© Borgis - Postępy Nauk Medycznych 4/2014, s. 216-220
*Katarzyna Drabko1, Anna Raciborska2, Katarzyna Bilska2, Jan Styczyński3, Marek Ussowicz4, Marta Choma1, Beata Wójcik1, Agnieszka Zaucha-Prażmo1, Ewa Gorczyńska4, Szymon Skoczeń5, Wojciech Woźniak2, Jolanta Goździk5, Jerzy Kowalczyk1
Analiza przyczyn niepowodzeń leczenia dzieci z mięsakiem Ewinga w Polsce w latach 1999-2006 według programu EURO-EWING 99
Causes of treatment failure in children with Ewing’s sarcoma in Poland treated with EURO-EWING 99 program (1999-2006)
1Department of Pediatric Hematology, Oncology and Transplantology, Medical University, Lublin
Head of Department: prof. Jerzy Kowalczyk, MD, PhD
2Department of Pediatrics and Adolescent Oncological Surgery, Institute of Mother and Child, Warszawa
Head of Department: Magdalena Rychłowska-Pruszyńska, MD, PhD
3Department of Pediatric Hematology and Oncology, Collegium Medicum, Nicolaus Copernicus University, Bydgoszcz
Head of Department: prof. Mariusz Wysocki, MD, PhD
4Department of Pediatric Bone Marrow Transplantation, Hematology and Oncology, Medical University, Wrocław
Head of Department: prof. Alicja Chybicka, MD, PhD
5Transplantation Centre, University Children’s Hospital, Kraków
Head of Department: Jolanta Goździk, MD, PhD
Streszczenie
Wstęp. Wyniki leczenia mięsaka Ewinga u dzieci i młodzieży są niezadowalające i konieczna jest współpraca wielodycyplinarna w celu poprawy losu tych pacjentów.
Cel pracy. Retrospektywna analiza niepowodzeń leczenia dzieci i młodzieży z mięsakiem Ewinga leczonych w polskich ośrodkach onkologii dziecięcej.
Materiał i metody. Analizą objęto grupę 119 pacjentów w wieku od 1,5 miesiąca do 19 lat z mięsakiem Ewinga, leczonych w latach 1999-2006 według programu EURO-EWING 99 w pięciu ośrodkach onkologii i hematologii dziecięcej oraz chirurgii onkologicznej. Pacjentami wysokiego ryzyka określono tych 78 chorych, u których stwierdzono co najmniej jeden z następujących czynników: obecność odległych przerzutów przy rozpoznaniu choroby, brak możliwości resekcji ogniska pierwotnego lub zła odpowiedź histologiczna na chemioterapię przedoperacyjną.
Wyniki. W badanej grupie zmarło 54 chorych. Większość zgonów u dzieci z mięsakiem Ewinga (50/54, 92%) była spowodowana brakiem remisji lub nawrotem choroby. U 4 pacjentów przyczyny śmierci były związane z leczeniem (8%). W grupie chorych z obecnymi czynnikami ryzyka wznowę stwierdzono u 44/78 (56%) pacjentów i 11/41 (27%) chorych z grupy standardowego ryzyka. Z powodu progresji choroby zmarło też 11 chorych, którzy nie odpowiedzieli na terapię I linii i ich leczenie nie było dalej prowadzone według programu EURO-EWING 99.
Wnioski. Niemal wszystkie niepowodzenia leczenia były spowodowane chorobą, co w naszej opinii uzasadnia intensyfikację terapii u pacjentów wysokiego ryzyka w pierwszej remisji, ze starannym monitorowaniem późnych skutków tego leczenia. Wykorzystanie megachemioterapii u pacjentów z czynnikami ryzyka nie zwiększyło liczby zgonów z powodu toksyczności i przyczyniło się do zmniejszenia liczby nawrotów w tej grupie pacjentów. Nowe metody, w tym terapie eksperymentalne są niezbędne dla pacjentów z mięsakiem Ewinga, którzy nie reagują na leczenie indukcyjne.
Summary
Introduction. Results of treatment of Ewing’s sarcoma in children and adolescents are unsatisfactory and multidisciplinary efforts are necessary to improve the outcome of this patients.
Aim. Retrospective analysis of treatment failure children and adolescence with Ewing’s sarcoma treated in Polish oncology centers.
Material and methods. Study included 119 patients with Ewing’s sarcoma treated in 1999-2006 according to the EURO-EWING 99 program in five pediatric oncology centers. High-risk patients (78) were identified as those, who had at least one of the following factors: the presence of distant metastases at diagnosis, lack of primary tumor resection or poor histologic response to preoperative chemotherapy.
Results. In the study group 54 patients died. Most of the treatment failures in children with Ewing’s sarcoma was caused by the underlying disease: lack of remission or relapse of the disease was the cause of death as many as 50 children (92%). Treatment related death occurred in 4 patients (8%). In high-risk patients relapse was observed in 44/78 (56%) patients and in 11/41 (27%) patients in standard risk group. The progression of the disease was the reason of death in 11 patients who did not respond to first-line therapy and their further treatment was not carried according to EURO-EWING 99.
Conclusions. Almost all treatment failures were due to the underlying disease, in our opinion intensification of therapy in high-risk patients in first remission is justified, with careful monitoring of late effects of this treatment. Use megachemotherapy in some patients with risk factors did not increase the number of deaths due to toxicity and contributed to reducing the number of relapses in these patients. New methods including experimental chemotherapy are required for patients with Ewing’s sarcoma, who not respond to induction treatment.

Introduction
Ewing’s sarcoma (ES) is the second most common malignancy of bone after osteosarcoma, and bone tumors are the eighth most common cancer in childhood after leukemia, CNS tumors, lymphomas, neuroblastoma, soft tissue sarcoma, embryonic tumors and kidney tumors. Incidence of ES is according to various sources from 1.6 to 2.9 new cases per 1 million children per year. More often boys than girls are affected 1.3-1.8 to 1 respectively. Most cases are diagnosed between 15-19 year age group (40%). Polish data from 1995-1999 indicate a higher incidence of bone tumors in children in our country (8.2% of all cancers in children) as compared to the European data (4.2%) (1-5).
ES is a systemic disease, as evidenced by the results of treatment in the years when they were available only local methods: surgery and radiotherapy. The majority (> 90%) described patients despite resection or irradiation of the primary foci died within a few to several months after diagnosis because of distant metastases (6-8).
Nowadays treatment of ES is complex, therapeutic programs are based on clinical trials, and consisted of a combination of preoperative chemotherapy, surgery and consolidation therapy, which is based on risk factors at the time of diagnosis and response to induction therapy. Group of European countries: United Kingdom, Germany, Austria, Netherlands, France, Switzerland and other countries affiliated to the EORTC-STBSG (European Organisation for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group) has prepared a program EURO-EWING 99, which was registered in 2001 and the planned closing date of this Protocol in 2017 (9).
The program EURO-EWING 99, which was also recommended by the Polish Pediatric Solid Tumours Group as standard of care of patients with ES, identified the following prognostic factors, which determines stratification treatment groups of patients:
– the presence of metastases at diagnosis ES,
– location of metastatic (lung or other locations),
– the possibility of surgical resection of the tumor,
– histological response to preoperative chemotherapy,
– the size of the tumor – in the cases without possibility of surgical resection.
Determination of risk factors in Poland also was based on the criteria of the EURO-EWING 99. In the Polish centers decision of consolidation therapy in patients with an unfavorable prognosis was taken by physicians and patients were not randomized for conventional consolidation consisting of cycles of standard chemotherapy (vincristin, actinomycin, and ifosfamide) or megachemotherapy (busulfan and melphalan).
Aim
The aim of this study was a retrospective analysis of treatment failures of children and adolescents with Ewing’s sarcoma treated at Polish pediatric oncology centers.
Material and methods
The analysis of failures of treatment was performed on a group of 119 patients with ES, including 71 boys, treated in five Polish pediatric centers of oncology and hematology: Department of Surgical Oncology Children and Youth, Institute of Mother and Child in Warsaw and departments of Pediatric Oncology and Hematology in Lublin, Wrocław, Bydgoszcz and Kraków. The median age of patients studied at diagnosis was 13 years and 2 months. The youngest patient at diagnosis was 1.5 months and the oldest 19 years and 9 months. The location of primary site of ES in the study group are shown in figure 1. In patients included to the analysis, the presence of following high-risk factors were determined:
– the presence of distant metastases at diagnosis,
– lack of surgical removal of a primary tumour,
– more than 10% viable tumor cells in the primary tumor after preoperative chemotherapy (poor histological response to preoperative chemotherapy).
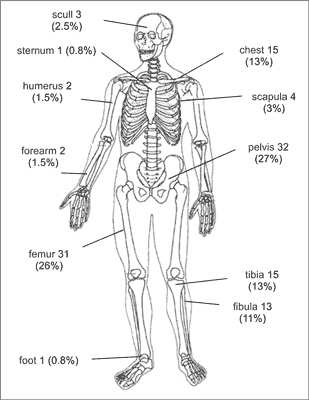
Fig. 1. Primary sites of Ewing’s sarcoma in analyzed group.
Due to the nature of the retrospective analysis, not all patients had sufficient data to determine the volume of the primary tumor, and therefore in this study we not included the tumor volume as a risk factor. Standard risk patients (SR) were defined children who did not have any of these adverse factors. High-risk patients (HR) were determined the patients revealed the presence of at least one of the above mentioned factors of poor prognosis. In the analyzed group of patients 41 children was included to SR group and the other 78 patients were in HR group.
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
24 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
59 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
119 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 28 zł
Piśmiennictwo
1. Kaatsch P: Epidemiology of childhood cancer. Cancer Treat Rev 2010 Jun; 36(4): 277-285.
2. Miller RW, Young JL Jr, Novakovic B: Childhood cancer. Cancer 1995 Jan 1; 75 (suppl. 1): 395-405.
3. Esiashvili N, Goodman M, Marcus RB Jr: Changes in incidence and survival of Ewing’s sarcoma patients over the past 3 decades: Surveillance Epidemiology and End Results data. J Pediatr Hematol Oncol 2008 Jun; 30(6): 425-430.
4. Kowalczyk JR, Dudkiewicz E, Balwierz W et al.: Incidence of childhood cancers in Poland in 1995-1999. Med Sci Monit 2002 Aug; 8(8): CR587-590.
5. Parkes SE, Parke S, Mangham DC et al.: Fifty years of paediatric malignant bone tumours in the West Midlands, UK, 1957-2006: incidence, treatment and outcome. Paediatr Perinat Epidemiol 2010 Sep; 24(5): 470-478.
6. Ewing J: Futher report of endothelial myeloma of bone. Proc N Y Pathol Soc 1924; 24: 93-100.
7. Perez CA, Tefft M, Nesbit ME Jr et al.: Radiation therapy in the multimodal management of Ewing’s sarcoma of bone: report of the Intergroup Ewing’s Sarcoma Study. Natl Cancer Inst Monogr 1981 Apr; (56): 263-271.
8. Patricio MB, Vilhena M, Neves M et al.: Ewing’s sarcoma in children: twenty-five years of experience at the Instituto Portugês de Oncologia de Francisco Gentil (I.P.O.F.G.). J Surg Oncol 1991 May; 47(1): 37-40.
9. http://controlled-trials.com/ISRCTN61438620/61438620, 2014.
10. Drabko K, Raciborska A, Bilska K et al.: Consolidation of first-line therapy with busulphan and melphalan, and autologous stem cell rescue in children with Ewing’s sarcoma. Bone Marrow Transplant 2012 Dec; 47(12): 1530-1534.
11. Delattre O, Zucman J, Melot T et al.: The Ewing family of tumors – a subgroup of small-round-cell tumors defined by specific chimeric transcripts. N Engl J Med 1994; 331: 294-299.
12. Le Deley MC, Delattre O, Schaefer KL et al.: Impact of EWS-ETS fusion type on disease progression in Ewing’s sarcoma/peripheral primitive neuroectodermal tumor: prospective results from the cooperative Euro-EWING 99 trial. J Clin Oncol 2010; 28: 1982-1988.
13. van Doorninck JA, Ji L, Schaub B et al.: Current treatment protocols have eliminated the prognostic advantage of type 1 fusions in Ewing’s sarcoma: a report from the Children’s Oncology Group. J Clin Oncol 2010; 28: 1989-1994.
14. Ladenstein R, Potschger U, Le Deley MC et al.: Primary Disseminated Ewing’s sarcoma: Results of the Euro-EWING 99 Trial. J Clin Oncol 2010; 28: 3284-3291.
15. Drabko K, Zaucha-Prażmo A, Choma M et al.: Megachemioterapia i autologiczne przeszczepienie hematopoetycznych komórek macierzystych u dzieci z mięsakiem Ewinga – doświadczenia polskie. Med Wieku Rozwoj 2008; XII(4), cz. 2: 1069-1073.
16. Drabko K, Zawitkowska-Klaczynska J, Wojcik B et al.: Megachemotherapy followed by autologous stem cell transplantation in children with Ewing’s sarcoma. Pediatr Transplant 2005 Oct; 9(5): 618-621.
17. Zawitkowska-Klaczyńska J, Drabko K, Katski K et al.: Outcome of Ewing’s sarcoma in children and adolescents with Ewing’s sarcoma: a five-year survival from a single institution. Pediatr Hematol Oncol 2004 Oct-Nov; 21(7): 627-633.

