Niedokrwienie trzustki ze skurczem naczyń we wczesnej fazie ciężkiej postaci ostrego zapalenia trzustki oraz rola ciągłego dotętniczego wlewu inhibitora proteaz i antybiotyku
Pancreatic ischemia with vasospasm in the early phase of severe acute pancreatitis and the role of continuous regional arterial infusion of a protease inhibitor and an antibiotic
In spite of development of medical intensive care, severe acute pancreatitis (SAP) is still a fatal disease with a high mortality rate. Severe acute pancreatitis is characterized by both systemic organ failure and pancreatic and/or peripancreatic necrosis with infection. Unfortunately, pharmacological therapy including anti-inflammatory drugs, antiproteases, and probiotics could not be clarified to having beneficial effects on the course of SAP as expected (1). Prophylactic antibiotics are also controversial in acute necrotizing pancreatitis (ANP) (2). A new strategy of treatment based on pathophysiology of the disease should be mandatory.
We have clarified pancreatic ischemia with vasospasm precedes necrotic change of the pancreas in the early phase of acute necrotizing pancreatitis (3). Microcirculatory derangement due to ischemia and an increase in intravascular coagulation associated with SAP may easily cause microthrombi formation resulting occlusion of microvessels and necrosis. Therefore, a therapeutic strategy to inhibit the intravascular coagulant activity in the pancreas may be expected to prevent evolution of pancreatic ischemia into necrosis. Nafamostat mesilate is a potent serine-protease inhibitor. In addition, it acts as an anticoagulant as well as antiprotease (4, 5, 6). Regional arterial infusion technique can provide enough amount of nafamostat to the ischemic area of the pancreas. Early initiation of continuous regional arterial infusion (CRAI) of a protease inhibitor and an antibiotic in ANP has been recommended in Japanese guidelines (7). Recently, Piascik et al reported that CRAI of protease inhibitor nafamostat and antibiotic imipenem was effective in preventing complications and reducing mortality rate in SAP based on a randomized controlled trial (8).
Angiographic appearances of arterial irregularities in acute pancreatitis have already been described since 1960’s (9, 10). However, main reason for angiography was examination of the presence of complications, such as a palpable mass, suspicion of abscess or pseudocyst. Nowadays contrast-enhanced computed tomography (CE-CT) enabled us to detect pancreatic abscess, pseudocyst and pancreatic necrosis easily. In addition, pancreatic ischemia is also detected by CE-CT in the early phase of ANP. Unfortunately, relationship between angiographic abnormalities and pancreatic ischemia and/or necrosis on CE-CT findings has never been discussed in the literature.
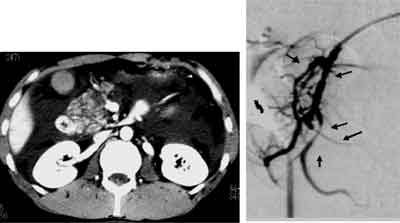
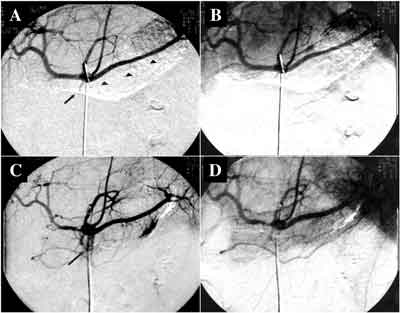
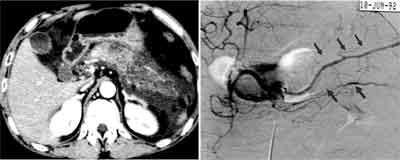
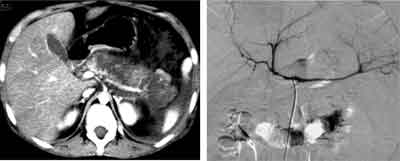
Angiographic features in the early phase of acute necrotizing pancreatitis were investigated in a few Japanese medical centers using digital subtraction angiography with selective catheterization into the celiac artery and superior mesenteric artery (3, 11, 12). In the early phase of ANP, angiographic appearances with irregularities in various grades were observed. Angiographic abnormalities such as sudden tapering, alternative narrowing and dilatation, angulations and diffuse caliber irregularities of the intrapancreatic branch arteries are characteristic of ANP. Severe ischemic change with diffuse vasospasm and/or obstruction was observed not only in the intra- and peripancreatic arteries, but also in the extrapancreatic arteries such as the superior mesenteric artery and its branches (3). Arterial irregularities with vasospasm were already visible on the initial angiography at 12 hours after the onset. Figure 1 shows a typical appearance of vasospasm on angiography. CE-CT revealed a heterogeneous enhancement in the pancreatic head. Selective angiography via the gastroduodenal artery showed alternative narrowing and dilatation and sudden tapering of the intrapancreatic branch arteries consistent with the poorly perfused area of the pancreas on CE-CT. Improvement of ischemic change with angiographic irregularities of the intrapancreatic branch arteries were also observed on follow-up angiography (fig. 2). These findings strongly suggested that the irregular narrowing of the arteries was not caused by structural or irreversible change due to postinflammatory fibrous adhesion but was caused by vasospasm. The extent of ischemic changes with vasospasm on angiographic appearance was correlated with the extent of the poorly perfused area of the pancreas on CE-CT findings and the severity of acute pancreatitis (fig. 3, 4). Non-occlusive mesenteric ischemia (NOMI) associated with SAP was also investigated based on the angiographic appearance by Hirota et al. (13) Severe vasospastic changes of both the celiac artery and the superior mesenteric artery were observed simultaneously in patients with NOMI accompanied by ANP. Severe ischemic changes in the intraabdominal vessels may aggravate microcirculation not only in the pancreas but also in the abdominal viscera leading to pancreatic necrosis and systemic organ failure.
Fig. 1. Vasospasm on angiographic appearance in a patient with acute necrotizing pancreatitis 12 hours after the onset. CE-CT revealed a heterogeneously perfused area of the pancreatic head. Selective angiography via the gastroduodenal artery demonstrated vasospasm such as sudden tapering (short arrow) and alternative narrowing and dilatation (long arrow) of the intrapancreatic branch arteries, consistent with the poorly perfused area on CE-CT.
Fig. 2. Changes of the vasospasm in acute pancreatitis. Initial angiography on admission (5 days after the onset) revealed sudden tapering of the dorsal pancreatic artery (short arrow) and impaired visualization of the intrapancreatic branch arteries (arrowhead) (A) and poor staining of the pancreatic parenchyma in the capillary phase (B). The patient was treated with CRAI therapy. Two weeks later, a follow-up angiography was carried out and it demonstrated improvement of the filling of the dorsal pancreatic artery (long arrow) and intrapancreatic branch arteries (C). Pancreatic gland was also stained homogenously in the capillary phase (D).
Fig. 3. A typical appearance of vasospasm on angiography. CE-CT showed a poorly perfused area in the pancreatic body and tail. Angiography showed diffuse caliber irregularities with narrowing in the intrtapancreatic branch arteries (long arrow). Focal ischemic change with diffuse narrowing of the splenic artery was also observed (46 hours after the onset).
Fig. 4. Severe ischemic change with diffuse narrowing of the extrapancreatic arteries. Angiography demonstrated diffuse narrowing of the splenic artery and common hepatic artery and impaired visualization of the ramifications (arrowhead), consistent with the poorly perfused area on CE-CT (56 hours after the onset).
1. Bang UC, Semb S, Nojgaard C et al.: Pharmacological approach to acute pancreatitis. World J Gastroenterology 2008; 21: 2968-2976.
2. Banks PA, Freeman ML: Practice guidelines in acute pancreatitis. Am J Gastroenterol 2006; 101: 2379-2400.
3. Takeda k, Mikami Y, Fukuyama S et al.: Pancreatic ischemia associated with vasospasm in the early phase of human acute necrotizing pancreatitis. Pancreas 2005; 30: 40-49.
4. Fujii S, Hitomi Y: New synthetic inhibitors of C1r, C1 esterase, thrombin, plasmin, kalikrein and trypsin. Biochim Biophys Acta 1981; 661: 342-345.
5. Aoyama T, Ino Y, Ozeki M et al.: Pharmacological studies of FUT-175, nafamostat mesialte. I Inhibition of protease activity in vitro and in vivo experiments. Jpn J Pharmacol 1984; 35: 203-227.
6. Iwaki M, Ino Y, Motoyoshi A et al.: Pharmacological studies of FUT-175, nafamostat mesialte. V. Effects on the pancreatic enzymes and experimental acute pancreatitis in rats. Jpn J Pharmacol 1986; 41: 155-162.
7. Hirota M, Takada T, Kitamura N et al.: Fundamental and intensive care of acute pancreatitis. J Hepatobiliary Pnacreatic Sci 2010; 17: 45-52.
8. Piascik M, Rydzewska G, Milewski J et al.: The results of severe acute pancreatitis treatment with continuous regional arterial infusion of protease inhibitor and antibiotic: A randomized controlled study. Pancreas 2010 (in press).
9. Reuter SE, Redman HC, Joseph RR: Angiographic findings in pancreatitis. Am J Roentgenol Radium Ther Nucl Med 1969; 107: 56-64.
10. Rosch J, Bret J: Arteriography of the pancreas. Am J Roentogenol Radium Ther Nucl Med 1965; 94: 182-193.
11. Inoue K, Hirota M, Beppu T et al.: Angiographic features in acute pancreatitis: The severity of abdominal vessel ischemic change reflects the severity of acute pancreatitis. JOP J Pancreas (Online) 2003; 4: 207-213.
12. Yasuda T, Takeyama Y, Ueda T et al.: Nonocclusive visceral ischemia associated with severe acute pancreatitis. Pancreas 2003; 26: 95-97.
13. Hirota M, Inoue K, Kimura Y et al.: Non-occlusive mesenteric ischemia and its associated intestinal gangrene in acute pancreatitis. Pancreatology 2003; 3: 316-322.
14. Toung T, Reilly PM, Fuh KC et al.: Mesenteric vasoconstriction in response to hemorrhagic shock. Shock 2000; 13: 267-273.
15. Gilsdorf RB, Long D, Moderg A: Central nervous system influence on experimentally induced pancreatitis. JAMA 1965; 192: 394-397.
16. Dumont AS, Dumont RJ, Chow MM et al.: Cerebral vasospasm after subarachnoid hemorrhage: putative role of inflammation. Neurosurgery 2003; 53: 123-133.
17. Yanamoto H, Kikuchi H, Okamoto S: Effects of protease inhibitor and immunosuppressant on cerebral vasospasm after subarachnoid hemorrhage in rabbits. Surg Neurol 1994; 41: 382-387.
18. Liu XH, Kimura T, Ishikawa H et al.: Effect of endothelin-1 on the development of hemorrhagic pancreatitis in rats. Scand J Gastroenterol 1995; 30: 276-282.
19. Ahn YM, Gajdusek C, London S et al.: Sustained arterial narrowing after prolonged exposure to perivascular endothelin. Neurosurgery 2002; 50: 843-848.
20. Foitzik T, Faulhaber J, Hubert G et al.: Endothelin receptor blockade improves fluid sequestration, pancreatic capillary blood flow, and survival in severe experimental acute pancreatitis. Ann Surg 1998; 228: 670-675.
21. Jenius H, Beall A, Woodrum D et al.: Thrombin-induced vasospasm: cellular signaling mechanism. Surgery 1998; 123: 46-50.
22. Knoepp L, Bagwell CD, Carlich P et al.: Thrombin contraction of vascular smooth muscle: implications for vasospasm. Int J Surg Investig 1999; 1: 285-290.
23. Eto M, Barandier C, Rathgeb L et al.: Thrombin suppresses endothelial nitric oxide synthase and upregulates endothelin-converting enzyme-1 expression by distinct pathways: role of Rho-Rock and mitogen-activated protein kinase. Circ Res 2001; 89: 583-590.
24. Inoue K, Hirota M, Kimura Y et al.: Further evidence for endothelin as an important mediator of pancreatic and intestinal gangrene in acute pancreatitis. Pancreatology 2003; 3: 316-322.
25. Kaller H, Patzschke K, Wegner LA et al.: Pharmacokinetic observations following intravenous administration of radioactively labeled aprotinin in volunteers. Eur J Drug Metab Pharmacokinet 1965; 3: 79-85.
26. Yamauchi J, Takeda K, Shibuya K et al.: Continuous regional applications of protease inhibitor in the treatment of acute pancreatitis. An experimental study using closed duodenal obstruction model in dogs. Pancreatology 2001; 1: 662-667.
27. Mikami Y, Takeda K, Matsuda K et al.: Rat experimental model of continuous regional arterial infusion of protease inhibitor and its effects on severe acute pancreatitis. Pancreas 2005; 30: 248-253.
28. Hayashi J, Kawarada Y, Isaji S et al.: Therapeutic effects of continuous intraarterial antibiotic infusion in preventing pancreatic infection in experimental acute necrotizing pancreatitis. Pancreas 1996; 13: 184-192.
29. Takagi K, Isaji S: Therapeutic efficacy of continuous arterial infusion of an antibiotic and a protease inhibitor via the superior mesenteric artery for acute pancreatitis in an animal model. Pancreas 2000; 21: 279-289.
30. Takeda K, Matsuno S, Sunamura M et al.: Continuous regional arterial infusion of protease inhibitor and antibiotics in acute necrotizing pancreatitis. Am J Surg 1996; 171: 394-398.
31. Takeda K, Matsuno S, Ogawa M et al.: Continuous regional arterial infusion (CRAI) therapy reduces the mortality rate of acute necrotizing pancreatitis: results of a cooperative survey in Japan. J Hepatobiliary Pancreat Surg 2001; 8: 216-220.
32. Imaizumi H, Kida M, Nishimaki H et al.: Efficacy of continuous regional arterial infusion of a protease inhibitor and antibiotic for severe acute pancreatitis in patients admitted to an intensive care unit. Pancreas 2004; 28: 369-373.
33. Yasuda T, Ueda T, Takeyama Y et al.: Treatment strategy against infection: clinical outcome of continuous regional arterial infusion, enteral nutrition, and surgery in severe acute pancreatitis. J Gastroenterol 2007; 42: 681-689.
34. Shirai K, Tanjou K, Shinohara K et al.: The effectiveness of continuous regional arterial infusion therapy with meropenem in severe acute pancreatitis compared with imipenem: A prospective, randomized study. Jpn J Gstroeneterol Surg 2008; 41: 1-11.
35. Ino Y, Arita Y, Akashi T et al.: Continuous regional arterial infusion therapy with gabexate mesilate for severe acute pancreatitis. World J Gastroenterol 2008; 14: 6382-6387.
36. Anai H, Sakaguchi H, Uchida H et al.: Continuous arterial infusion therapy for severe acute pancreatitis: correlation between CT arteriography and therapeutic effect. J Vasc Interv Radiol 1999; 10: 1335-1342.
37. Takeda K, Yamauchi J, Shibuya K et al.: Benefit of continuous regional arterial infusion of protease inhibitor and antibiotic in the management of acute necrotizing pancreatitis. Pancreatology 2001; 1: 668-673.
38. Ishikawa T, Imai M, Kamimura H et al.: Therapeutic efficacy of continuous arterial infusion of the protease inhibitor and the antibiotics and via celiac and superior mesenteric artery for severe acute pancreatitis – pilot study. Hepatogastroenterology 2009; 56: 524-528.
39. Fukuyama S, Takeda K, Matsuda K et al.: Does intra-arterial infusion therapy ameliorate pancreatic perfusion? J Abdom Emerg Med 2005; 25: 633-636.
40. British Society of Gastroenterology: United Kingdome guidelines for the management of acute pancreatitis. Gut 1998; 42: (suppl. 2): s1-s13.
41. Bize PE, Platon A, Becker CD et al.: Perfusion measurement in acute pancreatitis using dynamic perfusion MDCT. AJR Am J Roentgenol 2006; 186: 114-118.
42. Tsuji Y, Yamamoto H, Yazumi S et al.: Perfusion computerized tomography can predict pancreatic necrosis in early stages of severe acute pancreatitis. Clin Gastroenterol Hepatolo 2007; 5: 1484-1492.
43. Takeda K, Kimura K, Sato A: Dignosis of acute necrotizing pancreatitis by perfusion CT. J Jpn Panc Soc 2007; 22: 547-555.