*Aleksandra Oleśna, Małgorzata Wojtkiewicz-Malicka, Anna Zakrzewska, Dorota Czech
Atypical localizations of infantile hemangiomas in pediatric otorhinolaryngology – report of two cases
Nietypowe lokalizacje naczyniaków wczesnodziecięcych w laryngologii dziecięcej – opis dwóch przypadków
Department of Pediatric Otorhinolaryngology, Medical University of Lodz, Poland
Head of Department: Dorota Czech, MD, PhD
Streszczenie
Naczyniak wczesnodziecięcy to najczęstszy łagodny guz wieku niemowlęcego. Zmiany te zazwyczaj nie są obecne podczas porodu, pojawiają się w pierwszych tygodniach życia. Ponad połowa naczyniaków wczesnodziecięcych lokalizuje się w rejonie głowy i szyi. Mają one tendencję do rozwoju w skórze i tkance podskórnej, jednak mogą również występować w narządach wewnętrznych. Te zmiany mogą mieć nietypowe objawy, dlatego też stanowią często wyzwanie diagnostyczne. Celem tego artykułu jest przedstawienie trudności diagnostycznych w dwóch przypadkach dzieci z naczyniakiem wczesnodziecięcym. Pierwszy pacjent prezentował ciężką duszność i stridor, zaś drugi pacjent zgłosił się z guzem lewej ślinianki przyusznej. Żadne z tych dzieci początkowo nie było podejrzewane o obecność naczyniaka wczesnodziecięcego. Potwierdzenie histopatologiczne diagnozy było możliwe tylko u jednego pacjenta, jednak oboje otrzymali skuteczne leczenie propranololem.
Summary
Infantile hemangioma (IH) is the most common benign tumors of infancy. IH typically are not present during childbirth, they manifest during first weeks of life. More than half of hemangiomas are localized in head and neck. IH tends to develop in the skin and subcutaneous tissue, however it may rarely present in internal organs. Those lesions may have atypical symptoms and are often a diagnostic challenge. The aim of this article is to present the diagnostic difficulties in two cases of children diagnosed with IH. First patient presented with severe dyspnea and stridor and second patient presented with the tumor of the left parotid gland. Both of these children at first were not suspected of IHs. The histopathological confirmation of the diagnosis was possible only in one patient, however both received propranolol treatment with success.
Introduction
Infantile hemangioma (IH) is the most common benign tumors of infancy, with an incidence of 5 to 10% infants (1). This type of tumor tends to occur more frequently in females, twins, premature infants, children with low birth weight and white neonates (2, 3). IH typically are not present during childbirth, they manifest during first weeks of life. The usual course of IH is characterized by a proliferative phase followed by involution phase (4). IH grows for about 6 to 9 months and tends to involute in 90% by the age of 9 (5). More than half of hemangiomas are localized in head and neck (4). IH tends to develop in the skin and subcutaneous tissue, however it may rarely present in internal organs, such as gastrointestinal tract or respiratory tract, especially in larynx. Those lesions are commonly manifested by cough, dyspnea or stridor and for this reason, they may be misdiagnosed with infectious diseases like for instance croup (6). Only after extensive additional investigations, such as laryngoscopy and imaging studies, the possible diagnosis of a vascular lesion might be considered (7). Definitive confirmation of the character of the tumor is possible due to the immunohistochemical examination of the tumor specimen (8). Generally, IH do not require any treatment. However, even up to 20% of children have to undergo some type of medical intervention (9).
The aim of this article is to present the diagnostic difficulties in two cases of children diagnosed with IH.
Case report
Case report one
A 4-month-old boy was admitted to the Department of Pediatric Otorhinolaryngology for the diagnosis of inspiratory-expiratory dyspnea and stridor. The patient was previously hospitalized in the Pediatric Department due to the pneumonia with severe dyspnea, with no improvement after treatment. Performed chest X-ray revealed inflammatory lesions and magnified mediastinal shadow. On physical examination, in addition to dyspnea and stridor, increased work of additional respiratory muscles was detected. Furthermore, occasionally the boy presented oxygen saturation level of 80%. During the diagnostic process, laryngotracheoscopy was performed. This examination showed segmental narrowing of the tracheal lumen, due to external compression. Sequentially, a contrast-enhanced computed tomography (CT) revealed the presence of a solid focal lesion of 30 x 31 x 22 mm on the right side of the neck, with strong contrast-enhancement, reaching to the superior mediastinum; the tumor was located between two brachiocephalic veins. Modeling and displacing the trachea to the left with tracheal compression to 2 mm was also described. CT results were sent to the Department of Cardiac Surgery – the consultants found no indications for urgent surgery due to high surgical risk of the procedure. Based on the history and imaging studies, considering the highest probability of IH, the patient was started on oral propranolol treatment. After three days of treatment, there was a significant improvement in the child’s respiratory function – normal oxygen saturation levels and improvement reported by the patient’s mother. After five days of oral propranolol, contrast-enhanced magnetic resonance imaging (MRI) showed well-demarcated focal lesion measuring 23 x 15 x 28 mm, undergoing strong homogeneous contrast enhancement, tracheal compression to 3 mm and it’s displacement was still observed (fig. 1a). The patient was discharged in good condition and had checkup appointments at the Vascular Malformation Treatment Center. Every 2 months he undergoes follow-up ultrasound scans, which revealed gradual reduction of the tumor (fig. 1b).
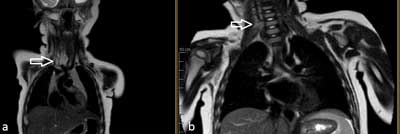
Fig. 1a, b. Patient one before and after the treatment (MRI)
Case report two
A previously healthy 4-month-old girl was admitted to the Department of Pediatric Otorhinolaryngology due to the presence of the tumor of the left parotid gland. Past medical history of the patient was irrelevant, she had no previous medical issues. On physical examination, she presented a tumor localized in the parotid region, measuring 2 x 3 cm; the overlying skin was not erythematous. An ultrasound scan (USS) revealed a hypoechogenic lesion approximately 20 x 26 x 32 mm with increased blood flow – the lesion was supposed to be hemangioma. Furthermore, a contrast-enhanced MRI was performed. This study showed a solid, encapsulated lesion in the superficial lobe of the parotid gland, segmentally involving the deep lobe, measuring 30 x 14 x 30 mm, with low signal on T1-dependent images and high signal on T2-dependent images; undergoing homogeneous contrast enhancement (fig. 2). According to the MRI, the tumor could be consistent with a pleomorphic adenoma. Due to discrepancies in imaging studies, the surgical biopsy of the tumor was performed. Histopathological findings revealed the expression of glucose transporter-1 (GLUT-1), which confirmed the diagnosis of IH. After cardiological consultation, the patient was started on oral propranolol therapy, initially at a dose of 1 mg/kg/day, then 2 mg/kg/day, with good clinical effect. During the postoperative period, the patient suffered from a mild upper motor neuron facial nerve palsy, which resolved completely after facial muscle massages, physiotherapy and administration of i.v. dexamethasone. After observation, the girl was discharged and received outpatient care at the Vascular Malformation Treatment Center.
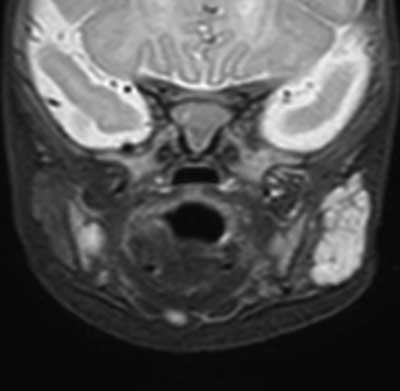
Fig. 2. Patient two – MRI
Discussion
IH is benign vascular tumor, which development is still being investigated. There are a few theories of the pathogenesis, which include: placental origin, intrinsic defect or somatic endothelial mutation, and extrinsic factors creating a supporting environment for growth of IH (10). If the placental hypothesis is confirmed, it would explain a higher rate of IHs in infants born following amniocentesis and chorionic villous sampling. There are also reports that IHs development takes place during the early stage of pregnancy (11). Furthermore, according to recent data, hemangioma and placental vessels show unique immunophenotypic similarity. Those include several markers, such as GLUT-1, merosin, Lewis Y antigen, Fcγ-RIIb, type III iodothyronine deiodinase, indoleamine 2,3-deoxygenase, and insulin-like growth factor 2 (IGF2) (12). Moreover, the expression of GLUT-1 is a way of distinguishing the IH from other vascular tumors (13).
IH are not usually present at birth, they occur during the first 2 weeks of life, grow during the proliferation phase and then tend to disappear during the involution phase. 60% of those lesions are located in the head and neck, mostly in the skin. However, they may develop in internal organs, such as parotid glands or respiratory tract (1, 3). Because of benign character and spontaneous involution, most IHs do not require treatment, it is necessary in some cases due to size, location and risk of possible complications. For instance, treatment should be implemented in tumors located in the respiratory tract (14). Furthermore, complications of IHs may be truly severe, 20 to even 40% of complications are life-threatening (15). The gold standard of systemic treatment is oral propranolol – nonselective β blocker, at the dosage for children in Western countries of 2-3 mg/kg/d. Side effects are not common, however may include bradycardia, hypotension or hypoglycemia (16). The exact mechanism of action been conclusively established, but several theories have been proposed. Current evidence suggest it’s role in vasoconstriction, inhibition of angiogenesis and nitric oxide, induction of apoptosis. Additionally, recent research indicates that propranolol can impact the transcription factor SOX18 directly, which disrupts the transformation of hemangioma stem cells into hemangioma endothelial cells (17). There are also other drugs that can be used, for instance corticosteroids or renin-angiotensin system acting agents. According to recent studies, sirolimus, an mTOR inhibitor, may be an alternative to propranolol. It has been shown to inhibit vasculogenesis, reduce differentiation and promotes regression of IH. Moreover, there are studies indicating, that sirolimus reduces proliferation of GLUT1 cells (18).
Other therapeutic options include topical therapy. Small and superficial IHs may be successfully treated with topical timolol. Treatment needs to be maintained up to 12 months, preferably until the involution phase (19). However, some IHs require surgical intervention – resection of the lesion or laser ablation should be considered, especially considering high risk tumors (20). Indications for surgical treatment are for instance life-threatening complications, functional impairment, pain or bleeding. There is no algorithm for the surgical approach, it must be considered individually, taking into consideration various factors like age, location, growth phase, condition of patient and many more, including potential psychosocial consequences of surgery (2).
While hemangiomas located in the skin are easy to identify and take further diagnostic and therapeutic measures, the localization of the lesions in internal organs poses a significant diagnostic issue. This is confirmed by the cases of the patients described in this article, since only comprehensive diagnostic tests and consultations enabled to make the diagnosis and initiate the proper treatment. Very important in the differential diagnosis is mentioned here marker GLUT-1, which became the base for the identification of IH in on of presented cases and allowed to implement the treatment (13). The problem appeared to be even more difficult in case of the second patient, in whom it was not possible to obtain a specimen for immunohistochemical studies. That is why, multispecialty consultations played an important role in the suspicion of IH and the initiation of treatment.
IHs of mediastinum are extremely rare, there are not many cases described in literature. According to Suárez Ramírez et al., treatment of choice appears to be surgical excision, however, due to the rarity of this lesion, further studies are essential to establish proper protocol of treatment (21). On the other hand, there are two described cases of pulmonary IHs, which were treated successfully with oral propranolol, despite the fact that in the majority of similar cases, surgical removal of the tumor was conducted. Those cases confirm that propranolol should be considered as a first-line therapy for IHs (22, 23).
Salivary gland hemangiomas account for 1% of salivary gland tumors (24), and those lesions are located mainly in the parotid gland. Moreover, it should be noted that IH is the most common tumor of the parotid gland in childhood. Surgical resections during the proliferative phase often led to severe complications, including blood loss, facial nerve injury or death (25). That is why, many researchers claim that conservative management should be treatment of choice in those tumors. On the other hand, there are reports that IH of the parotid gland tend to be less responsive to pharmacotherapy than hemangiomas of any other organs (26).
Conclusions
In conclusion, we report two cases of children with infantile hemangiomas of atypical localization – mediastinum and parotid gland. Both of those patients have been successfully treated with propranolol and they did not require radical surgery.
Piśmiennictwo
1. Jung HL: Update on infantile hemangioma. Clin Exp Pediatr 2021; 64: 559-572.
2. Haggstrom AN, Drolet BA, Baselga E et al.: Hemangioma Investigator Group. Prospective study of infantile hemangiomas: demographic, prenatal, and perinatal characteristics. J Pediatr 2007; 150(3): 291-294.
3. Krowchuk DP, Frieden IJ, Mancini AJ et al.: Clinical practice guideline for the management of infantile hemangiomas. Pediatrics 2019; 143: e20183475
4. Fulkerson DH, Agim NG, Al-Shamy G et al.: Emergent medical and surgical management of mediastinal infantile hemangioma with symptomatic spinal cord compression: case report and literature review. Child’s Nerv Syst 2010; 26: 17996-1805.
5. Bota M, Popa G, Blag C et al.: Infantile hemangioma: A brief review. Clujul Med 2015; 88: 23-27.
6. Darrow DH: Management of Infantile Hemangiomas of the Airway. Otolaryngol Clin North Am 2018; 51(1): 133-146.
7. Onder SS, Gergin O, Karabulut B: A Life Threatening Subglottic and Mediastinal Hemangioma in an Infant. J Craniofac Surg 2019; 30(5): e402-e404.
8. Johnson EF, Davis DM, Tollefson MM et al.: Vascular Tumors in Infants: Case Report and Review of Clinical, Histopathologic, and Immunohistochemical Characteristics of Infantile Hemangioma, Pyogenic Granuloma, Noninvoluting Congenital Hemangioma, Tufted Angioma, and Kaposiform Hemangioendothelioma. Am J Dermatopathol 2018; 40(4): 231-239.
9. Satterfield KR, Chambers CB: Current treatment and management of infantile hemangiomas. Surv Ophthalmol 2019; 64(5): 608?618.
10. Lo K, Mihm M, Fay A: Current theories on the pathogenesis of infantile hemangioma. Semin Ophthalmol 2009; 24(3): 172-177.
11. Itinteang T, Withers AHJ, Davis PF et al.: Biology of infantile hemangioma. Front Surg 2014; 1: 38.
12. Barnès CM, Huang S, Kaipainen A et al.: Evidence by molecular profiling for a placental origin of infantile hemangioma. Proc Natl Acad Sci USA 2005; 102(52): 19097-19102.
13. Steiner JE, Drolet BA: Classification of vascular anomalies: an update. Semin Intervent Radiol 2017; 34: 225-232.
14. Smithson SL, Rademaker M, Adams S et al.: Consensus statement for the treatment of infantile haemangiomas with propranolol. Australas J Dermatol 2017; 58: 155-159.
15. Tiemann L, Hein S: Infantile Hemangioma: A Review of Current Pharmacotherapy Treatment and Practice Pearls. J Pediatr Pharmacol Ther 2020; 25: 586-599.
16. Chen ZY, Wang QN, Zhu YH et al.: Progress in the treatment of infantile hemangioma. Ann Transl Med 2019; 7: 692.
17. Hasbani DJ, Hamie L: Infantile Hemangiomas. Dermatol Clin 2022; 40(4): 383-392.
18. Sebaratnam DF, Rodríguez Bandera AL, Wong LF, Wargon O: Infantile hemangioma. Part 2: Management. J Am Acad Dermatol 2021; 85(6): 1395-1404.
19. Satterfield KR, Chambers CB: Current treatment and management of infantile hemangiomas. Surv Ophthalmol 2019; 64(5): 608-618.
20. Chinnadurai S, Sathe NA, Surawicz T: Laser treatment of infantile hemangioma: A systematic review. Lasers Surg Med 2016; 48(3): 221-233.
21. Suárez Ramírez R, de Lucio Delgado A, González Cruz M et al.: Hemangioma infantil del mediastino posterior en un lactante de 11 meses, un hallazgo inusual. Reporte de un caso y revisión de la literatura (Posterior mediastinum infantile hemangioma in an 11 months infant, an unusual finding. A case report and review of the literature). Cir Pediatr 2019; 32(1): 46-48.
22. Galdeano F, Herón A, Moreno S et al.: Multiple pulmonary infantile hemangiomas responsive to oral propranolol. Pediatr Dermatol 2020; 00: 1-3.
23. Emiralioglu N, Oguz B, Akyüz C et al.: Successful treatment of pulmonary hemangioma with propranolol. Pediatr Pulmonol 2014; 49(8): 829-833.
24. Zarepur E, Moghimi M: Trismus Resulting from Infantile Hemangioma of the Parotid: A Rare Case Report. Iran J Ped Hematol Oncol 2015; 5(4): 249-253.
25. Greene AK, Rogers GF, Mulliken JB: Management of parotid hemangioma in 100 children. Plast Reconstr Surg 2004; 113(1): 53-60.
26. Sinno H, Thibaudeau S, Coughlin R et al.: Management of infantile parotid gland hemangiomas: a 40-year experience. Plast Reconstr Surg 2010; 125(1): 265-273.

