© Borgis - Postępy Nauk Medycznych 7/2012, s. 563-568
*Joanna Kopeć-Szlęzak, Urszula Podstawka, Agnieszka Gajewska, Anika Sikorska
Ekspresja molekuł CD79b, CD25 i CD100 na komórkach B w przewlekłej białaczce limfocytowej (PBL-B)
Expression of CD79b, CD25 and CD100 molecules on B cells in chronic lymphocytic leukemia (B-CLL)
Laboratory of Immunophenotyping, Institute of Hematology and Transfusion Medicine, Warsaw
Head: prof. Joanna Kopeć-Szlęzak, MD, PhD
Streszczenie
Wstęp. Molekule CD79b, jako składnikowi kompleksu BCR, przypisuje się istotny udział w dojrzewaniu limfocytów B i w przebiegu PBL-B. Ostatnio uwagę skupia również ekspresja CD25 (łańcuch α receptora dla IL-2) oraz semaforyny CD100, jako molekuł aktywujących interakcje białaczkowych limfocytów B z mikrośrodowiskiem, które może stymulować progresję PBL-B.
Cel pracy. Celem pracy była analiza ekspresji CD79b oraz analiza ekspresji tej cząsteczki i występowania na limfocytach białaczkowych B molekuł CD38, ZAP-70, molekuł uznanych za niekorzystne czynniki rokownicze, a także CD25 i CD100, istotnych w kontaktach z mikrośrodowiskiem.
Materiał i metody. Materiał stanowiła krew obwodowa 113 chorych na przewlekłą białaczkę limfocytową, rozpoznaną metodami rutynowymi. Metodą badawczą była trójkolorowa cytometria przepływowa z zastosowaniem przeciwciał monoklonalnych.
Wyniki. W grupie chorych z wysoką ekspresją CD79b na komórkach białaczkowych B częściej występowała ekspresja CD38 i dodatnia ekspresja ZAP-70 oraz częściej notowano progresję choroby i więcej zgonów niż w grupie CD79b ujemnej. Stwierdzono również, że u chorych o wysokiej ekspresji molekuły CD79b był istotnie wyższy odsetek komórek z ekspresją receptora dla IL-2 (CD25), aniżeli u chorych z ujemną ekspresją CD79b. W grupie 50 chorych, u których na komórkach B oznaczano ekspresję CD100 stwierdzono dodatnią korelację pomiędzy ekspresją CD38 a ekspresją CD100, a także u 90% chorych tej grupy ekspresję CD79b i CD25.
Wnioski. Uzyskane wyniki sugerują, że u chorych CD79b+ występuje „nagromadzenie” ekspresji czynników niekorzystnych rokowniczo i warunkujących interakcje komórek białaczkowych B z mikrośrodowiskiem PBL-B, co sprzyja progresji choroby.
Summary
Introduction. The CD79b, a component of the BCR complex plays an important role in lymphocyte B maturation and in the course of B-CLL. The α chain IL-2 receptor (CD25) and semaphorin Sema4D (CD100) are involved in B-CLL cells – microenvironment connections.
Aim. The aim of this study was to analyze the relationship between CD79b expression and adverse prognostic factors CD38, ZAP-70 as well as CD25 and CD100 molecules.
Material and methods. We analyzed peripheral blood samples from 113 B-CLL patients using three color flow cytometry with monoclonal antibodies.
Results. In the patients with CD79b expression on B-CLL cells the CD38, ZAP-70 expression and disease progression were more frequently observed, as compared to those without CD79b expression. We also observed in CD79b positive patients a significantly higher percentage of cells with CD25 expression than in patients from groupCD79b negative. In 40/50 analyzed patients with CD100 expression on B-CLL cells, a positive correlation between CD38 expression and CD100 expression was defined, as well as high expression of CD25.
Conclusions. Our results indicate that in CD79b positive patients is observed an „accumulation” of negative prognostic factors as well as expression of molecules responsible for interactions between B-CLL cells and the B-CLL microenvironment.

Introduction
Pathogenic factors associated with the clinical progression of B-cell chronic lymphocytic leukemia include intrinsic factors, depending on changes in the genome of leukemic cells and extrinsic, responsible for interactions between B-CLL cells and microenvironment. The latter include interactions of B cells via the surface BCR receptor that depend on its structure as well as on other factors related to this receptor e.g. ZAP-70 (1, 2).
The CD79b molecule in the BCR receptor complex is the part of CD79a/CD79b heterodimer. CD79b is an important functional factor of B lymphocytes (3) and is involved in the intracellular signaling and transfer of signals from BCR (4).
B-CLL progression is connected not only with CD38 and ZAP-70 expression (5, 6) but probably also with other molecules such as CD25 and CD100 (7, 8, 9). CD38 binds to CD31 on “nurse-like cells” and activates ZAP-70-dependent B cell signal and BCR receptor. It stimulates B-CLL cells proliferation (6). The Semaphorin (CD100) is involved in B-CLL progression via plexin β1 ligand binding on “nurse like cells” which stimulates B-CLL cell survival. CD38 and CD100 form a complex on B-CLL cell surface for signals from the microenvironment which additionally promotes B-CLL cells survival in B-CLL (7).
The presence of CD25 molecule (the α chain receptor for IL-2) favors apoptosis arrest through induction of growth of antiapoptotic protein levels in B cells and is considered an adverse factor for B-CLL development (8).
Although CD79b expression is also observed in many B lymphoproliferative disorders, the CD79b intensity expression in such cases may be too low to induce any response to anti CD79b antibody treatment (10, 11).
To our best knowledge, literature data on CD79b expression by B-CLL cells – did not provide an analysis of such expression in relation to the expression of known prognostic factors as CD38, ZAP-70 and other molecules important for B cell growth, for example CD25 and CD100 (12, 13).
The aim of our study was to analyze the relationship between CD79b expression and the expression of CD38, ZAP-70 and CD25 (IL-2 α receptor) and semaphorin CD100 (Sema4D) as molecules engaged in B-CLL cells and microenvironment relation.
Material and methods
The study material included peripheral blood samples from 113 B-CLL patients diagnosed by routine methods and distributed according to Rai disease classification system. Blood for analysis was received from samples used to routine diagnostic methods. Patients’ characteristics are presented in table 1.
Table 1. Clinical data on B-CLL patients.
| Characteristic | Patient number | % patients |
| Women/men | 50/63 | 44/56 |
| Rai stage | | |
| 0 | 24 | 21 |
| I | 46 | 41 |
| II | 26 | 23 |
| III/IV | 17 | 15 |
| | median | range |
| Age, year | 62 | 33-89 |
| WBC, x 103/l | 26.9 | 10.9-259.0 |
| Time observation (months) | 34 | 30-48 |
B-CLL cells were defined as having CD19+CD5+CD23+ CD20+weak CD79b± weak immunophenotype (14). We used monoclonal antibodies combined with fluorochromes. Three-color immunophenotyping with CD19 gating was performed for the following markers: CD5PE-Cy7, CD19PE, CD20PE, CD23APC, CD38PE and CD100FITC (Becton Dickinson) CD79bPE Beckman/Coulter, CD22FITC and CD25PE (DAKOCytomation) and ZAP-70 (Caltag Laboratories). Analyses were conducted with FACS Canto flow cytometer and FACS Diva software (Becton Dickinson).
We distinguished two groups of patients: 1) the CD79 positive group where the expression of 30% of B-CLL cells was designated for antigen threshold positivity and 2) the CD79b negative group where the percentage of CD79b+ B-CLL cells was below 30%. A proportion of 30% B-CLL cells was the designated threshold for positivity of CD38; for CD25 and CD100 expression was the designated as used in the common practice positively threshold of 20% B-CLL cells. According to literature, also 20% was the designated threshold for positivity of ZAP-70 in B-CLL cells (15). CD79b, CD25 and CD100 intensity expression was determined as stain index (SI) which normalizes the positive population signal and includes the degree of dispersion of the negative population (16).
Where: SI = Stain Index; Mean(pos) = the mean channel of positive population, Mean(neg) = the mean channel of negative population, SD(neg) = the standard deviation of the negative population.
Examples of CD79b expression on the surface of B-CLL cells are presented in figure 1.
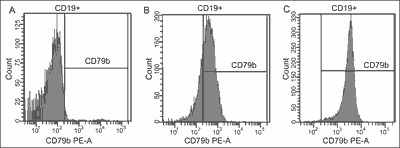
Fig. 1. CD79b expression on B-CLL cells in B-CLL patients, A – negative expression, B – positive CD79b middle expression, C – positive CD79b bright expression.
ZAP-70 expression was determined in 57 patients with Fix&Perm and anti-ZAP-70 antibodies and control, as recommended in literature (17). We also evaluated B-CLL cells count in patients with and without CD79b expression. In both, CD79b-positive and CD79b-negative groups, we evaluated disease stage at diagnosis according to Rai classification, time from diagnosis to the onset of treatment and response to treatment. Patients were monitored for 30 to 48 months.
Additionally, in 50 B-CLL patients with 0/I disease Rai` stage CD100 expression on B-CLL cells was analyzed. In this patient group CD38, CD79b, CD25 and ZAP-70 expression was determined. The control group comprised 10 healthy donors.
Statistical analysis was based on the nonparametric U Mann-Whitney test for independent trials. The p < 0.05 value was accepted as statistically significant. The correlation coefficient was calculated according to Pearson.
Results
CD79b expression on B-CLL cells
CD79b expression on B-CLL cells was detected in 93 patients (82%) while absence of CD79b expression was observed in 20 patients (18%). In the group of CD79b positive patients the mean proportion of CD79b+ B-CLL cells was 78.4 ± 19.7%, (median 83.7). In 80% of CD79b positive patients the value of CD79b+ B-CLL cell percentage was over 60%.
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Dal-Bo M, Bertoni F, Forconi F et al.: Intrinsic and extrinsic factors influencing the clinical course of B-cell chronic lymphocytic leukemia: prognostic markers with pathogenetic relevance. J Trans Med 2009; 7: 76-90.
2. Gobessi S, Laurenti L, Longo PG et al.: ZAP-70 enhances B-cell-receptor signaling despite absent or inefficient tyrosine kinase activation in chronic lymphocytic leukemia and lymphoma B cells. Blood 2007; 109: 2032-2039.
3. Minuzzo S, Indraccolo S, Tosello V et al.: Heterogeneous intracellular expression of B-cell receptor components in B-cell chronic lymphocytic leukaemia (B-CLL) cells. Br J Haematol 2005; 130: 5504-5509.
4. Treanor B, Depoil D, Gonzalez-Granja A et al.: The membrane skeleton controls diffusion dynamics and signaling through the B receptor. Immunity 2010; 32: 187-199.
5. Boonstra JG, Van Lom K, Langerak AW et al.: CD38 as a prognostic factor in B cell chronic lymphocytic leukemia (B-CLL: Comparison of three approaches to analyze its expression. Cytometry B Clin Cytom 2006; 70: 136-141.
6. Richardson S, Matthews C, Catherwood M et al.: ZAP-70 expression is associated with enhanced ability to respond to migratory and survival signals in B-cell chronic lymphocytic leukemia. Blood 2006; 107: 3584-3592.
7. Deaglio S, Vaisitti T, Bergui I et al.: CD38 i CD100 lead a network of surface receptors relaying positive signals for B-CLL growth and survival. Blood 2005; 105: 3042-3050.
8. Decker T, Bogner C, Oelsner M et al.: Antiapoptotic effect of interleukine-2 (IL-2) in B-CLL cells with low and high affinity IL-2 receptors. Ann Hematol 2010; 89: 1125-1132.
9. Burger JA, Ghia P, Roswald A, Caligaris-Capio F: The microenvironment in mature B-cell malignancies; a target for new treatment strategies. Blood 2009; 114: 3367-3375.
10. Zheng B, Fuji RN, Elkins K et al.: In vivo effects of targeting CD79b with antibodies and antibody-drug conjugates. Mol Cancer Ther 2009; 8: 2937-2946.
11. Dornan D, Bennet F, Chen Y: Therapeutic potential of an anti-CD79b antibody-drug conjugate for the treatment of non-Hodgkin Lymphoma. Blood 2009; 114: 2721-2729.
12. Kurtova A, Balakrishnan K, Chen R et al.: Diverse marrow stromal cells protect CLL cells from spontaneous and drug-induced apoptosis. Blood 2009; 114: 4441-4450.
13. Stevenson FK, Krysov S, Davies AJ et al.: B-cell receptor signaling in chronic lymphocytic leukemia. Blood 2011; 118: 4313-4320.
14. Habib LK, Finn WG: Unsupervised immunophenotypic profiling of chronic lymphocytic leukemia. Cytometry Part B, 2006; 70B: 124-135.
15. Schroers I, Griesinger F, Trumper L et al.: Combined analysis of ZAP-70 and CD38 expression as predictor of disease progression in B-cell chronic lymphocytic leukemia. Leukemia 2005; 19: 750-758.
16. Bigos M, Stovel R, Parks D: Evaluating multicolor fluorescence data quality among different instruments and different laser powers – methods and results. Cytometry 2004; 59A: 42-48.
17. Le Garff-Tavernier M, Ticchioni M, Brissard M et al.: National standardization of ZAP-70 flow cytometry determination: the French experience. Cytometry B Clin Cytom 2007; 72: 103-108.
18. Quijamo C, Lopez A, Rasilio A et al.: Impact of trisomy 12,del(13q0,del(17)p and del(11q) on the immunophenotyp DNA ploidy status and proliferative rate of leukemic-B-cells in chronic lymphocytic leukemia. Cytometry B Clin Cytom 2008; 74: 1441-1445.
19. Kopeć-Szlęzak J. Podstawka U, Sikorska A et al.: Znaczenie CD38 i ZAP-70 jako czynników rokowniczych w przewlekłej białaczce limfocytowej B (PBL-B). Post Nauk Med 2007; 20: 309-314.
20. Van Bockstaele F, Verhasselt B, Philippe J: Prognostic markers in chronic lymphocytic leukemia. Blood Rev 2009; 23: 25-47.
21. Shanafelt TD: Predicting clinical outcome in CLL. Hematology 2009; 75: 421-429.
22. Deaglio S, Malavasi F: Chronic lymphocytic leukemia microenvironment: shifting the balance from apoptpsis to proliferation. Haematologica 2009; 94: 752-756.
23. Patten PE, Buggins AG, Richards J et al.: CD38 expression in chronic lymphocytic leukemia is regulated by the tumor microenvironment. Blood 2008; 111: 5173-5181.
24. Malavasi F, Deaglio S, Damle R et al.: CD38 and chronic lymphocytic leukemia: a decade later. Blood 2011; 118: 3470-3478.
25. Herishanu I, Perez-Galan P, Liu D et al.: The lymph node microenvironment promotes B-cell receptor signaling and tumor proliferation in chronic lymphocytic leukemia. Blood 2011; 117: 563-574.
26. Caligaris- Cappio F.: Inflammation, the microenvironment and chronic lymphocytic leukemia. Haematologica 2011; 96: 353-355.
27. Ching ES, Kumanooh A. Roles of Sema4D and Plexin-B1 in tumor progression. Mol Cancer 2010; 9: 251-260.

