*Małgorzata Matuszczyk, Anna Rybak, Michał Szczepański, Anna Wiernicka, Jarosław Kierkuś
Home Enteral Nutrition in children – 6 years of clinical experience
Żywienie dojelitowe dzieci w warunkach domowych – 6 lat doświadczeń klinicznych
Department of Gastroenterology, Hepatology, Feeding Disorders and Pediatrics, The Children’s Memorial Health Institute, Warsaw
Head of Department: prof. Józef Ryżko, MD, PhD
Streszczenie
Wstęp. Ostatnie lata to czas intensywnego rozwoju terapii żywieniowej w warunkach domowych.
Cel pracy. Celem pracy było przedstawienie doświadczeń własnych w domowym żywieniu dojelitowym (HEN) dzieci.
Materiał i metody. 148 pacjentów włączonych do HEN w okresie od 1 października 2007 r. do 30 września 2013 r. scharakteryzowano pod kątem wieku, wskazań do żywienia dojelitowego, stanu odżywienia, dostępu do przewodu pokarmowego, rodzaju diety, powikłań, czasu trwania oraz przyczyn zakończenia programu.
Wyniki. Najczęstszymi wskazaniami do żywienia dojelitowego były zaburzenia neurologiczne (n = 71) oraz schorzenia gastroenterologiczne (n = 34). W dniu zakwalifikowania do programu niedożywienie stwierdzono u 65% chorych. 60 dzieci żywionych było przez sondę nosowo-żołądkową, 87 przez gastrostomię i jedno dziecko przez jejunostomię. 129 pacjentów otrzymywało przemysłową dietę polimeryczną, w 97 przypadkach wykorzystywano preparaty wysokokaloryczne, a dla 27 dzieci dieta wzbogacana była w błonnik pokarmowy.
U 130 chorych preparaty przemysłowe podawane były w bolusach, 13 pacjentów żywiono metodą wlewu ciągłego, a 5 metodą łączoną. Powikłania żywienia dojelitowego stwierdzono w sumie u 98 dzieci, a do najczęstszych należały: stany zapalne wokół stomii (n = 63), uszkodzenia mechaniczne gastrostomii (n = 53) oraz nietolerancja diety przemysłowej (n = 38). 67 dzieci zakończyło HEN, a najczęstszym powodem było uzyskanie oczekiwanych efektów terapii (n = 29).
Wnioski. HEN jest bezpieczną i dobrze tolerowaną metodą żywienia u dzieci cierpiących na choroby przewlekłe, kiedy skuteczne żywienie doustne nie jest możliwe, lub ze względu na duże zapotrzebowanie na energię i składniki odżywcze wysokie jest ryzyko niedożywienia.
Summary
Introduction. In the last years there was an intensive development of Home Enteral Nutrition (HEN).
Aim. The aim of the study was the presentation of our clinical experience in Home Enteral Nutrition in children.
Material and methods. 148 patients treated in HEN since 1st October 2007 till 30th September 2013 were characterized according to age, indications for enteral nutrition, nutritional status, access to gastrointestinal tract, type of diet, complications, duration of HEN and reasons for discontinuation the Program.
Results. The main indications for enteral nutrition were neurological diseases (n = 71) and gastrointestinal disorders (n = 34). At the time of introducing HEN the undernutrition was stated in 65% of patients. 60 children were fed by nosogastric tube, 87 by gastrostomy and 1 by jejunostomy. 129 patients received the polymeric industrial diet, in 97 cases the hypercaloric formula was used and in 27 the diet was enriched in fiber. Diet was given in bolus in 130 patients, 13 were fed by continuous infusion and 5 by mixed method. The complications of enteral nutrition were reported in 98 patients. The main problems included: peristomal inflammation (n = 63), mechanic damages of gastrostomy (n = 53) and intolerance of industrial diet (n = 38). In 67 children the HEN was interrupted and the main reason was obtaining an anticipated effects of the therapy (n = 29).
Conclusions. HEN is safe and well tolerated method of nutrition for children suffering from chronic diseases, when the effective oral feeding is impossible or the risk of nutritional deficiencies is high due to the higher energy requirements.

Introduction
Treatment with liquid diets delivered trough the other way than oral have its origins in the ancient times (1). In 1598 the first attemps of supplying the liquid food directly into the esophagus were taken (2) and first descriptions of gastrostomy and jejunostomy placement methods have already come from the nineteenth century (3). The placement of gastrostomy by inoperable method – percutaneous endoscopic gastrostomy (PEG) has started in the 70s of the 20th century and was a milestone in the area of enteral nutrition. The second half of the last century was also the time of significant progress in the production of enteral preparations, modifications of their composition and recipes as well as widening the range of their applications in different clinical conditions (1).
According to the ESPGHAN Committee on Nutrition statement, the term of enteral nutrition (EN) includes feeding of patients with specialized industrial preparations, both by the tube/stomy and orally (4). However in Poland, EN is recognized as the administration of enteral diet through any other than oral way (5, 6). This form of therapy is intended for patients with at least partially functioning digestive tract, in whose the effective oral feeding is impossible or the standard diet does not cover the individual requirements for energy and nutrients (1).
Poor nutritional status is one of the most common problem among children suffering from chronic diseases (7). In many cases the inability to provide the adequate amounts of energy and nutrients by oral feeding refers not only to the disease’s exacerbation, but can also be maintained during the reconvalescence period or even through the whole life. In these patients, nutritional therapy has usually the long-term character, contributes to the reduction of the quality of life and generates the high costs of treatment (3). For these reasons, in the 80s of the last century the USA and Canada started the Home Enteral Nutrition (HEN), and since then we observed a rapid development of this kind of nutritional teratment around the whole world (8). Experiences of other centers showed, that in children suffering from chronic diseases HEN is safe and effective method of feeding, that reduces the degree of malnutrition and allows to normal participation in family and social life (3, 4).
Aim
Until recently, in Poland (mostly due to the high costs) the prevalence of HEN was relatively low. However, this procedure has started to be reimbursed by the Public Health Care in 2007 (7) and since then it has become an important alternative for an inpatient enteral nutrition. Over these past six years the significant progress in the area of HEN has been seen in our country. Nevertheless, the availability of the data in this method of therapy among pediatric patients is still very limited. Therefore the aim of the study was the multidimensional analysis of clinical experiences in HEN in children and the assessment of safety and efficacy of this method of nutritional treatmenet.
Material and methods
We retrospectively analyzed the data from all patients younger than 18 years who entered HEN Program in our Department, since its beginning, on the 1st October 2007, till 30th September 2013. A total of 148 patients (75 males) were studied, all by the same nutritional team.
The data (including age, gender, indications for enteral feeding, nutritional status, access to gastrointestinal tract, type of diet and methods of delivery, complications, duration of HEN and causes of any discontinuation the Program) were obtained from the HEN computerized data-base, and from the medical and dietetic charts of all patients. The indications for Home Enteral Nutrition were classified as neurological diseases (including cerebral palsy), genetic congenital malformations, gastrointestinal diseases, cystic fibrosis, hepatic diseases, metabolic disorders and congenital heart malformations. In most patients the nutritional status was assesses based on BMI index reffered to the WHO standards. Diet was given by naso-gastric tube (NG), gastrostomy (GT) or jejunal access (JA). The methods of delivery included: continuous EN (> 16 hours per day), overnight EN (< 12 hours per day), day time bolus EN and day time bolus with overnight EN. For continuous EN peristaltic pumps were used and for overnight EN the industrial diet was given both, in gravity infusion and by pumps. Commercially available diets were analyzed according to 3 features: type of contained protein – polymeric or hydrolyzed-protein paediatric formula, energy density – normocaloric (≤ 1 kcal/1 ml) or hypercaloric (> 1 kcal/1 ml) industrial diet and content of the dietary fiber – with or without addittion of fiber formula. All this products were adapted to the age and disease. The number of patients (in first year of life) were fed with hydrolyzed-protein infant formula because of lack of hydrolyzed-protein industrial diet for patients under first year of life – in this cases diet may have eventually been concentrated or enriched in supplements feeds (like additionally sources of energy/carbohydrate or fat units). Complications of HEN were considered as: peristomal inflammation, mechanic damages, intolerance of industrial diet and constipation. Reasons of discontinuation of the Program were divided into the following groups: obtaining an anticipated effects of therapy, death of the child, intolerance of industrial diet and changing the HEN center.
Results are presented as means and standard deviations.
Results
Population
The mean age of patients at the beginning of HEN was 5.26 years ± 5.52 (the range from 6 weeks to 17 years) with the median of 2.8 years. 42 children (28%) were under 1st year of life and 34 (23%) were older than 10 years. The age varied from 0.66 ± 0.55 in patients with congenital heart malformations to 7.4 ± 6.44 in children with gastrointetinal diseases (fig. 1). Among patients still in HEN on 30th September 2013 (n = 81) the mean age was 7.85 ± 5.76 (the range from 3 months to 18.5 years) with a median of 6.32 years; 6 children (7.4%) were under 1st year of life and 29 (36%) were older than 10 years.
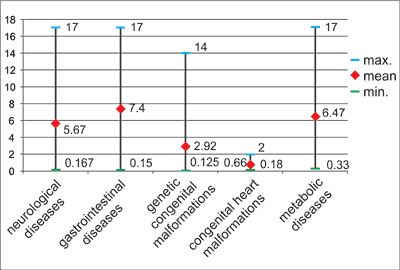
Fig. 1. Mean age of patients at the time of introducing HEN, according to the underlying disease
The mean duration of HEN was 397.31 ± 371.66 days (the range from 10 to 1545 days) with the median of 282 days. 10 children (7%) received HEN for less than 30 days and 28 patients (19%) for more than 2 years. The duration of HEN range from 130.83 ± 96.21 days (the range from 21 to 245 days) among children with metabolic diseases to 498.38 ± 400.89 days (the range from 16 to 1545 days) in patients with neurological diseases (fig. 2).
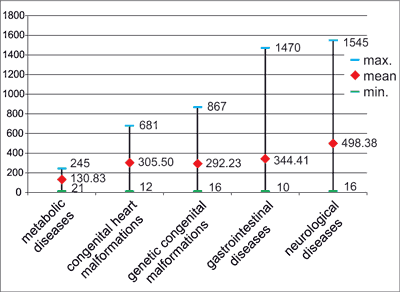
Fig. 2. Mean duration of HEN, according to the underlying disease
Indications
Indications for HEN administration were: neurological diseases in 71 (48%) children, including cerebral palsy in 36 (24%); gastrointestinal disorders in 34 patients (23%); genetic congenital malformations in 27 (18%); congenital heart malformations in 10 (7%) and metabolic diseases in 6 (4%). Most heterogeneous was the group of gastrointestinal diseases and included: cystic fibrosis in 10 children (29%), hepatic diseases in 8 (23%), feeding disorders in 6 (18%), Crohn’s disease in 4 (12%), burned esophagus in 2 (6%), congenital esophageal atresia in 2 (6%) and single cases of esophageal stenosis and recurrent pancreatitis.
Nutitional status
In 96 (65%) of patients commencing HEN the undernitrition was stated in 74 cases (77%) based on BMI status referred to the WHO standards (BMI < 3 pc). In the rest of children the health condition resulting from underlying disease did not allow to measure the growth and thus to calculate the BMI index. Among them the nutritional status was assessed using both the information included in medical charts (that are filled up for each patient on the day of the HEN introduction) and the laboratory tests. Most often the undernutrition was confirmed among patients with congenital heart malformations and metabolic diseases (n = 9; 90% and n = 5; 83%, respectively). Also in 3 other groups the poor nutritional status constituted the important problem in more than the half of children – malnutrition was claimed in 22 patients with gastrointestinal diseases (65%), 17 with congenital genetic malformations (63%) and 39 who suffer from neurological disorders (55%).
Made of administration
At the time of introducing HEN the industrial diet was delivered by NG in 60 (40.5%) children, GT in 80 (59%) and JA in 1 (0.5%). NGT was the most common enteral access among patients with congenital heart malformations (n = 8; 80%), whereas GT was preferred in children with neurological diseases (n = 52; 73%). In the three remaining groups the equal distribution on children fed by NG and GT was observed (fig. 3). Initially, only one boy with Down’s syndrome was fed by the jejunal acces.
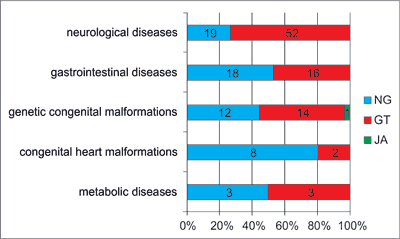
Fig. 3. Type of enteral access, according to the underlying disease
In the group of children commencing HEN with NG, in 34 (57%) cases this kind of enteral access was used during the whole time of treatment in the Program. The NG was sufficient method of feeding in around half of patients with gastrointestinal disorders and congenital heart malformations (n = 16; 47% and n = 3; 50%, respectively). It was also an only way of food administration among 2 children with metabolic diseases (33%) and 6 patients with congenital genetic malformations (22%). In children with neurological disorders NG was used during the entire time of terapy only in 5 children (7%). The mean duration of HEN in this group of patients was 105.09 ± 98.01 days (the range from 10 to 338 days) with the median of 56 days. The duration of HEN range from 83.5 ± 88.39 days in children with metabolic diseases to 144.4 ± 135.28 in patients suffering from congenital heart malformations (tab. 1). The percentage of cases, in which the feeding by NG was continiued longer than 6 weeks, was 65% (n = 22).
Tab. 1. Profile of patients in whose the NG was used as an only method of diet’s delivery during the whole duration of HEN, according to the underlying disease
| Indication | Total number of patients with NG | Number of patients fed by NG during the whole therapy in HEN | % | Mean duration of HEN | Number of patients fed by NG longer than 6 weeks | % |
| Neurological diseases | 19 | 5 | 26 | 89.00 | 2 | 10.5 |
| Gastrointestinal diseases | 18 | 16 | 89 | 96.63 | 10 | 62.5 |
| Genetic congenital malformations | 12 | 6 | 50 | 115.50 | 5 | 41 |
| Congenital heart malformations | 8 | 5 | 62.5 | 144.40 | 4 | 50 |
| Metabolic diseases | 3 | 2 | 67 | 83.50 | 1 | 33 |
Among 26 (43%) children with NG the GT (PEG in 13 cases and surgical gastrostomy in 13) was placed on average after 108 days from the begining of HEN (the range from 27 to 262 days). Most often the placement of GT was necessary in children with neurological diseases (n = 14; 74%) and genetic congenital malformations (n = 6; 50%). Because of persisting feeding’s problems the further holding of NG was impossible in around one third of patients with congenital heart malformations and metabolic diseases, n = 3 (38%) and 1 (n = 33), respectively. However, only in 11% of children with gastrointestinal diseases, NG was not sufficient type of enteral access during the entire duration of HEN.
The mean time between introduction of HEN with NG and placement of GT was 107.5 ± 76.47 days (the range from 27 to 262 days) with the median of 82.5 days. This time range from 72 days among children suffering from congenital health malformations to 234 days in patients with metabolic diseases (tab. 2). The percentage of cases in which the feeding by NG was continiuing longer than 6 weeks in this group was 80% (n = 20).
Tab. 2. Profile of patients with NG at the day of introducing HEN in whose the GT was placed, according to the underlying disease
| Indication | Total number of patients with NG | Number of patients in whom the gastrostomy was placed | % | Mean time between introduction of HEN and GT placement | Number of patients fed by NG longer than 6 weeks | % |
| Neurological diseases | 19 | 14 | 74 | 100.79 | 9 | 47 |
| Gastrointestinal diseases | 18 | 2 | 11 | 130.5 | 2 | 11 |
| Genetic congenital malformations | 12 | 6 | 50 | 112.17 | 6 | 50 |
| Congenital heart malformations | 8 | 3 | 38 | 72 | 2 | 25 |
| Metabolic diseases | 3 | 1 | 33 | 234 | 1 | 33 |
Diets
Techniques of infusion regime included day time bolus in 130 children (88%), overnight EN in 8 (6%), continuous EN in 5 (3%) and mixed method (day time bolus with overnight EN) in 5 (3%). Day time bolus was the most common way of diet’s delivery in all groups of patients – in 70 children with neurological disorders (99%), 20 with gastrointestinal diseases (59%), 25 with genetic congenital malformations (93%), all children with congenital heart diseases (n = 10) and 5 with metabolic problems (83%). Other methods of diet’s infusion were mainly used in patients with gastrointestinal diseases (n = 14; 41%), and only in single cases in the remaining groups of children (fig. 4). In all patients with continuous EN the peristaltic pumps were used, whereas 2 children (25%) with overnight EN and 4 (80%) with mixed method was fed trough gravity infusion.
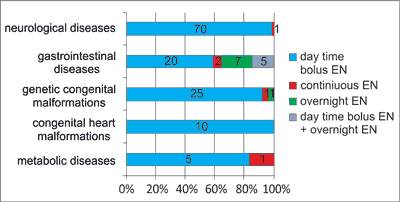
Fig. 4. Type of infusion regime, according to the underlying disease
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
24 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
59 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
119 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 28 zł
Piśmiennictwo
1. Spodaryk M: Podstawy leczenia żywieniowego u dzieci. Wyd. Uniwersytetu Jagiellońskiego, Kraków 2001.
2. Barnabas G: Navigating home Care: Enteral nutrition. Part One. Pract Gastroenterol 2003; 27: 13-35.
3. Pertkiewicz M: Żywienie dojelitowe w warunkach domowych. Post Żyw Klin 2006; 1(1): 5-14.
4. ESPGHAN Committee on Nutrition: Practical Approach to Paediatric Enteral Nutrition: A Comment by the ESPGHAN Committee on Nutrition. JPGN 2010; 15(1): 110-122.
5. Pertkiewicz M, Korta M, Książyk J: Standardy żywienia pozajelitowego i dojelitowego. Wydawnictwo Lekarskie PZWL, Warszawa 2005.
6. Książyk J: Standardy leczenia żywieniowego w pediatrii 2013. Wydawnictwo Lekarskie PZWL, Warszawa 2005.
7. Borkowska A, Szlagatys-Sidorkiewicz A, Zagirski M et al.: Żywienie dojelitowe dzieci w warunkach domowych – doświadczenia kliniczne. Przeg Ped 2010; 40(1): 50-53.
8. Paccagnella A, Baruffi C, Pizzolato D et al.: Home enteral nutrition in adults: A five-year (2001-2005) epidemiological analysis. Clin Nutr 2008; 27: 378-385.
9. Daveluy W, Guimber D, Mention K et al.: Home enteral nutrition in children – an 11-year experience with 416 patients. Clin Nutr 2005; 24: 48-54.
10. Salmon Zaban AL, Garbi Novaes MR: Home enteral nutrition in children: a one-year experience with 184 patients in Distrito Federal, Brazil. Exper Biol Med 2010; 235: 584-589.
11. McCarey DW, Buchanan E, Gregory M et al.: Home enteral feeding of children in the West of Scotland. Scott Med J 1996; 41(5): 147-149.
12. Pedrón-Giner C, Calderón C, Martínez-Zazo A et al.: Home enteral nutrition in children: a 10 year experience with 304 pediatric patients. Nutr Hosp 2012; 27(5): 1444-1450.
13. Szlagatys-Sidorkiewicz A, Popińska K, Toporowska-Kowalska E et al.: Home enteral nutrition in children – 2010 nationwide survey of the polish siciety for clinical nutrition in chiuldren. Eur J Pediatr 2012; 171: 719-723.
14. Diamanti A, Di Ciommo VM, Tentolini A et al.: Home enteral nutrition in children – 14-year multicenter survey. Eur J Clin Nutr 2013; 67: 53-57.
15. Matuszczyk M, Szczepański M, Janowska A et al.: Domowe żywienie dojelitowe dzieci ze schorzeniami gastroenterologicznymi. Post Żyw Klin 2011; 4(20): 25-35.
16. Matuszczyk M: Żywienie w mukowiscydozie. Stand Med Ped 2013; 4: 445-458.
17. Lucendo AJ, de Rezende LC: Importance of nutrition in inflammatory bowel disease. World J Gastroenterol 2009; 15(17): 2081-2088.
18. Ryżko J, Kierkuś J, Ryżko J: Postępowanie dietetyczne i żywieniowe w nieswoistych zapaleniach jelit u dzieci z uwzględnieniem European Crohn’s And Colitis Organisation (ECCO). Pediatr Współcz Gastr Hepat Żyw Dziecka 2011; 13(2): 103-110.
19. Akobeng AK, Thoas AG: Enteral nutrition for maintenance of remission in Crohn's disease. Cochrane database Syst Rev 2007, CD005984.
20. Rosenfeld M, Casey S, Pele M, Ramsey BW: Nutritional effects of long-term gastrostomy feeding in children with cystic fibrosis. J Am Diet Ass 1999; 99(2): 191-194.
21. Conway S, Morton A, Wolfe S: Enteral tube feeding for cystic fibrosis. Cochrane Database Syst Rev 2012, 12, 12: CD001198.
22. Sinaasappel M, Stern M, Littlewood J et al.: Nutrition in patients with cystic fibrosis: a European Consensus. J Cys Fib 2002; 1: 51-75.
23. Milanowski A, Pogorzelski A, Orlik T et al.: Zasady rozpoznawania i leczenia mukowiscydozy. Stanowisko Polskiej Grupy Roboczej Mukowiscydozy. Stand Med Ped 2002; 10: 600-612.
24. Evans S, Daly A, Davies P, MacDonald A: Fibre content ofenteral feeds for the older child. J Hum Nutr Diet 2009; 22(5): 414-421.
25. Pentiuk S, O’Flaherty T, Santoro K et al.: Purred by Gastrostomy Tube Diet Improves Gagging and Retching in Children With Foundoplication. J Parent ent Nutr 2012; 35(3): 375-379.




