatic Basal Cell Carcinoma. Four Case Reports, Review of Literature, and Immunohistochemucal Evaluation. Arch Pathol Lab Med 2006; 130(1): 45-51.
64. Weedon D, Wall D: Metastatic basal cell carcinoma. Med J Aust. 1975; 2: 177-179.
65. Domarus H, Stevens PJ: Metastatic basal cell carcinoma. Report of five cases and review of 170 cases in the literature. J Am Acad Dermatol 1984; 10: 1043-60.
66. Randle HW: Basal cell carcinoma: identification and treatment of the highrisk patient. Dermatol Surg 1996; 22: 255-261.
67. Snow SN et al.: Metastatic basal cell carcinoma. Report of five cases. Cancer. 1994; 73: 328-35.
68. Safai B, Good RA: Basal cell carcinoma with metastasis: review of literature. Arch Pathol Lab Med 1977; 101: 327-331.
69. Weinstock MA et al.: Nonmelanoma skin cancer mortality: a population-based study. Arch Dermatol. 1991; 127: 1194-1197.
70. Kricker A et al.: Skin cancer in Geraldton, Western Australia: a survey of incidence and prevalence. Med J Aust 1990; 152: 399-407.
71. Presser SE, Taylor JR: Clinical diagnostic accuracy of basal cell carcinoma. J Am Acad Dermatol 1987; 16: 988-990.
72. Mogensen M, Jemec GB: Diagnosis of nonmelanoma skin cancer/keratinocyte carcinoma: a review of diagnostic accuracy of nonmelanoma skin cancer diagnostic tests and technologies. Dermatol Surg 2007; 33(10): 1158-1174.
73. Costantino D, Lowe L, Brown DL: Basosquamous carcinoma – an under-recognized, high-risk cutaneous neoplasm: case study and review of the literature. J Plast Reconstr Aesthet Surg 2006; 59: 424-8.
74. Bers MH, Berkow R: The Merck Manual of Diagnosis and Therapy. 2001; Section 10 Chapter 126: 1004-1010.
75. The skin Cancer Foundation: Basal Cell Carcinoma: www.skincancer.org
76. Salasche SJ, Amonette RA: Morpheaform basal-cell epitheliomas. A study of subclinical extensions in a series of 51 cases. J Dermatol Surg Oncol 1981; 7: 387-394.
77. Wagner RF, Casciato DA: Skin cancers. [In:] Casciato DA, Lowitz BB, eds.: Manual of Clinical Oncology. 4th ed. Philadelphia: Lippincott 2000; p. 336-373.
78. Wade T, Ackerman AB: The many faces of basal-cell carcinoma. J Dermatol Surg Oncol 1978; 4: 23.
79. McCormack CJ, Kelly JW, Dorevitch AP: Differences in age and body site distribution of the histological subtypes of basal cell carcinoma. A possible indicator of differing causes. Arch Dermatol 1997; 133: 593-596.
80. Australian Cancer Network Management of Non-Melanoma Skin Cancer Working Party. Non-melanoma skin cancer: Guidelines for treatment and management in Australia; 24 October 2002.
81. American Joint Committee on Cancer, AJCC Cancer Staging Manual, 6th ed. New York: Springer-Verlag 2002.

