© Borgis - New Medicine 1/2005, s. 9-16
Joanna Kusowska
Cost-effectiveness analysis of therapy with standard antibiotics versus fluoroquinolone at Clinical Unit of diabetology, Medical University of ŁÓdŹ, Poland
Clinical Unit of Diabetology, Department of Clinical Pharmacology, Medical University of Łódź, Poland
Summary
Pharmacoeconomic studies of effectiveness, safety and costs of treatment have been scarce in Poland. Development of therapeutic standards for bacterial infections, based on pharmacoeconomic analysis and clinical studies evaluating treatment effectiveness and safety, allows for a more rational pharmacotherapy. The Alexander study of bacterial resistance to antibiotics conducted in Poland showed a high sensitivity of bacterial strains to simple and cheap antibiotics.
Aim
Clinical questions to be answered:
? Is the empirical therapy with cheap standard antibiotics [SAT] more effective than that with fluoroquinolone [FT] in patients with severe bacterial infections?
? What is the frequency of therapeutic failure in patients in severe clinical condition who were offered SAT or FT?
? What are the direct costs of the treatment of bacterial infections at the Clinical Unit in Łódź?
? What financial consequences may result from the introduction of treatment guidelines in bacterial infections, and a 5-day shorter hospital stay?
Material and methods
A prospective, randomized, single blind, pharmacoeconomic study was carried out in 420 patients with bacterial infections, admitted to our hospital from 1 July 2002 to 31 December 2002, who were given SAT versus FT.
Therapeutic effectiveness was evaluated daily from complaints reported by patients, complete physical examination, selected diagnostic and laboratory tests. The effectiveness of pharmacotherapy and the frequency of adverse drug reactions (ADR) were recorded.
The economic evaluation was done by assessing the direct cost of the treatment of patients with severe bacterial infections. Finally, the statistical significance was evaluated.
Results
Effectiveness: Empirical sequential SAT vs FT therapy administered at the Clinical Unit of Diabetology, Łódź, Poland showed similar effectiveness, i.e. 87% and 90% respectively.
Cost estimation: The direct costs of the therapy were low - approximately 85 USD vs. 83 USD with therapeutic success, and 126 USD vs. 97 USD with therapeutic failure. The cost of specialist consultation per patient during the treatment accounted for 0.85% - 1.38% of the resources allocated by the Health Care Fund for patient hospital stay. Sequential pefloxacin therapy resulted in direct cost savings (excluding hotel cost) of about 2450 USD per 1000 patients, which was economically significant.
Conclusion
In order to reduce the costs and increase the effectiveness at the Clinical Unit of Diabetology, we followed the therapeutic guidelines for bacterial infections, which resulted in high effectiveness and reduced hospital stay from 13 to 8 days, with cost-effectiveness of approximately 101,850 USD per 1000 patients.
INTRODUCTION
The Alexander resistance study of bacterial strains demonstrated a high susceptibility of the strains responsible for lower respiratory tract infections ( Haemophilus influenzae, Streptococcus pneumoniae, Moraxella cataralis), urinary tract infections (Escherichia coli, Proteus vulgaris) and other infections (Staphylococcus aureus) to cheap standard antibiotics (aminopenicillins) and pefloxacin which are rarely used in everyday clinical practice in Poland.
In patients with severe bacterial infections and at risk of developing side effects, administration of chemotherapy requires increased pharmacovigilance, monitoring of the treatment by performing clinical examination and laboratory tests, which increases treatment costs.
AIM
Clinical questions to be answered:
? Is administration of empirical cheap standard antibiotic therapy (SAT) vs fluoroquinolone (FT) effective in patients with severe bacterial infections?
? What is the frequency of therapeutic failure in patients in severe clinical condition who were given SAT or FT?
? What are the direct costs of the treatment of bacterial infections at the Clinical Unit of Diabetology?
? What may be the financial consequences of introducing treatment guidelines for bacterial infections?
METHODS
A prospective, randomized, single-blind pharmacoeconomic study was performed in 416 patients (420 patients were enrolled in the study; 2 patients died of stroke and there were 2 sudden cardiac deaths in the course of the therapy).
Inclusion criteria: patients with severe infections, age> 45 years, the Caucasian race, severe clinical condition and/or> 75 years of age (diabetes with ketoacidosis, stroke) receiving prophylactic antibiotic treatment.
Exclusion criteria: symptoms and signs of renal failure (creatinine> 2.0 mg/dl), dehydration, stroke with tetraplegia, diabetes with ketoacidosis or hyperosmolar coma, hypotonia, neoplasia, mental disease, administration of antidepressants for 30 days prior to the inclusion in the study, hypersensitivity to antibiotics and fluoroquinolones, severe dermatitis and bacterial resistance to selected chemotherapeutic agents.
Characteristics of the study population: A group of 416 patients with infectious diseases treated from 1st July to 31st December, 2002 at the Clinical Unit of Diabetology, Department of Clinical Pharmacology, Medical University of Łódź, Poland.
Group I included 208 patients with severe bacterial infections who had not received antibiotics prior to hospitalization, and who were treated randomly with intravenous-to-oral ampicillin/amoxycillin. Group II included 130 patients with severe infections in whom aminopenicillin treatment prior to hospitalization had proved to be ineffective, and who received an intravenous-to-oral switch therapy with pefloxacin (FT).
In the therapeutic failure group of 37 patients with no response to the SAT or FT on day 4, an alternative antibiotic therapy was instituted (other antibiotic therapy - OAT) with cefuroxime, cefotaxime, occasionally given in combination with penicillin G.
OAT was given to these 37 patients, as follows:
Cefuroxime 20 patients
Cefotaxime 9 patients
Cefotaxime + Penicillin G 1 patient
Pefloxacin + cefuroxime + cloxacilline 7 patients
Sixteen severely ill patients (with congestive heart failure class IV according to NYHA and/or respiratory failure) were simultaneously given clindamycin against anaerobic bacteria.
Antibiotics were given using the empirical sequential therapy, pharmacodynamic/pharmacokinetic properties of the agents, as well as microbial culture and sensitivity of the strains. Once the clinical improvement had been achieved, the intravenous drug administration was converted to the oral route [1-3]. All the patients met the criteria for hospital admission, and were also treated for their concomitant diseases, i.e.; cardio-vascular disease (congestive heart failure class III/IV according to the NYHA criteria, chronic heart disease and arterial hypertension) and diabetes [1-3].
Study endpoints:
1. The effectiveness of pharmacotherapy and the frequency of ADR were recorded:
? a therapeutic success was noted with the regression of all the symptoms of bacterial infection;
? a progressive improvement was found with the remission of all the clinical symptoms and signs as well as some positive laboratory findings;
? the frequency of ADR was recorded.
2. Economic assessment:
? direct costs of treatment of bacterial infections with SAT versus FT;
? financial savings resulting from the introduction of guidelines in the treatment of infections increased the therapeutic effectiveness and reduced the hospital stay from 13 to 8 days.
Therapeutic effectiveness was assessed daily by monitoring fever, dyspnoea, chest pain, productive cough, wheezing, chills, dysuria and other reported complaints as well as daily evaluated physical findings. Laboratory results, i.e. complete blood count (CBC), urinalysis, comprehensive metabolic profile (CMP), including fasting blood glucose, acid-base balance, levels of urea and creatinine, electrolytes, liver function tests (levels of bilirubin, alanine and aspartic transaminase) were monitored on days: 1, 4, 7, 14 of hospital stay or on day 3 after the regression of infection. Prior to drug administration, chest X-ray, microbial culture and sensitivity had been assessed. Complete physical examination and laboratory tests were performed before and 30 days after discharge from hospital.
The economic assessment was performed of the direct treatment costs in patients with bacterial infections hospitalized at the Clinical Unit of Diabetology [4-8].
Baseline characteristics of the study population (mean values for both study groups).
| | SAT | FT |
| Age (yrs) | 67 ? 8 | 69 ? 4 |
| Males | 68% | 65% |
| Lower respiratory tract infections [LRTIs] | Community acquired pneumonia | 49% | 51% |
| Bronchitis and chronic obstructive pulmonary diseases | 11% | 10% |
| Urinary tract infections [UTIs] | 28% | 25% |
| Prophylactic chemotherapy [PCT] | 12% | 14% |
The direct costs included:
1. medical and nursing care, and specialist consultation;
2. diagnostic and laboratory tests: CBC, CMP, antibiograms, imaging techniques (X-rays, ultrasound USG), computed tomography (CT), magnetic resonance imaging (MRI), electrocardiogram (ECG) including 24-hour Holter monitoring and 24-hour arterial blood pressure recording [9];
3. medication, i.e. antibiotics or fluoroquinolone [10].
Symptomatic treatment included pharmacotherapy with theophylline, mucolytics, b-adrenomimetics, cholinolytics, glycocorticosteroids, cromolyn sodium and antihistamine agents, NSAID (non-steroidal anti-inflammatory drugs), non-opioid analgesics, anxiolytics, hypnotics and antitussive drugs.
The costs of the medical, specialist, and nursing care were calculated from the following equation:
C = I / Np x Nph x T
C - cost of the medical and nursing care per patient during hospitalization,
I - income of physicians and/or nurses per annum (basic salary, on call duties, bonuses, additional rewards),
Np - number of patients hospitalized per year in the Clinical Unit of Diabetology,
Nph - number of physicians (and nurses) working at the Clinical Unit,
T - the length of hospital stay (days).
The costs of the following procedures were also estimated: intravenous infusion, single intravenous (IV), intra muscular (IM) drug administration, nursing procedures, measurement of blood pressure and temperature, and oxygen therapy. The cost of the procedures included the cost of pharmacological agents and materials, and nurses´ working hours. The costs of drug preparation, storage, distribution and administration were also included in the calculation.
The Health Care Fund established the total cost of the therapy per one hospital day at 385 USD in 2002 [9]. The hospitalization cost calculated by the HCF included the costs not related to the disease, number and type of procedures, diagnostic tests, specialist consultations and drug therapy, etc.
The discounting was not performed because the achieved benefits appeared to be parallel to the cost in the same calendar year.
Statistical analysis was done using the SPSS.PC v.8.0 computer programme. Demographic data and discrepancies in clinical effectiveness were analyzed by means of the chi-square test. The baseline characteristics for groups of continuous variables were evaluated using the Duncan test or the Kruskal - Wallis test. The cost differences were analyzed using the one-way Anova multigeneral linear model. The statistical significance was evaluated at p <0.05.
The study was approved by the Health Research Ethics Board of the Medical University of Łódź, Poland.
RESULTS
The effectiveness of treatment in patients with bacterial infections was assessed either as a therapeutic success or failure. The therapeutic success was consistent with regression of all the clinical and laboratory signs of infections, and the progressive improvement of the patients´ health was based on physical findings and results of accessory investigations.
Owing to the empirical intravenous-to-oral antibiotic therapy, the success was achieved in 87% patients treated with ampicillin/amoxycillin versus 90% of those receiving sequential pefloxacin.
The patients whose health did not improve, or deteriorated, were classified as therapeutic failure. The treatment failure was observed in 9%-13% of patients with a slight improvement after therapy and/or adverse drugs reactions (hypersensitivity, dyspepsia, headache, sleep disorders and insomnia). The average length of hospital stay of those patients was about 18 days.
ECONOMIC IMPACT
Patients treated with SAT for approximately 11 days, were hospitalized for 14.3 days and the direct cost of standard antibiotic therapy per one patient successfully treated or with constant improvement totalled 85.51 USD. Pefloxacin was administered for 7.8 days, the direct cost was 83.06 USD, the number of hospital days did not decrease significantly.
The direct costs of treatment per patient without therapeutic success or with adverse reactions increased by 41.03 USD with SAT and by 14.86 USD with FT.
Table 1. Clinical effectiveness of standard antibiotic /SAT/ vs fluoroquinolone therapy /FT/ in the Clinical Unit of Diabetology, Łódź, Poland.
| Direct costs | SAT success (USd) | SAT failure (USd) | FT success (USd) | FT failure (USd) |
| Physician* | 3.78 ? 0.19 | 5.31 ? 0.69 | 3.30 ? 0.28 | 3.34 ? 0.90 |
| Nursing care* | 3.56 ? 0.18 | 5.08 ? 0.66 | 13.16 ? 0.27 | 3.20 ? 0.86 |
| Specialist consultation* | 23.89?2.2 | 36.55?8.73 | 21.04?3.21 | 21.76?11.70 |
| Subtotal | 30.00?2.31 | 46.94?9.17 | 27.50?3.42 | 28.29?11.92 |
| Laboratory tests | 29.92?1.50 | 42.36 ? 5.45 | 28.52?2.31 | 48.09 ? 12.15 |
| Microbial tests | 0.47 ? 0.10 | 1.09 ? 0.28 | 0.95 ? 0.19 | 0.41 ? 0.17 |
| X-ray and other | 7.15 ? 0.93 | 7.26 ? 3.10 | 5.99 ? 1.29 | 1.14 ? 1.14 |
| Subtotal | 37.55?1.76 | 50.71?7.64 | 35.46?2.56 | 49.64?12.32 |
| Antibacterial therapy | 8.08 ? 0.83 | 19.49? 7.88 | 16.01 ? 3.35 | 11.77 ? 8.19 |
| Symptomatic therapy | 6.10 ? 1.61 | 4.07 ? 1.81 | 4.09 ? 1.82 | 8.21 ? 6.7 |
| Subtotal | 14.18 ? 2.06 | 23.58 ? 8.13 | 20.09 ? 3.89 | 19.98 ? 14.48 |
| TOTAL | 85.51?4.73 | 126.54?22.96 | 83.06?6.69 | 97.92?29.96 |
FT* – Sequential Fluoroquinolone Therapy
SAT* – Sequential Standard Antibiotic Therapy
ADR* – Adverse Drug Reaction (14 patients with allergy, dyspepsia, headache, insomnia)
In patients effectively treated with ampicillin, the resources obtained for pharmacotherapy constituted only about 17%, those spent on specialist physicians, nursing and consultant services accounted for 35%, and those spent on diagnostic investigations amounted to 44% of the total direct costs. In patients treated with pefloxacin 33% of resources was spent on medical services, 43% on diagnostic investigations, and 24% on pharmacotherapy (Table 2).
Table 2. Direct cost in SAT vs FT at the Clinical Unit of Diabetology, Łódź, Poland.
| Treatment of infections | No of patients (%) |
| SAT* | FT* |
| Therapeutic success | Cured | 87 | 91 |
| Improved |
| Therapeutic failure | Not improved | 13 | 9 |
| Failed |
| Adverse Drug Reaction (ADR) |
*Net salary
**Cost calculated on the 2000 USD exchange rate mean ± 1USD = 4.16 PLN
The cost of administration is included
Discounting was not necessary since the benefits occurred during the same time period as the costs.
The cost/effectiveness ratio in patients effectively treated with aminopenicillins versus fluoroquinolone was calculated.
The incremental cost/effectiveness ratio (ICER) in the therapeutic success of SAT and FT:
ICER = (C2 - C1 = D C)/(E2 - E1 = D E)
C2 -direct cost of FT, C1 - direct cost of SAT, E2 - effectiveness of FT, E1 - effectiveness of SAT, DC - difference of cost, DE - difference of effectiveness.
The treatment with fluoroquinolone appeared to be more cost-effective, but the difference was not significant.
The ARR index (Absolute Risk Reduction index; ARR = ARControl - ARTherapeutic) was calculated for therapeutic failure:
ARR = 13% - 9% = 4 = 0.04
Relative Risk RR = 13%: 9% = 1.44
The RRR index (Relative Risk Reduction; RRR = ARC - ART/ARC) for therapeutic failure was also calculated:
WRR = 0.04: 0.13 = 0.31
as well as the number of patients in whom therapeutic failure could be avoided:
Number Needed to Treat = NNT=1: 0.04 = 25
Reduction in the risk of therapeutic failure 0.13 vs 0.09 ARR 0.04, RRR 0.31; 95% CI:1.0 - 11.3, NNT 25.
In patients given FT, the reduction in the risk of therapeutic failure was 0.31. In order to avoid one failure, it was necessary to administer FT in 25 patients.
The assessment of therapeutic effectiveness revealed that 3% of patients (90%-87%) might be additionally cured with fluoroquinolone. Thus, an additional therapeutic effect might be achieved, together with a simultaneous reduction in costs by USD 2.45.
ATR (Absolute Therapeutic Ratio) = 90% - 87% = 3 = 0.03
RTR relative therapeutic ratio = 003: 0.87 = 0.034 The absolute therapeutic success is 3%, which means that, additionally, more than 33 patients need to be treated (NNT - number needed to treat 1: 0.03 = 33).
The total difference in costs to be incurred in both groups, is 2.45 USD per patient and constitutes 2.87% of SAT.
Therapeutic failure was found in 52 patients who were given SAT, and in 38 subjects treated with FT. The direct costs were increased by 28.62 USD in the SAT patients.
In 2002 the Health Care Fund paid about 385 USD for hospitalization per patient.
The total mean cost of hospitalization = direct costs + hotel accommodation cost
Hotel cost = total cost - (men direct cost of therapeutic success of SAT and FT)
Mean hotel costs per patient with therapeutic success;
Hotel cost = 385 USD - 84.29 USD = 300.71 USD
The cost of treatment per patient with bacterial infection was about 85.51 USD with SAT and 83.06 USD with FT, out of which direct costs constituted only 22% of the money allocated by the Health Care Funds for hospitalization, and hotel costs accounted for as much as 78% of the total.
In the patients with bacterial infection and therapeutic failure, the costs of antibiotic therapy and therapy of symptoms were increased by 41.03 USD with SAT and only by 13.95 USD with FT (Table 2).
Table 3. Incremental cost /effectiveness ratio (ICER).
| | SAT (USD) | FT (USD) | D |
| Cost of therapy | 85.51 | 83.06 | 2.45 |
| Success of therapy | 0.87 | 0.91 | 0.04 |
| ICER | | | 0.613 |
The money received for hospitalization of 416 patients with bacterial infections amounted to 160,160 USD and the direct costs of treatment of bacterial infections in all the patients without improvement of their physical condition following the empirical sequential chemotherapy of the first choice, or in those with adverse drug reactions, were 10,301.04 USD and represented only 6.43% of the money acquired from the Health Care Funds.
The results achieved at the Clinical Unit of Diabetology helped design a decision-tree model for the treatment of bacterial infections (Fig. 1).
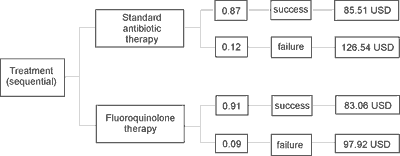
Fig.1. A decision-tree model of the treatment of bacterial infections in Poland.
DISCUSSION
Economic evaluation of selected medical procedures, diagnoses and treatments have been studied in some European countries and also in the United States of America [1, 5, 11-14].
Data on the economic impact of antibacterial treatment, and its impact on the Polish Health Care System are scarce. In our study we have made an attempt to answer the questions whether the administration of cheap, safe drugs of confirmed efficacy in vitro (aminopenicillin SAT) or alternative FT, is effective and safe, and whether it may reduce the treatment costs [15-18].
The cost-effectiveness analysis indicates where significant cost savings may be made in the future by implementing new, and modifying older therapeutic standards at the Clinical Unit of Diabetology with the Department of Clinical Pharmacology, Medical University of Łódź, Poland.
EVALUATION OF THERAPEUTIC SUCCESS
Ampicillin treatment in patients with bacterial infections was effective and accounted for as much as 87% of our patients; pefloxacin therapy was effective in about 90% of the patients.
We were the first researchers in Poland to assess the effectiveness and real current costs of treatment of bacterial infections in patients treated with SAT or FT. Our study showed that the empirical, standard antibiotic therapy is highly effective in bacterial infections, and the cost is justified due to a quickly achieved therapeutic success.
Paladino et al. reported that their therapeutic success in 72% to 76% of study patients indicated highly significant effectiveness of chemotherapy. Other reports showed that in the majority of cases intravenous antibiotics such as cefuroxime, gentamicin or ampicillin and oral cefuroxime and pefloxacin were administered more frequently than carbapenems or clindamycin [19, 20].
In our patients with antibiotic-resistant bacterial infections in whom the success was not achieved on day 4, a wide spectrum antibiotic therapy was introduced in combination with the 2nd and 3rd generation cephalosporins, semisynthetic penicillins, fluoroquinolones, penicillin or clindamycin.
The Alexander study of bacterial strains resistance conducted in Poland proved the highest " in vitro ” effectiveness of aminopenicillins against pathogenic strains of Streptoccocus pneumoniae (97%) and Haemophilus influenzae (86%) in lower respiratory tract infections. The research also showed significant effectiveness of ampicillin and pefloxacin in the treatment of urinary tract infections caused by Escherichia coli [21, 22].
The present study of empirical SAT or FT in severe infections proved it to be highly effective and cost-justified. Statistical analysis showed no significant differences between the effectiveness of SAT and FT therapies (p> 0.05).
In our study we attempted to calculate the current and real expenditure on services offered by specialist physicians, nurses and specialist consultants. However, we did not consider the costs incurred by the medical personnel associated with postgraduate training and self-education [4, 23].
At the Clinical Unit of Diabetology, the cost of specialist consultation per patient during the treatment accounted for 0.85%-1.38% of the resources allocated for patient hospital stay by the Health Care Fund. Decreased spending on diagnostics and laboratory tests may also reduce direct costs. However, it should be emphasized that in Poland, the costs allocated for physician, nursing care and specialist consultations are drastically low and cannot be the source of savings.
The cost of one dose of intravenously administered antibiotic was estimated at about 7 USD, orally administered 0.65 USD, and medicines preparation and storage costs were also calculated [25, 26].
Reduction in direct costs may also be obtained by decreasing resources for diagnostic investigations and laboratory tests.
In Poland, expenditure on effective pharmacotherapy of bacterial infections and concomitant diseases is low and accounts for 16.85% of the direct costs with SAT, and 24.19% with FT; it is also an extremely low percentage of expenditure allocated for antibiotic therapy in the EU countries and the USA.
Drug prices and pharmacotherapy cost may significantly influence direct costs only when the change in SAT guarantees a higher effectiveness, i.e. over 25% [27, 28].
Economic aspects impose reduction in the course of intravenous antibiotic administration at hospital, and later, a conversion to oral therapy provided by an outpatient physician as a follow-up care.
Many authors have reported that hospital stay reduced by one day may yield significant economic profits per year [12, 13, 15-17, 29-31].
However, attention should be drawn to the fact, that outpatient treatment requires continuation of pharmacotherapy, i.e. a monitored follow-up for 2 to 6 weeks after discharge from hospital. The outpatient treatment poses the risk of recurrent disease and readmission to hospital, as well as adverse reactions due to the therapy. The cost of the outpatient treatment, development of late ADR and asymptomatic infections in patients with diabetes also need to be considered [6, 12-17, 28].
Hendrickson and North observed that in the empirical therapy of patients with pneumonia the conversion from intravenously administered ceftriaxone to oral cefpodin made an economic difference. They calculated that the intravenous-to-oral switch in antibiotic therapy allowed saving 46.05 USD per patient, reduced hospital stay by one day, and produced no recurrence of the infection for about 30 days [3].
Fligelman et al. reported, that administration of sequential therapy with ciprofloxacin provides savings of 1159.97 USD as compared to the treatment with ciprofloxacin given only intravenously. They showed that it was cost-justified and acceptable for savings of USD 293 per patient with severe bacterial infections [2].
Jensen studied an economic impact of treatment with ciprofloxacin and enoxacin in comparison to a standard intravenous antibiotic therapy. They showed that the length of hospital antibiotic therapy course or outpatient treatment has a crucial effect on its costs: the average treatment cost was 4818 USD [19].
In our study, we found statistically significant differences between therapy effectiveness, the mode of chemotherapy and direct costs of treatment in patients with bacterial infections. However, some authors deny the idea of obtaining statistical significance in pharmacoeconomic investigations [1, 32].
We also found that sequential administration of pefloxacin for 7 days or ampicillin for about 11 days allows achieving therapeutic success. However, a short-term and highly effective pefloxacin therapy did not reduce the length of patients´ hospital stay.
In the UE countries, the number of days spent at hospital by patients with infections has been systematically reduced since it is cost-justified. However, physicians in Poland are not interested in decreasing treatment costs. The results of pharmacoeconomic analysis show that cheap and effective antibiotic therapy, change in work organisation at clinical units and establishing short-term therapy units will improve the economic condition of Polish hospitals. It is likely that earlier scheduled contracts with imposed admission quotas in clinical units do not promote quick diagnosis, therapeutic success or short-term hospital stay at clinical units in Poland.
Another study confirmed that the length of hospital stay has the greatest impact on decrease in the total costs. Reduced hospital stay at the Clinical Unit of Diabetology with the Department of Clinical Pharmacology, Medical University of Łódź, Poland, will generate significant savings in the future.
The following shows a calculation of savings for a shorter hospital stay (5 days):
C = Dc + Hc
C = total cost, Dc = direct cost, Hc = hotel cost
Hotel cost/day = 385 USD - 85.51 USD: 14.7 days = 20.37 USD/day
Hotel cost/day = 20.37 USD
Savings per patient = 20.37 USD x 5 days = 101.85 USD
The savings of 101.85 USD were obtained due to a reduced hospital stay per patient by an average time of 5 days. Each year, the staff of the Clinical Unit of admit and treat about 1000 patients with bacterial infections.
It should be emphasized that it is cost-justified, providing that the Health Care Fund pays for each patient´s hospital stay [12, 13, 18]. Saving 101,850 USD only by reducing the length of hospital stay of 1000 patients by 5 days with assurance of achieving therapeutic success in 87% is of significant economic importance for the deficit Health Care System in Poland. The savings will be allocated for necessary investment in Clinical Units in order to increase treatment effectiveness and to improve the poor economic situation of physicians and nurses in the future. The income of the medical staff should not become a source of savings in the Health Care System in Poland. However, due to insufficient hospital resources allocated from the national budget, even small savings made within a year at all levels of the Health Care Service may be of a considerable benefit. The lack of cost-effectiveness analyses in Poland has rendered it impossible to identify and implement the most effective and cost-justified methods of treatment.
CONCLUSIONS
1. In Poland, empirical sequential SAT versus FT revealed the same effectiveness and low costs in the treatment of bacterial infections.
2. Sequential pefloxacin therapy resulted in direct cost savings (without hotel costs) of about 2450 USD per 1000 patients over 6 months, which was certainly a significant economical result.
3. Empirical sequential aminopenicillin or pefloxacin therapy will reduce the number of hospital days and yield cost savings of about 101,850 USD per 1000 patients.
4. The mean cost of infection treatment in Polish hospital units is lower than that in any of the EU countries. 5. Implementation of treatment guidelines for infections at the Clinical Unit of Internal Medicine, Łódź, Poland, and setting up short-term therapy units may reduce the hospital stay only by 5 days, which may yield cost savings of approximately 104,300 USD per 1000 patients.
Piśmiennictwo
1. Drummond M., Davies L.: Evaluation of the costs and benefits of reducing hospital infection. J Hosp Infect 1991; 18 (supp.A): 85-93. 2.Fliegelman R.M., Matinngly P.M., Dempsey C.L., et al.: Economic impact of oral ciprofloxacin following standard intravenous therapy. Diagn. Microbiol. Infect. Dis., 1990; 13: 187-9. 3.Hendrickson J.R., North D.S.: Pharmacoeconomic benefit of antibiotics step-down therapy: converting patients from intravenous ceftriaxone to oral cefpodoxine proxetil. Ann Pharmacother 1995; 29:561-5. 4.Drummond M., Stoddart G., Turrance Q.: Methods for the economic evaluation of health care programmes. Oxford, Oxford University Press, 1987: 171. 5.Davey P., Duncan I., Edward D., et al.: Cost benefit analysis of cefradine and mezlocillin prophylaxis for abdominal and vaginal hysterectomy. Br. J. Obster. Gynecol. 1988; 95: 1170-7. 6.Davey P., Malek M.M., Parker S.T.: Pharmcoeconomics of antibacterial treatment. Pharmacoeconomics 1992; 1: 409-37. 7.Kusowska J.: Metody pomiaru kosztów w oddziałach szpitalnych. Farmacja Polska 2000; 4: 328-35. 8.Kusowska J.: Metody monitorowania skutków i kosztów terapii. Problemy Terapii Monitorowanej 2002; 1: 135-164. 9.Wycena Świadczeń Zdrowotnych ZOZ-u dla Szkół Wyższych, Łódź Styczeń 2002. 10.Medicines; Wykaz cen oferta dla aptek 2002; 4. 11.Kusowska J., Matusewicz W.: Podstawy farmakoekonomii dla lekarzy praktyków. Magazyn Medyczny 1998; 3: 29-30. 12.Kusowska J.: Cost-effectiveness analysis of the standard antibiotic versus fluoroquinolone therapy in Central European Countries (abstract 378). In: Program and abstract of the joint meeting of VII World Conference on Clinical Pharmacology and Therapeutics IUPHAR - Division of Clinical Pharmacology, CPT 2000 (Florence, Italy), DC British Journal of Clinical Pharmacology 2000: 1: 99-100. 13.Kusowska J.: Ekonomiczna ocena leczenia zakażeń dolnych dróg oddechowych. Farmakoekomomika 2001; 1: 52-8. 14.Kusowska J.: Analiza opłacalności TDM w zapobieganiu nefrotoksyczności po aminoglikozydach. (abstract 36). In program and abstract of the" VII Ogólnopolski Zjazd TTM" Spała, Poland, DC: Problemy Terapii Monitorowanej 2001: 9. 15.Kusowska J., Dąbrowski J.: Porównanie kosztów antybiotykoterapii w dwóch oddziałach chorób wewnętrznych. (abstract 32) . In: Program and abstracts of the International Symposium on Pharmacoeconomics in Central European Countries. Warsaw, Poland 1995; 10: 125-126. 16.Kusowska J., Matusewicz W.: Monitorowanie kosztów i korzyści w okołooperacyjnej profilaktyce przeciwbakteryjnej. Problemy Terapii Monitorowanej 1997; 8: 186-192. 17.Kusowska J., Tkaczyński W.: Analiza farmakoekonomiczna koszt - skuteczność w leczeniu powikłań cukrzycy typu 2. Medycyna Metaboliczna 2000; 1: 10-15. 18.Farmakoekonomika. Ekonomiczna ocena programów ochrony zdrowia. Praca zbiorowa pod redakcją M. Czecha. Oficyna Wydawnicza Politechniki Warszawskiej, Warszawa 2004. 19. Jensen K.M., Paladino J.A.: Cost-effectiveness analysis of abbreviating the duration of intravenous antibiotics with oral fluoroquinolones. PharmacoEconom. 1997; 11: 64-74. 20.Paretsch DJ, Paladino JA. Cost -effectiveness comparison of sequential ofloxacin versus standard switch therapy. PharmacoEconom. 1997; 31: 1137-1145. 21.Projekt "Alexander". Polska Grupa Robocza ds. Rekomendacji i Standardów Profilaktyki Racjonalnej Terapii Zakażeń. Warszawa 1999; 6. 22.Gold H.S., Moellering R.C.Jr.: Antimicrobial drug resistance. N.Engl. J.Med. 1996; 19: 1445-53. 23.Paladini J.A., Sperry H.E., Backes J.M., et al.: Clinical and economic evaluation of oral ciprofloxacin after an abbreviated course of intravenous antibiotics. Am. J. Med. 1991;91:462-70. 24.Kuti J.l., Capitano B., Nicolau D.P.: Cost-effective approaches to the treatment of community - acquired pneumonia in the era of resistance. PharmacoEconomics 2002; 20:514-27. 25.Kirschner C.G., Burkett R.C., Coy J.A., et al.: Physicians Current procedural terminology. Chicago:American Medical Association 1994. 26. Bureau of labor statistics data. Available from URL; Www.Bls.Gov/Cgi-Bin/Dsrv (accessed 2001). 27.Balas E.A., Kretschmer R.A., Gnann W., et al.: Interpreting cost analyses of clinical interventions. JAMA 1998; 1: 54-56. 28.Empy P.E., Jennings H.R., Thornton A.C., et al.: Levofloxacin failure in a patient with pneumococcal pneumonia. Ann. Pharmacother. 2001; 35: 687-90. 29. Scott W.G., Tilyard M.W., Dovley S.M., et al.: Roxithriomycin versus cefaclor in lower respiratory tract infection a general practice pharmacoeconomics study use comments; published erratum appears in Pharmacoeconomics. PharmacoEconom. 1993; 4: 122-30. 30.Morris S., Anderson P., Irwin D.E.: Acute exacerbations of chronic bronchitis: a pharmacoeconomic review of antibacterial use. PharmacoEconomics 2002; 20(3): 153-68. 31.Young M., Plosker G.L.: Piperacillin/tazobactam; a pharmacoeconomic review of its use in moderate to severe bacterial infections. PharmacoEconomics 2000; 19(11): 1135-75. 32.Johnson J.A.: Statistical significance in pharmacoeconomic analyses (letter) Can. J. Hosp. Pharm. 1996; 49: 57-8.
