*Małgorzata Kołodziejczak1, 2, Przemysław Ciesielski3
Complications after anal abscesses and fistulae treatment
Powikłania po leczeniu ropni i przetok odbytu
1Warsaw Proctology Centre, Saint Elizabeth’s Hospital, Mokotów Medical Centre
Head of Centre: Associate Professor Małgorzata Kołodziejczak, PhD
2National Institute of Geriatrics, Rheumatology and Rehabilitation, Warsaw
Head of Institute: Piotr Bednarski, MD, PhD
3Department of General Surgery, County Hospital in Wołomin
Head of Department: Krzysztof Górnicki, MD, PhD
Streszczenie
Według koncepcji odkryptowej etiopatogenezy chorób zapalnych odbytu ropień i przetoka odbytu są ostrą i przewlekłą fazą tej samej choroby, której przyczyną w większości przypadków jest zapalenie krypty odbytu. Inne przyczyny to: nieswoiste zapalenia jelit (choroba Leśniowskiego-Crohna, wrzodziejące zapalenie jelita grubego), uraz, ciało obce, radioterapia, nowotwór odbytu, odbytnicy lub narządów sąsiednich, choroby obniżające odporność (HIV) oraz przyczyny kazuistyczne: endometrioza, promienica, gruźlica. Pomimo wspólnej etiopatogenezy ropni i przetok odbytu powikłania mogące wystąpić w związku z leczeniem tych chorób są odmienne. W artykule omówiono powikłania związane z leczeniem ropni odbytu, do których należą: ponowne powstanie ropnia, powstanie przetoki, defekty kosmetyczne (deformujące okolicę odbytu blizny), powikłania septyczne (wstrząs septyczny, zespół Fourniera) oraz nietrzymanie gazów i stolca. Powikłania związane z leczeniem przetok odbytu to powikłania wczesne, typowe dla wszystkich zabiegów proktologicznych: krwawienie z rany, infekcja i wtórny do niej ropień, zatrzymanie moczu, zakrzepica żył okołoodbytowych. Powikłania późne są specyficzne dla operacji przetok i należą do nich: nawrót przetoki, objawy niewydolności zwieraczy, zniekształcenie odbytu, zwężenie odbytu, wypadanie błony śluzowej oraz przetoka jatrogenna. W artykule omówiono przyczyny i profilaktykę możliwych komplikacji pooperacyjnych. Podkreślono też rolę jakości życia pacjenta po operacji i związanej z tym satysfakcji pacjenta po zabiegu.
Summary
According to the crypt concept of anal inflammations aetiopathogenesis, anal abscess and fistula both constitute an acute and chronic phases of the same disease, which is caused in most cases by an inflammation of the anal crypt. Other causes include non-specific enteritis (Crohn’s disease, ulcerative colitis), trauma, foreign body, radiotherapy, anal cancer, rectal cancer or of adjacent organs, immunity lowering diseases (HIV) and casuistic reasons: endometriosis, actinomycosis, tuberculosis. Despite the common aetiopathogenesis of anal abscess and fistulae, complications that may arise in connection with the treatment of these diseases are different. The article discusses the complications associated with the treatment of anal abscesses, which include: re-emergence of an abscess, fistula, cosmetic defects (deforming scars in anal region), septic complications (septic shock, Fournier’s syndrome) and incontinence of gases and stool. Complications associated with the treatment of anal fistulae are early complications, typical of all proctological procedures: bleeding from the wound, infection and secondary abscess, urinary retention, venous thrombosis of perianal veins. Late complications are specific for the operations of fistulae and include: recurrent fistula, sphincteric failure, anal stenosis, mucosa prolapse and iatrogenic fistula. The article discusses the causes and prevention of possible post-operative complications. Authors stress the role of the quality of life of the patient after the surgery and related patient satisfaction following surgery.

Introduction
According to the crypt concept of anal inflammations aetiopathogenesis, anal abscess and fistula constitute an acute and chronic phase of the same disease, the cause of which in the majority of cases is anal crypt inflammation. Other reasons for the development of anal abscesses and fistulae are non-specific enteritis (Crohn’s disease, ulcerative colitis), trauma, foreign body, radiotherapy, neoplasm of the anus, rectum or adjacent organs, diseases lowering the immunity (e.g. in the course of HIV infection) and casuistic reasons: endometriosis, actionomycosis, TB and others). Fistula treatment has been absorbing surgeons for centuries due to the extremely large number of postoperative complications, mainly in the form of recrudescence and the symptoms of incontinence of gases and stool. Despite the common aetiopathogenesis of anal abscesses and fistulae, complications related to the treatment of the diseases are various and require separate discussion.
Anal abscess
Anal abscess is an acute surgical condition and should be voided as quickly as possible. Own observations show that patients having abscesses are often ignored by doctors, deemed to be patients with a ”minor surgical condition”. Correct incision of the abscess is decisive as regards the course of the disease and further life of the patient. The majority of patients with abscesses, exclusive of surface abscesses, located subcutaneously, should be operated on under the conditions of an operating theatre, under nerve block (epidural anaesthesia) or general anaesthesia, which allows one to fully control the anal canal, penetrate hourglass abscess cavity and their extensive drainage. In particular, in the case of patients uncertain diagnostically with abscesses located high, which are invisible while examining the anal area (e.g. high abscess, ischiorectal or pelvirectal), anaesthesia is a necessary diagnostic element. Abscesses treatment by ”puncturing” and only aspirating the purulent content under local anaesthesia is inadmissible. The incision should be wide, radial and close to the anal canal (under the control of a finger inserted to the rectum, with no damage to the sphincters), which ”makes easier” and shortens the way of the possible future fistula (1). The particularly hard to be treated horseshoe abscesses should be drained from separate incisions on both sides of the anus, thus avoiding one, surrounding incision connecting both sides of the anus due to possible functional and cosmetic complications (anal canal deformations) and slowed wound healing. Post-incision wound should be healing for several weeks and the patient should be constantly contacting the surgeon. The patient should also be instructed as regards the technique of changing the dressing.
Complications following abscess incision
The most common complications following abscess incision include:
– re-formation of abscess (early or later after the previous procedure),
– creation of a secondary abscess fistula,
– cosmetic defects (scars deforming the anal area).
Rare complications include:
– septic complications: septic shock, Fournier’s syndrome,
– incontinence of gases and stool: sensory, related to the damage of pudendal nerves and kinaesthetic related to the damage of sphincter muscles.
The next abscess
Literature on the subject matter includes data as regards the recrudescence of abscesses to be approx. 7.5-10% (2, 3) (tab. 1). According to the author, it is necessary to differentiate between abscesses developed early, up to 2 weeks following the procedure and the ones created later, over 2 weeks following the procedure. An abscess developed several or ten or so days following the previously drained abscess is usually the same abscess, improperly drained (4). Ineffective drainage may be proven by lack of mood improvements in the patient after the procedure, not total pain subsiding and elevated temperature. In such cases, the disease usually runs in accordance with the following: there is interim, few-days improvement following the operation, and then temperature increases again and the ailments relapse. Such situations are most often observed by surgeons in patients with hourglass abscesses with open and drained lower cavity of the abscess and no control over the fundus of the wound and the upper cistern was not drained, e.g. draining the surface abscess, while leaving the ischiorectal one, or draining the ischiorectal abscess, while leaving the pelvirectal one. A helpful examination in such cases is transrectal ultrasound exam making it possible to visualise high fluid cisterns.
Tab. 1. List of types of abscess operations and the frequency of recrudescence
| Type of operation | Leading indications | Recrudescence (%) |
| Abscess incision | Anal abscess, every | 10% (1) |
| Abscess incision with seton drain | Anal abscess with visible fistula internal opening | 2-8% (2) |
| Abscess incision with fistulotomy | Anal abscess with low simple fistula or intersphincteric fistula | 2-8% (2) |
| Abscess incision with counterincision | Anal horseshoe abscess | nd |
nd – no data
Fistula
The creation of a secondary abscess fistula is assessed in the literature on the subject matter to amount to 34-50% (3, 5).
It should be emphasised that in many cases a fistula is created even in the situation of correct abscess drainage. However, delay in the drainage of the purulent cistern is conducive to the creation of a branched fistula, much harder to be treated. It has been discussed for many years, whether in the course of abscess incision one should seek and drain the fistula opening. Own research suggests that during abscess incision, finding the fistula opening and a one-step seton insertion with later fistulotomy provides a statistically better therapeutic result. However, it needs to be highlighted that the cited research was performed on a group of patients operated on by an experienced team of colorectal surgeons (4). In the case the abscess is located near the surface with a finely visible surface fistula tract and internal opening – a one-step fistulotomy makes it possible to later avoid the creation of a fistula, though opinions in this respect are various (6). Some reports show that the commonly used packing of the abscess cavity is conducive to the creation of a subsequent fistula (7). The creation of a fistula should be suspected in the case the healing of a wound following abscess incision is very long (2-3 months) or there are other abscesses being created in the same place. Image examinations prove helpful in confirming the presence of fistula tract: transrectal ultrasound or MRI. The creation of a fistula following the incision of an abscess is prevented by appropriate change of dressing, rinsing the wound often and performing surgical controls, which results in the wound being healed ”from the bottom”.
Septic complications
The occurrence of septic complications is not affected by routine use of antibiotics therapy in patients with anal abscesses. Antibiotics therapy, recommended many years ago by such classics of coloproctology as Goligher (8), is currently being deemed conservative treatment and not recommended by the majority of surgeons owing to the practical lack of possibility of the antibiotic to penetrate into the abscess cavity (9). Antibiotics are administered only to the patients with lowered immunity, diabetes mellitus, heart valve diseases and in cases of very extensive inflammations.
The occurrence of septic complication secondary to anal abscess is due to:
– no abscess drainage or delayed abscess incision. It may happen in the case when the patient – despite the presence of symptoms – fails to see the doctor or the doctor gives inappropriate diagnosis (e.g. in the case of high abscesses hard to be diagnosed),
– insufficient drainage (too spring incision, failure to diagnose higher cavity of the abscess, e.g. ischiorectal or pelvirectal),
– lowered patient’s immunity.
Group at risk of septic complications are patients treated with immunosuppressors, steroids, with lowered immunity (with HIV infection), with diabetes mellitus and patients with inflammatory infiltrations with no signs of fluctuation.
The most severe septic complications are septic shock and Fournier’s gangrene.
Fournier’s gangrene is a severe septic complication being the result of bacterial microembolism in perineal tissues vessels, which leads to extensive perineal tissues necrosis. Treatment must be aggressive, multi-layer and cover septic shock treatment (wide antibiotics therapy, first empirically, later in accordance with culture), fluid therapy, administering pressor amines and surgical excisions of the perineal necrotic tissues. Fournier’s syndrome is a rare complication, yet with a high mortality rate, the likelihood of its occurrence may be estimated on the basis of a scale according to Lao et al. (10).
Complications following anal fistulae treatment
Complications following anal fistulae surgeries may be generally classified as early and late.
Early complications generally do not differ from the ones following other proctological surgeries and they include:
– bleeding from the wound,
– infections and secondary abscess,
– retention of urine (most frequently related to epidural anaesthesia or subarachnoid anaesthesia),
– perianal veins thrombosis.
Pain following the surgery is smaller than after other proctological procedures, since the basic cause of pain is internal sphincter contraction, and fistulae surgery usually decreases the tonus of sphincters.
Late complications are specific of fistulae surgeries and they include:
– fistula recurrence (fig. 1),
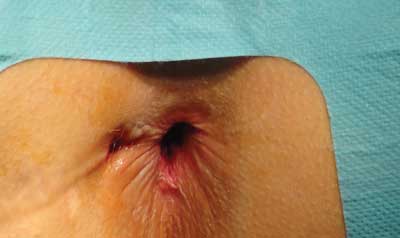
Fig. 1. Fistula recurrence and sphincteric failure
– symptoms of sphincteric failure (fig. 2),
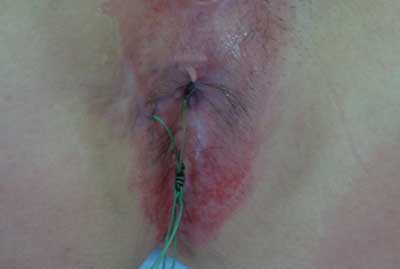
Fig. 2. Sphincter weakening related perianal dermal lesions
– anus deformation leading to a cosmetic defect or functional defect,
– anal stricture,
– mucous membrane prolapse,
– iatrogenic fistula.
Fistula recurrence
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
24 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
59 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
119 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 28 zł
Piśmiennictwo
1. Whiteford MH: Perianal Abscess/Fistula Disease Benign Anorectal Conditions. Guest Editor Bradford Sklow. Clin Colon Rectal Surg 2007 May; 20(2): 102-109. 2. Onaca N, Hirhberg A, Adar R: Early reoperation for perirectal abscess: a preventable complication. Dis Colon Rectum 2001; 44: 1469-1473. 3. Vasilevsky CA, Gordon PH: The incidence of recurrent abscesses or fistula-in-ano following anorectal suppuration. Dis Colon Rectum 1984; 27: 126-130. 4. Kołodziejczak M, Kosim A, Grochowicz P: Drenaż nitkowy w chirurgicznym leczeniu ropni odbytu – wyniki operacji 133 ropni odbytu w Oddziale Proktologii Szpitala Śródmiejskiego w Warszawie. Proktologia 2003; 4(4): 307-314. 5. Ramanujam PS, Prasad ML, Abcarian H, Tan AB: Perianal abscesses and fistulas. A study of 1023 patients. Dis Colon Rectum 1984; 27(9): 593-597. 6. McElwain JR, Alexander RM, Maclean MD et al.: Experience with primary fistulectomy of anorectal abscess: a report of 1000 cases. Dis Colon Rectum 1975; 18: 646-649. 7. Tonkin DM, Murphy E, Brooke-Smith M et al.: Perianal abscess: a pilot study comparing packing with nonpacking of the abscess cavity. Dis Colon Rectum 2004; 47: 1510-1514. 8. Goligher JC (ed.): Abscess. Surgery of the Anus, Rectum and Colon. 3rd ed. Charles C Thomas, Springfield, IL 1975: 200-201. 9. Blumetti J, Abcarian A, Quinteros F et al.: Evolution of treatment of fistula in ano. World J Surg 2012; 36: 1162-1167. 10. Lo O, Wei R, Foo D, Law WL: Ligation of intersphincteric fistula tract procedure for the management of cryptoglandular anal fistula. Surg Pract 2012; 16: 120-121. 11. Ortiz H, Marzo J, Ciga MA et al.: Randomized clinical trial of anal fistula plug versus endorectal advancement flap for the treatment of high cryptoglandular fistula in ano. Br J Surg 2009; 96: 608-612. DOI: 10.1002/bjs.6613. 12. Ommer A, Herold A, Joos A et al.: Gore BioA Fistula Plug in the treatment of high anal fistulas – initial results from a German multicenter-study. Ger Med Sci 2012; 10: Doc13. DOI: 10.3205/000164. Epub 2012 Sep 11. 13. Kołodziejczak M, Sudoł-Szopińska I: Diagnostyka i leczenie ropni i przetok odbytu. Borgis, Warszawa 2008: 233-236. 14. Rosa G, Lolli P, Piccinelli D et al.: Fistula in ano: anatomoclinical aspects, surgical therapy and results in 844 patients. Tech Coloproctol 2006; 10(3): 213-215. 15. Iwadare J: Sphincter-preserving techniques for anal fistulas in Japan. Dis Colon Rectum 2000; 4(10): 69-77. 16. van der Hagen SJ, Baeten CG, Soeters PB, van Gemert WG: Long-term outcome following mucosal advancement flap for high perianal fistulas and fistulotomy for low perianal fistulas: recurrent perianal fistulas: failure of treatment or recurrent patient disease? Int J Colorectal Dis 2006; 21: 784-790. 17. Ortíz H, Marzo J: Endorectal flap advancement repair and fistulectomy for high trans-sphincteric and suprasphincteric fistulas. Br J Surg 2000; 87: 1680-1683. 18. Soltani A, Kaiser AM: Endorectal advancement flap for cryptoglandular or Crohn’s fistula-in-ano. Dis Colon Rectum 2010; 53: 486-495. 19. Vial M, Parès D, Pera M, Grande L: Faecal incontinence after seton treatment for anal fistulae with and without surgical division of internal anal sphincter: a systematic review. Colorectal Dis 2010 Mar; 12(3): 172-178. 20. García-Aguilar J, Davey CS, Le CT et al.: Patient satisfaction after surgical treatment for fistula-in-ano. Dis Colon Rectum 2000 Sep; 43(9): 1206-1212. 21. Limura E, Giordano P: Modern management of anal fistula. World J Gastroenterol 2015 Jan 7; 21(1): 12-20. 22. Blumetti J, Abcarian A, Quinteros F et al.: Evolution of treatment of fistula in ano. World J Surg 2012; 36: 1162-1167. 23. Rojanasakul A, Pattanaarun J, Sahakitrungruang C, Tantiphlachiva K: Total anal sphincter saving technique for fistula-in-ano; the ligation of intersphincteric fistula tract. J Med Assoc Thai 2007 Mar; 90(3): 581-586. 24. Hong KD, Kang S, Kalaskar S, Wexner SD: Ligation of intersphincteric fistula tract (LIFT) to treat anal fistula: systematic review and meta-analysis. Tech Coloproctol 2014; 18: 685-691. 25. Van Koperen PJ, D’Hoore A, Wolthuis AM et al.: Anal fistula plug for closure of difficult anorectal fistula: a prospective study. Dis Colon Rectum 2007; 50(12): 2168-2172. 26. Lindsey I, Smilgin-Humphreys MM, Cunningham C et al.: A randomized, controlled trial of fibrin glue vs. conventional treatment for anal fistula. Dis Colon Rectum 2002; 45: 1608-1615. 27. Wilhelm A: A new technique for sphincter-preserving anal fistula repair using a novel radial emitting laser probe. Tech Coloproctol 2011; 15: 445-449. 28. Herreros MD, Garcia-Arranz M, Guadalajara H et al.: Autologous expanded adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistulas: a phase III randomized clinical trial (FATT 1: fistula Advanced Therapy Trial 1) and long-term evaluation. Dis Colon Rectum 2012; 55: 762-772. 29. García-Aguilar J, Belmonte C, Wong DW et al.: Cutting seton versus two-stage seton fistulotomy in the surgical management of high anal fistula. Br J Surg 1998 Feb; 85(2): 243-245. 30. Nwaejike N, Gilliland R: Surgery for fistula in ano: an audit of practise of colorectal and general surgeons. Colorectal Dis 2007; 9(8): 749-753. 31. Garcia-Aguilar J, Belmonte C, Wong WD et al.: Anal fistula surgery. Factors associated with recurrence and incontinence. Dis Colon Rectum 1996 Jul; 39(7): 723-729. 32. Ellis CN: Outcomes with the use of bioprosthetic grafts to reinforce the ligation of the intersphincteric fistula tract (BioLIFT procedure) for the management of complex anal fistulas. Dis Colon Rectum 2010; 53: 1361-1364. 33. Buchberg B, Masoomi H, Choi J et al.: A tale of two (anal fistula) plugs: is there a difference in short-term outcomes? Am Surg 2010; 76: 1150-1153. 34. Buchanan GN, Bartram CI, Phillips RK et al.: Efficacy of fibrin sealant in the management of complex anal fistula: a prospective trial. Dis Colon Rectum 2003; 46(9): 1167-1174 [PubMed]. 35. Patrlj L, Kocman B, Martinac M et al.: Fibrin glue-antibiotic mixture in the treatment of anal fistulae: experience with 69 cases. Dig Surg 2000; 17: 77-80. 36. Meinero P, Mori L: Video-assisted anal fistula treatment (VAAFT): a novel sphincter-saving procedure for treating complex anal fistulas. Tech Coloproctol 2011; 15: 417-422. 37. The Surgisis AFP anal fistula plug: report of a consensus conference. Colorectal Dis 2008; 10: 17-20.


