© Borgis - Postępy Nauk Medycznych 8/2013, s. 543-547
*Aneta Obcowska1, Małgorzata Kołodziejczak2, Iwona Sudoł-Szopińska2, 3, 4
Przydatność przedoperacyjnego badania endosonograficznego w różnicowaniu przetok odbytu niskich z wysokimi
The usefulness of preoperative endosonographic differentiation of low and high anal fistula
1Department of General Surgery, Vascular Surgery Unit, Praski Hospital, Warsaw
Head of Department: Rafał Górewicz, MD, PhD
2Department of General Surgery, Proctology Unit, Solec Hospital, Warsaw
Head of Department: Jacek Bierca, MD, PhD
Head of Unit: Małgorzata Kołodziejczak, MD, PhD
3Department of Radiology, Institute of Rheumatology, Warsaw
Head of Department: prof. Iwona Sudoł-Szopińska, MD, PhD
4Department of Diagnostic Imaging, II Medicine Faculty, Medical University, Warsaw
Head of Department: prof. Wiesław Jakubowski, MD, PhD
Streszczenie
Wstęp. Przedoperacyjne określenie wysokości przetoki ma duże znaczenie w wyborze sposobu leczenia oraz ocenie ryzyka pooperacyjnej inkontynencji. Ze zwiększonym ryzykiem wystąpienia tego powikłania wiąże się leczenie wysokich przetok przezzwieraczowych i międzyzwieraczowych, przetok nadzwieraczowych, rozgałęzionych, w tym podkowiastych, oraz przetok przednich u kobiet.
Cel. Celem pracy było określenie czułości i swoistości badania endosonograficznego (ang. endoanal ultrasound – EUS) w ocenie typu kanału pierwotnego oraz wysokości przetoki odbytu.
Materiał i metody. Materiał stanowiła grupa 424 pacjentów, w tym 132 kobiet i 292 mężczyzn w średnim wieku 45,8 lat, zoperowanych z powodu przetoki odbytu w okresie od 1 grudnia 2008 do 8 kwietnia 2011 roku. Przeprowadzono retrospektywną analizę danych z 424 przedoperacyjnych badań EUS oraz protokołów operacyjnych u tych samych pacjentów. Sprawdzono zgodność oceny przetok pod względem typu kanału pierwotnego oraz jego wysokości.
Wyniki. W EUS stwierdzono 63,1% przetok niskich i 36,9% wysokich, zaś śródoperacyjnie odpowiednio 57,6 i 42,2% przetok (w 0,2% przypadków stwierdzono przetokę mnogą). Zgodność EUS ze stanem śródoperacyjnym w ocenie wysokości przetoki uzyskano w 344 przypadkach (84,3%). Czułość i swoistość tej metody w ocenie wysokości przetok wyniosły odpowiednio 75 i 91%.
Wnioski. Badanie EUS jest dobrym badaniem dodatkowym do określania wysokości przetoki, niezależnie od jej typu. Czułość i swoistość metody w ocenie wysokości przetok wynosi odpowiednio 75 i 91%.
Summary
Introduction. Preoperative determination of the fistula is of great importance in the selection of treatment and the risk of postoperative incontinence. High transsphincteric and intersphincteric fistulas, suprasphincteric fistulas, branching ones, including horseshoe ones and front fistulas in women are associated with an increased risk of complications related to treatment.
Aim. The aim of this study was to determine the sensitivity and specificity of endosonographic examinations (EUS – endoanal ultrasound) to determine the type of the original canal and the location of anal fistula.
Material and methods. The material was a group of 424 patients, including 132 women and 292 men with a mean age of 45.8 years, operated because of anal fistula in the period from 1 December 2008 to 8 April 2011. A retrospective study of 424 preoperative EUS examinations and operational protocols in the same patients was conducted. The compatibility of the assessment of fistulas in terms of type of canal and its original location was verified.
Results. In EUS examinations 63.1% low fistulas and 36.9% high fistulas were determined, and intraoperatively, respectively 57.6 and 42.2% of fistulas (in 0.2% of cases plural fistula was determined). EUS compliance with the state of the intraoperative assessment of the fistula was obtained in 344 cases (84.3%). The sensitivity and specificity of this method in the evaluation of fistulas were respectively 75 and 91%.
Conclusions. EUS is a good additional examination for the determination of the fistula location, regardless of its type. The sensitivity and specificity of the method in the assessment of fistula is respectively 75 and 91%.

Introduction
Anal fistula is described has been described in the medical literature for more than 2.5 thousand years.
To date, the disease has gone through a number of pathophysiological theories, classifications and a series of proposed treatments. The treatment of anal fistula is often problematic and leaves the surgeon with a choice between the courageous operational procedure with a lower risk of disease recurrence with simultaneous increased risk of postoperative incontinence (urinary and bowel gas) and cautious procedure, giving more relapses, but less complications of incontinence. The recurrence rate quoted in the literature depends on, among other things, the type of operation performed, and in the case of fistulotomy eq3uals 0-11% (1, 2), using the surgical technique of a displaced muscle flap 1.5-4.6% (3, 4), anodermal flap-plastry 0-3% (5-10), the Hippocratic operation method 4-39% (2, 11-13). Leaving the internal opening and insufficient expertise of a surgeon are also factors increasing the risk of reoccurrence (1).
The most serious postoperative complication is urinary incontinence due to sphincter damage. The first scale to evaluate the severity of fecal and gas incontinence was developed by Parks and Browning (14). The currently most commonly used classifications are the ones of Wexner or Vaizeya (15, 16). The risk of incontinence and anal fistula after the operation is 0 to 25% (urinary incontinence), and even up to 60% (periodic dirty linen) (1, 6, 17, 18). High fistulas, apart from the branching ones (including horseshoe ones), front fistulas in women, recurrent fistulas, also formed after irradiation, and in the course of Crohn’s disease are known as the so-called “complex” fistulas (19).
Their operation is associated with increased rates of postoperative incontinence. Some authors believe that another risk factor for incontinence are more previous incision and drainage of anus abscess (20). In centers specializing in colorectal surgery postoperative complication rate is much lower than in the general chirurgic branches.
The determination of a fistula location is essential for the proper planning of the operation and evaluating the risk of complications. The canal is described as high when it covers more than 30% of the length of the anal external sphincter muscle (21) (fig. 1, 2).
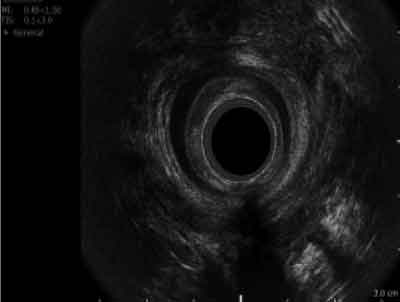
Fig. 1. Back high transsphincteric fistula.
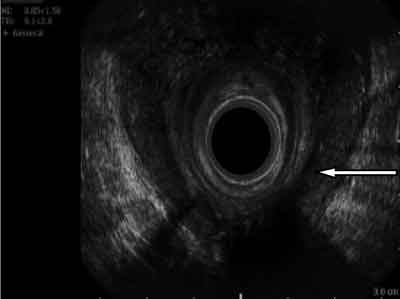
Fig. 2. Back high horseshoe intrasphincteric fistula.
Aim
The aim of this study was to determine the sensitivity and specificity of endosonography examinations (EUS – endoanal ultrasound) to determine the type and height of the original canal of anal fistula.
Material and methods
The material was a group of 424 patients, including 132 women and 292 men, aged between 17 and 83 years (mean 45.8 years), operated because of anal fistula in the period from 1 December 2008 to 8 April 2011. The analysis does not include vaginal-rectal fistulas, fistula arising in the course of Crohn’s disease, tuberculosis, actinomycosis, cancer, against the presence of a foreign structure, and after irradiation of the anal area. The study included only patients scheduled for elective surgery, except of emergency cases operated because of concomitant anal abscess.
A retrospective comparative analysis of data from 424 EUS examinations and operational protocols in the same patients was conducted.
All EUS examinations were performed by a single, experienced radiologist with Profocus Ultraview 2202-7 apparatus with endorectal 3D mechanical head, with a frequency of 16 MHz. The examination was performed in the Sims position, without bowel preparation. Transrectal probe was coated with ultrasound gel, and a latex sheath, which was also applied to the gel. The type of fistula was determined, the height, the location of the internal opening, the presence of any branches, morphological condition of sphincters. A similar analysis was conducted during the operation. Parks classification was used in the assessment (22). Fistulas were included in the group of high ones when covered more than 1/3 the length of the external anal sphincter muscle.
Concordance between the preoperative assessment of anal fistula in study EUS for the entire group was determined, as well as for different types of original canals. The sensitivity and specificity of EUS in these groups were calculated. For the calculations, the χ2 of Pearson independence test was used.
Results
In the EUS examination the following were diagnosed: 100 intersphincteric fistulas (23.6%), 266 transsphincteric fistulas (62.8%), 45 suprasphincteric fistulas (10.6%), 9 extrasphincteric fistulas (2.1%) and 4 unusual (submucosal) ones (0.9%). Intraoperatively following were diagnosed, respectively, 67 (15.8%) intersphincteric fistulas, 265 (62.5%) transsphincteric fistulas, 74 (17.5%) suprasphincteric fistulas, 17 (4.0%) extrasphincteric fistulas, 1 (0.2%) unusual fistula (submucosal).
For the entire group conformity assessment primary channel type was found in 312 cases (73.6%).
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
24 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
59 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
119 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 28 zł
Piśmiennictwo
1. Garcia-Aguilar J, Belmonte C, Wong WD et al.: Anal fistula surgery: factors associated with recurrence and incontinence. Dis Colon Rectum 1996; 39: 723-729.
2. Quah HM, Tang CL, Eu KW et al.: Meta-analysis of randomized clinical trials comparing drainage alone vs. primary sphincter-cutting procedures for anorectal abscess-fistula. Int J Colorectal Dis 2006; 21: 602-609.
3. Iwadare J, Sumikoshi Y, Sahara R: Muscle-filling procedure for transsphincteric fistulas. Dis Colon Rectum 1997; 40 (suppl.): 102-103.
4. Wang D, Yamana T, Iwadare J: Long-Term Results and Quality-of-Life Outcomes in Patients With Transsphincteric Fistulas After Muscle-Filling Procedure. Poster presentation at the meeting of The American Society of Colon and Rectal Surgeons, Chicago, Illinois, June 3-8, 2002.
5. Oh C: Management of high recurrent anal fistula. Surgery 1983; 93: 330-332.
6. Ho KS, Ho YH: Controlled, randomized trial of island flap anoplasty for treatment of trans-sphincteric fistula-in-ano: early results. Tech Coloproctol 2005; 9: 166-168.
7. Wedell J, Meier zu Eissen P, Banzhaf G, Kleine L: Sliding flap advancement for the treatment of high level fistulae. Br J Surg 1987; 74: 390.
8. Aguilar PS, Plasencia G, Hardy TG Jr et al.: Mucosal advancement in the treatment of anal fistula. Dis Colon Rectum 1985; 28: 496-498.
9. Sonoda T, Hull T, Piedmonte MR et al.: Outcomes of Primary Repair of Anorectal and Rectovaginal Fistulas Using the Endorectal Advancement Flap. Poster presentation at the meeting of The American Society of Colon and Rectal Surgeons, San Diego, California, June 2-7, 2001.
10. Dupsky PC, Stift A, Friedl J et al.: Endoanal advancement Flaps In Treatment of high anal fistula of Cryptoglandular Origin: Full thickness vs. Mucosal-Rectum Flaps. Diseases Colon and Recum 2008; 51: 852-857.
11. Garcia-Aguilar J, Belmonte C, Wong WD et al.: Cutting seton versus two-stage seton fistulotomy in the surgical management of high anal fistula. Br J Surg 1998; 85: 243-245.
12. Theerapol A, So BYI, Ngoi SS: Routine use of setons for the treatment of anal fistulae. Singapore Med J 2002; 43: 305-307.
13. Williams JG, MacLeod CA, Rothenberger DA: Seton treatment of high anal fistulae. Br J Surg 1991; 78: 1159-1161.
14. Browning G, Parks A: Postanal repair for neuropathic faecal incontinence: correlation of clinical results and anal canal pressures. Br J Surg 1983; 70: 101-104.
15. Jorge JMN, Wexner SD: Etiology and management of fecal incontinence. Dis Colon Rectum 1993; 36: 77-97.
16. Vaizey CJ, Carapeti E, Cahill JA, Kamm MA: Prospective comparison of faecal incontinence grading systems. Gut 1999; 44: 77-80.
17. Perez F, Arroyo A, Serrano P et al.: Fistulotomy with primary sphincter reconstruction in the management of complex fistula-in-ano: prospective study of clinical and manometric results. J Am Coll Surg 2005; 200(6): 897-903.
18. Arroyo A, Pérez-Legaz J, Moya P et al.: Fistulotomy and sphincter reconstruction in the treatment of complex fistula-in-ano: long-term clinical and manometric results. Ann Surg 2012; 255(5): 935-939.
19. Whiteford MH, Kilkenny III J, Hyman N et al.: The standards practice task force, The American Society of Colon ane Rectal Surgeons. Practice parametersfor the treatment of perianal abscess and fistula-in-ano (Revised). Dis Colon Rectum 2005; 48: 1337-1342.
20. Van Tets WF, Kuijpers HC: Continence disorders after anal fistulotomy. Dis Colon Rectum 1994; 37(12): 1194-1197.
21. Chung W, Kazemi P, Ko D et al.: Anal fistula plug and fibryn glue versus conventional treatment In repair of complex anal fistulas. Am J Surgery 2009; 197: 604-608.
22. Parks AG, Gozdon PH, Hardcastle JD: A classification of fistula-in-ano. Br J Surg 1976; 63: 1-12.
23. Hussein SM, Stoker J, Schouten WR et al.: Fistula in ano: endoanal sonography versus endoanal MR imaging in classification. Radiology 1996; 200: 475-481.
24. Cataldo PA, Senagore A, Luchtefeld MA: Intrarectal ultrasound in the evaluation of perirectal abscesses. Dis Colon Rectum 1993; 36: 554-558.
25. Lunniss PJ, Barker PG, Sultan AH et al.: Magnetic resonance imaging of fistula-in-ano. Dis Colon Rectum 1994; 37: 708-718.
26. Buchanan GN, Bartram CI, Williams AB et al.: Value of hydrogen peroxide enhancement of three-dimensional endoanal ultrasound in fistula-in-ano. Dis Colon Rectum 2005; 48: 141-147.
27. Gustafsson UM, Kahvecioglu B, Astrom G et al.: Endoanal ultrasound or magnetic resonance imaging for preoperative assessment of anal fistula: a comparative study. Colorectal Dis 2001; 3: 189-197.
28. Navarro-Luna A, Garcia-Domingo MI, Rius-Macias J, Marco-Molina C: Ultrasound study of anal fistulas with hydrogen peroxide enhancement. Dis Colon Rectum 2004; 47: 108-114.
29. Choen S, Burnett S, Bartram CI, Nicholls RJ: Comparison between anal endosonography and digital examination in the evaluation of anal fistulae. Br J Surg 1994; 78: 445-447.
30. Cho DY: Endosonographic criteria for an internal opening of fistula-in-ano. Dis Colon Rectum 1999; 42: 515-518.
31. Chew SS, Yang JL, Newstead GL, Douglas PR: Anal fistula: Levovist-enhanced endoanal ultrasound: a pilot study. Dis Colon Rectum 2003; 46: 377-384.
32. Toyonaga T, Matsushima M, Tanaka Y et al.: Non-sphincter splitting fistulectomy vs. conventional fistulotomy for high trans-sphincteric fistula-in-ano: a prospective functional and manometric study. Int J Colorectal Dis 2007; 22: 1097-1102.
33. Deen KI, Williams JG, Hutchinson R et al.: Fistulas in ano: endoanal ultrasonographic assessment assists decision making for surgery. Gut 1994; 35: 391-394.
34. Ratto C, Grillo E, Parello A et al.: Endoanal ultrasound-guided surgery for anal fistula. Endoscopy 2005; 37: 722-728.
35. Ratto C, Gentile E, Merico M et al.: How can the assessment of fistula-in-ano be improved? Dis Colon Rectum 2000; 43: 1375-1382.
36. West RL, Zimmerman DD, Dwarkasing S et al.: Prospective comparison of hydrogen peroxide-enhanced threedimensional endoanal ultrasonography and endoanal magnetic resonance imaging of perianal fistulas. Dis Colon Rectum 2003; 46: 1407-1415.
37. Giordano P, Grondona P, Hetzer H et al.: Three-dimensional endoanal ultrasonography is better than conventional anal endosonography in the assessment of fistula in ano. Dis Colon Rectum 2004; 47: 607-608.
38. Subasinghe D, Samarasekera DN: Comparison of Preoperative Endoanal Ultrasonography with Intraoperative Findings for Fistula In Ano. World J Surg 2010; 34: 1123-1127.
39. West RL, Zimmerman DDE, Dwarkasing S et al.: Endoanal Magnetic Resonance Imaging of Perianal Fistulas. Dis Colon Rectum 2003: 1407-1415.
40. Beets-Tan RGH, Beets GL, van der Hoop AG et al.: Preoperative MR Imaging of Anal Fistulas: Does It Really Help the Surgeon? Radiology 2000; 218(1): 75-84.
41. Buchanan GN, Halligan S, Bartram CI et al.: Clinical examination, endosonography, and MR imaging in preoperative assessment of fistula in ano: comparison with outcome-based reference standard. Radiology 2004; 233(3): 674-681.
42. Sahni VA, Ahamad R, Burling D: Which method is the best for imaging of perianal fistula? Abdom Imaging 2008; 33(1): 26-30.
43. Halligan S, Stoker J: Imaging of fistula in ano. Radiology 2006; 239: 18-33.
44. Morris J, Spencer JA, Ambrose NS: MR imaging classification of perianal fistulas and its implications for patient management. Radiographics 2000; 20: 623-635.
45. Stoker J, Rociu E, Zwamborn AW et al.: Endoluminal MR imaging of the rectum and anus: technique MRI of fistula-in-ano: a comparison of endoanal coil with external phased array coil techniques, applications, and pit-falls. Radiographics 1999; 19(2): 383-398.


