*Monika Bilewicz-Stebel1, Beata Bergler-Czop2, Katarzyna Sierant3, Dominika Wcisło-Dziadecka4
Vitamin D in dermatology
Witamina D w dermatologii
1Department of Internal Medicine, Dermatology and Allergology, Specialized Hospital in Zabrze
Head of Department: Professor Jerzy Jarząb, MD, PhD
2Department of Dermatology, School of Medicine in Katowice, Medical University of Silesia in Katowice
Head of Department: Professor Ligia Brzezińska-Wcisło, MD, PhD
3Chair and Department of Dermatology, Andrzej Mielęcki Silesian Independent Public Hospital in Katowice
Head of Department: Professor Ligia Brzezińska-Wcisło, MD, PhD
4Department of Skin Structural Studies, School of Pharmacy with the Division of Laboratory Medicine in Sosnowiec, Medical University of Silesia in Katowice
Head of Department: Krzysztof Jasik, assistant professor
Streszczenie
Witamina D pierwotnie znana była ze swej roli w utrzymaniu prawidłowej gospodarki kostnej poprzez regulację metabolizmu parathormonu (PTH), wapnia i fosforu, zapewniając integralność układu kostnego. W ostatnich dziesięcioleciach witamina D ponownie wzbudziła uwagę badaczy z powodu swojego wpływu na wiele zaburzeń zdrowotnych. Badania potwierdziły jej ingerencję w prawidłowe funkcjonowanie większości tkanek w organizmie człowieka, m.in. mózgu, sercu, mięśniach, układzie odpornościowym, a także w skórze. Wskazanie wielu nowych aktywności witaminy D było możliwe dzięki odkryciu obecności receptora witaminy D (VDR) w większości komórek organizmu i enzymów, które syntetyzują aktywną formę witaminy w innych niż nerkowe tkankach, zwłaszcza w skórze. Doprowadziło to do poszukiwania, jaką rolę odgrywa witamina D w zmniejszaniu ryzyka przewlekłych i śmiertelnych stanów chorobowych, takich jak: nowotwory, choroby autoimmunologiczne i zakaźne czy choroby układu krążenia. Zasadniczo uwaga dermatologów skupiała się na znaczeniu witaminy D w łuszczycy, ale prowadzone badania poszerzają jej terapeutyczne wskazania w chorobach skórnych, takich jak nowotwory czy atopowe zapalenie skóry.
Summary
Vitamin D was primarily acknowledged for its role in bones formation by regulating parathyroid hormone (PTH), calcium and phosphorous metabolism leading to maintain the integrity of the skeletal system. Over the past decades, once more the vitamin D attracted a great attention because of its implication in a numerous medical disorders. Researches confirmed its interference in the proper functioning of the most tissues in human body including brain, heart, muscles, immune system and also the skin. Pointing multiple new roles of the vitamin was possible because of discovery of vitamin D receptors (VDRs) in most cells of the body and the presence of enzymes that synthesize the active form of vitamin D in other than renal tissue, especially in the skin. It led to an interest in its role in decreasing the risk of chronic, highly morbid conditions such as carcinomas, autoimmune diseases, infectious diseases and cardiovascular diseases. Basically, its importance for dermatologists focused on its significance in psoriasis but further researches broadens its therapeutic effects in skin diseases such as skin cancers or atopic dermatitis.

Introduction
Vitamin D3 – chemically 1α,25-dihydroxycholecalciferol – is an organic steroid substance, historically classified as a vitamin, in fact being a hormonal compound. Its precursor – cholesterol – is a steroid lipid compound supplied to the human organism with food (exogenous cholesterol) or produced in a process of biosynthesis (endogenous cholesterol) (1, 2). At the basal layer of epidermis, the cholesterol is converted to provitamin D3 (7-dehydrocholesterol), which undergoes a photochemical transformation due to ultraviolet B radiation (UVB) into the intermediate compound previtamin D3. Then a slow isomerization process under the influence of temperature results in a production of cholecalciferol (traditionally called vitamin D3), lumisterol and tachysterol. This process takes place at UVB range between 290-320 nm, with the pick reaction at 297 nm. The process is presented at the figure 1. It is estimated that cholecalciferol biosynthesis in the skin covers 80-100% of the vitamin D demand. In the vitamin D metabolic pathway exists the reaction which protects against the formation of toxic amounts of this substance during a prolonged UVB exposure. It is a mechanism of increased production and accumulation of lumisterol, at the expense of vitamin D and tachysterol. But beyond skin biosynthesis, vitamin D is also delivered by absorption in the intestines from food and dietary supplements or medical products (3).
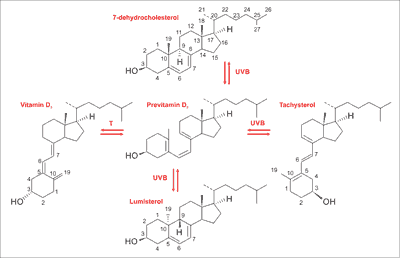
Fig. 1. Cycle of skin conversion 7-dehydrocholesterol to cholecalciferol – vitamin D3 (3)
Vitamin D3 is a form of prohormone – a chemical compound with a very low activity. It can be converted into a fully active hormone by enzymatic and non-enzymatic photochemical processes. Cholecalciferol synthesized in the skin is transferred to a circulatory system. The first activation is made in hepatic cells into 25-hydroxycholecalciferol – calcidiol [25(OH)D], which is the main form of vitamin circulating in the blood. The second stage occurs in the kidney cells with production of 1α,25-dihydroxycholecalciferol – calcitriol [1,25(OH)D], which is the main active form of the vitamin and a specific ligand for a nuclear vitamin D receptor (VDR). The receptor was initially found in intestinal epithelial cells, renal cells and bone tissue. For this reason, vitamin D was perceived by its activity in the calcium-phosphate and bone mineral metabolism (1). Researches which discovered the presence of VDR in most tissues of the human body made an increased interest in vitamin D action and underlined its importance in numerous diseases (3, 4). Vitamin D deficiency is a proved risk factor for such affection as cancers, skin diseases, autoimmune diseases, type 1 and type 2 diabetes mellitus, cardiovascular disease, hypertension, metabolic or infectious diseases, neurological syndromes and mental disorders (4, 5).
1,25(OH)D induces its endocrine effects on target tissues by intracrine, autocrine and paracrine routes in two ways – by modifying transcription of genes (genomic mechanism) and non-transcription (non-genomic mechanism) (1, 3).
Gene transcription is effected via binding calcitriol to VDR in the target cell cytoplasm what initiates the intracellular heterodimerization of VDR and the retinoic X receptor (RXR) leading to form a complex able to penetrate into the cell nucleus. Formed VDR-RXR heterodimer is capable to recognize specific DNA sequences – the VDRE (vitamin D response element) and along with multiple regulatory proteins, impacts the gene transcription, mainly by chromatin acetylation (4, 6). This process may result in the activation or inhibition of gene expression. The inhibition is probably responsible for anti-inflammatory and anti-proliferative features of the vitamin (3, 7).
As a non-transcriptional pathway the literature describes the ability of 1,25-dihydroxycholecalciferol to affect intracellular calcium levels, as well as intracellular signaling kinases and phosphatases. The facility of activation different pathways in different types of cells confirmed the pleiotropic features of vitamin D (7-9).
Vitamin D within the skin exerts control of cell proliferation, differentiation and maturation, control of angiogenesis, regulation of cellular apoptosis, induction of tumor cell differentiation, participation in the process of epidermal barrier renewal. The most recent literature reports highlight the immunomodulating effect of this compound, including stimulating immune system cell differentiation and inhibiting the production of proinflammatory cytokines that contribute to the development and progression of inflammatory process in skin diseases (10, 11).
Because of the proven impact of vitamin D on numerous disorders and its multiple activities, it is important to sick for its role in skin diseases.
Vitamin D and psoriasis
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects about 2-3% of the general population (12). Although the countless researches, the pathogenesis of psoriasis is still not fully known. Evidences suggest the dysregulation of the immune cells in the skin, especially lymphocyte T cells (2). Characteristic histopathological image shows hyperproliferation associated with incomplete differentiation of keratinocytes and decreased keratinocyte apoptosis, skin infiltration by activated inflammatory cells and impaired epidermal barrier function at the sites of skin lesions (2, 12).
Studies which focused on the possible role of vitamin D in psoriasis show several pathways of possible mechanism (13-15). It involves the anti-proliferative function of vitamin D – human keratinocytes exposed to an active form of the vitamin showed inhibition of growth and faster maturation (16). Moreover, the anti-inflammatory and anti-angiogenic activity of calcitriol counteract with inflammation and angiogenesis present in psoriatic lesions. It suppresses the proinflammatory Th1 and Th17 cell proliferation, as well as induce the regulatory T cells.
Topically used vitamin D analogues significantly decreases cutaneous levels of proinflammatory IL-17 and IL-8 (2, 17). In addition, dendritic cells differentiation, maturation and antigen presentation seem to be reduced by active 1,25(OH)D (12).
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
24 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
59 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
119 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 28 zł
Piśmiennictwo
1. Wadhwa B, Relhan V, Goel K et al.: Vitamin D and skin diseases: A review. Indian J Dermatol Venereol Leprol 2015; 81(4): 344-355.
2. Mostafa WZ, Hegazy RA: Vitamin D and the skin: Focus on a complex relationship: A review. J Adv Res 2015; 6(6): 793-804.
3. Piotrowska A: Modulacyjny wpływ witaminy D na działanie reaktywnych form tlenu w keratynocytach. Rozprawa doktorska. Gdański Uniwersytet Medyczny 2014.
4. Sintov AC, Yarmolinsky L, Dahan A et al.: Pharmacological effects of vitamin D and its analogs: recent developments. Drug Discov Today 2014; 19(11): 1769-1774.
5. Williams HC, Dellavalle RP, Garner S: Acne vulgaris Lancet 2012; 379(9813): 361-372.
6. Tukaj C: Właściwy poziom witaminy D warunkiem zachowania zdrowia. Postepy Hig Med Dosw 2008; 62: 502-510.
7. Ryan JW, Anderson PH, Morris HA: Pleiotropic Activities of Vitamin D Receptors – Adequate Activation for Multiple Health Outcomes. Clin Biochem Rev 2015; 36(2): 53-61.
8. Abelha-Aleixo J, Fonseca R, Bernardo A et al.: Vitamin D – immunomodulatory actions and new potentialities. Acta Reumatol Port 2014; 39(4): 355-356.
9. Maestro MA, Molnar F, Mourino A et al.: Vitamin D receptor 2016: novel ligands and structural insights. Expert Opin Ther Pat 2016; 26(11): 1291-1306.
10. Schlager JG, Rosumeck S, Werner RN et al.: Topical treatments for scalp psoriasis: summary of a Cochrane Systematic Review. Br J Dermatol 2017; 176(3): 604-614.
11. Cannell JJ, Hollis BW: Use of Vitamin D in Clinical Practice. Altern Med Rev 2008; 13(1): 6-20.
12. Barrea L, Savanelli MC, Di Somma C et al.: Vitamin D and its role in psoriasis: An overview of the dermatologist and nutritionist. Rev Endocr Metab Disord 2017; 18(2): 195-205.
13. Gisondi P, Rossini M, Di Cesare A et al.: Vitamin D status in patients with chronic plaque psoriasis. Br J Dermatol 2012; 166: 505-510.
14. Orgaz-Molina J, Buendía-Eisman A, Arrabal-Polo MA et al.: Deficiency of serum concentration of 25-hydroxyvitamin D in psoriatic patients: a case-control study. J Am Acad Dermatol 2012; 67: 931-938.
15. El-Moaty Zaher HA, El-Komy MH, Hegazy RA et al.: Assessment of interleukin-17 and vitamin D serum levels in psoriatic patients. J Am Acad Dermatol 2013; 69: 840-842.
16. Holick MF: Vitamin D: a millenium perspective. J Cell Biochem 2003; 88: 296-307.
17. Sugiyama H, Gyulai R, Toichi E et al.: Dysfunctional blood and target tissue CD4+ CD25 high regulatory T cells in psoriasis: mechanism underlying unrestrained pathogenic effector T cell proliferation. J Immunol 2005; 174: 164-173.
18. Soleymani T, Hung T, Soung J: The role of vitamin D in psoriasis: a review. Int J Dermatol 2015; 54(4): 383-392.
19. Autier P, Boniol M, Pizot C, Mullie P: Vitamin D status and ill health: a systematic review. Lancet Diabetes Endocrinol 2014; 2(1): 76-89.
20. Hambly R, Kirby B: The relevance of serum vitamin D in psoriasis: a review. Arch Dermatol Res 2017. DOI: 10.1007/s00403-017-1751-2.
21. Lehmann B: Role of the vitamin D3 pathway in healthy and diseased skin-facts, contradictions and hypotheses. Exp Dermatol 2009; 18(2): 97-108.
22. Reichrath J, Saternus R, Vogt T: Endocrine actions of vitamin D in skin: Relevance for photocarcinogenesis of non-melanoma skin cancer, and beyond. Mol Cell Endocrinol 2017; 453: 96-102.
23. Bikle D: Vitamin D and skin cancer. J Nutr 2004; 12: 3472S-3478S.
24. Wong G, Gupta R, Dixon KM et al.: 1,25-Dihydroxyvitamin D and three low-calcemic analogs decrease UV-induced DNA damage via the rapid response pathway. J Steroid Biochem Mol Biol 2004; 89-90: 567-570.
25. Dixon KM, Deo SS, Norman AW et al.: In vivo relevance for photoprotection by the vitamin D rapid response pathway. J Steroid Biochem Mol Biol 2007; 103: 451-456.
26. Gupta R, Dixon KM, Deo SS et al.: Photoprotection by 1,25 dihydroxyvitamin D3 is associated with an increase in p53 and a decrease in nitric oxideproducts. J Invest Dermatol 2007; 127: 707-715.
27. Caini S, Boniol M, Tosti G: Vitamin D and melanoma and non-melanoma skin cancer risk and prognosis: a comprehensive review and meta-analysis. J Cancer 2014; 50(15): 2649-2658.
28. Sharma CK, Sharma M, Aggarwal B, Sharma V: Different Advanced Therapeutic Approaches to Treat Vitiligo. J Environ Pathol Toxicol Oncol 2015; 34(4): 321-334.
29. Grimes PE, Nashawati R: The Role of Diet and Supplements in Vitiligo Management. Dermatol Clin 2017; 35(2): 235-243.
30. Silverberg JI, Silverberg AI, Malka E, Silverberg NB: A pilot study assessing the role of 25 hydroxy vitamin D levels in patients with vitiligo vulgaris. J Am Acad Dermatol 2010; 62: 937-941.
31. Li K, Shi Q, Yang L et al.: The association of vitamin D receptor gene polymorphisms and serum 25-hydroxyvitamin D levels with generalized vitiligo. Br J Dermatol 2012; 167: 815-821.
32. Birlea SA, Costin GE, Norris DA: Cellular and molecular mechanisms involved in the action of vitamin D analogs targeting vitiligo depigmentation. Curr Drug Targets 2008; 9: 345-359.
33. Alghamdi K, Kumar A, Moussa N: The role of vitamin D in melanogenesis with an emphasis on vitiligo. Indian J Dermatol Venereol Leprol 2013; 79: 750-758.
34. Tanghetti EA: The role of inflammation in the pathology of acne. J Clin Aesthet Dermatol 2013; 6(9): 27-35.
35. de Menezes AF, Oliveira de Carvalho F, Barreto RS et al.: Pharmacologic Treatment of Vitiligo in Children and Adolescents: A Systematic Review. Pediatr Dermatol 2017; 34(1): 13-24.
36. Gargoom AM, Duweb GA, Elzorghany AH et al.: Calcipotriol in the treatment of childhood vitiligo. Int J Clin Pharmacol Res 2004; 24: 11-14.
37. Finamor DC, Sinigaglia-Coimbra R, Neves LC et al.: A pilot study assessing the effect of prolonged administration of high daily doses of vitamin D on the clinical course of vitiligo and psoriasis. Dermatoendocrinol 2013; 5: 222-234.
38. Szepietowski J, Kapińska-Mrowiecka M, Kaszuba A et al.: Trądzik zwyczajny: patogeneza i leczenie. Konsensus Polskiego Towarzystwa Dermatologicznego. Przegl Dermatol 2012; 99: 649-673.
39. Metiko B, Brooks K, Burkhart CG et al.: Is the current model for acne pathogenesis backwards? J Am Acad Dermatol 2015; 72(6): 167.
40. Reichrath J: Vitamin D and the skin: an ancient friend, revisited. Exp Dermatol 2007; 16(7): 618-625.
41. Kramer C, Seltmann H, Seifert M et al.: Characterization of the vitamin D endocrine system in human sebocytes in vitro. J Steroid Biochem Mol Biol 2009; 113(1-2): 9-16.
42. Yildizgören MT, Togral AK: Preliminary evidence for vitamin D deficiency in nodulocystic acne. Dermatoendocrinol 2015; 6(1): e983687.
43. Bhat YJ, Latief I, Hassan I: Update on etiopathogenesis and treatment of Acne. Indian J Dermatol Venereol Leprol 2017; 83(3): 298-306.
44. Kim MJ, Kim SN, Lee YW et al.: Vitamin D Status and Efficacy of Vitamin D Supplementation in Atopic Dermatitis: A systematic Review and Meta-Analysis. Nutrients 2016; 8(12): 789.
45. Van Etten E, Decallone B, Verlinden L et al.: Analogs of 1α,25-dihydroxy- vitamin D3 as pluripotent immunomodulators. J Cell Biochem 2003; 88: 223-226.
46. Liu PT, Stenger S, Li H et al.: Toll-like receptor triggering of vitamin D mediated human antimicrobial response. Science 2006; 311: 1770-1773.
47. Schauber J, Dorschner RA, Coda AB et al.: Injury enhances TLR2 function and antimicrobial peptide expression through a vitamin D dependent mechanism. J Clin Invest 2007; 117: 803-811.
48. Howell MD, Wollenberg A, Gallo RL et al.: Cathelicidin deficiency predisposes to eczema herpeticum. J Allergy Clin Immunol 2006; 117: 836-841.
49. Samochocki Z, Bogaczewicz J, Jeziorkowska R et al.: Vitamin D effects in atopic dermatitis. J Am Acad Dermatol 2013; 69: 238-244.
50. Back O, Blomquist HK, Hernell O, Stenberg B: Does Vitamin D intake during infancy promote the development of atopic allergy? Acta Derm Venereol 2009; 89: 28-32.

