© Borgis - Postępy Nauk Medycznych 1/2017, s. 22-26
*Michał Wąsowski, Marek Tałałaj
Bone fractures after stroke
Złamania kości u osób po udarze mózgu
Department of Geriatrics, Internal Medicine and Metabolic Bone Diseases, Centre of Postgraduate Medical Education, Warsaw
Head of Department: Associate Professor Marek Tałałaj, MD, PhD
Streszczenie
Udar mózgu jest jedną z głównych przyczyn niesprawności i śmierci. Pacjenci po udarze mózgu są podatni na przyspieszony ubytek masy kostnej, silniej zaznaczony po stronie niedowładu i w kończynach dolnych, oraz na złamania osteoporotyczne. Głównymi czynnikami, które wpływają na obniżanie się gęstości mineralnej kości (BMD), są: czas trwania unieruchomienia w następstwie niedowładu połowiczego, szybkość i stopień odzyskiwania sprawności ruchowej oraz zaawansowanie deficytów funkcjonalnych.
Strategia postępowania mająca na celu zapobieganie złamaniom u osób po udarze powinna być ukierunkowana na zahamowanie ubytku masy kostnej i zmniejszenie częstości upadków. Odpowiednio prowadzona rehabilitacja ruchowa może poprawić stan układu kostnego oraz mobilność osób po udarze. Ochraniacze bioder mogą być stosowane jako skuteczne amortyzatory ograniczające siłę urazu. Niedobór witaminy D musi zostać wyrównany. U pacjentów z niską wartością BMD i wysokim ryzykiem wystąpienia złamań należy wdrożyć leczenie z wykorzystaniem bisfosfonianów, takich jak: ryzedronian, alendronian, ibandronian lub kwas zoledronowy, a także suplementację witaminy B12 i kwasu foliowego.
Summary
Stroke is a major cause of disability and death. Patients after stroke are susceptible to accelerated bone loss, more evident at the paretic side and at lower extremities, as well as to osteoporotic fractures. Main factors that influence decrease in bone mineral density (BMD) are duration of hemiplegia-induced immobilization, time and degree of functional recovery, and severity of functional deficits.
Strategies for prevention of post-stroke fragility fractures should be focused on inhibition of bone mass loss and reduction of incidence of falls. Effective management, including physical exercise regimens, can improve bone health as well as patients’ mobility. Hip protectors can be used as effective shock absorbers. Vitamin D deficiency has to be corrected. In patients with low BMD and increased fracture risk antiresorptive treatment with bisphosphonates, such as risedronate, alendronate, ibandronate or zoledronic acid, as well as vitamin B12 and folate supplementation need to be used.

“I was looking forward to my 61st birthday in two days time. I enjoyed life-walks with the wind in my hair, driving to see loved ones, working in my garden for hours. Then my life stopped and I had to learn everything again. How to walk, how to dress and feed myself – everything I took for granted. But having to ask for help was hard – I’d been so independent all my life.
The stroke had wiped out all my left side. I walked with a frame with someone beside me and moving the log of wood that was my leg was exhausting. One doctor said it would be five years before my hand would move. I thought ‘never’ and worked on it hour after hour until I got one finger to move a fraction. The rest of my fingers came back in three months. I set myself targets and when I walked with a stick three months before the target month I was well chuffed. But last August, a freak fall broke my left hip. This has hit me harder than the stroke did...”.
(Letter to Stroke News, Volume 19.2, February 2001)
INTRODUCTION
Stroke is a cause of long-term disability and leaves 90% of post-stroke patients with functional limitations such as muscle weakness, pain, spasticity, cognitive dysfunction, poor balance and frequent falls (1, 2).
The incidence of strokes increases with age. The association of age-related bone loss and sarcopenia together with functional deficits make the patients with stroke vulnerable to falls and osteoporotic fractures.
FALLING
Bone fracture risk in stroke patients depends considerably on their tendency to falls (3). It was found that approximately 40% of patients experience at least one fall within the first year following stroke (4). Whether a fracture occurs when a person falls depends largely on the type and severity of falls (5, 6). As a result of impaired locomotor function, persons with stroke tend to fall towards the paretic side and demonstrate reduced ability to stretch the arm on the weak side in order to absorb the shock impact (3, 7).
Many factors are considered to be predictors of falls among stroke patients, such as: older age, male sex, right hemispheric stroke, post-stroke seizures, previous strokes, widespread white matter lesions, significant motor and mental dysfunctions, intercurrent infections and treatment with numerous drugs, especially analgesics, sedatives and antihypertensives (8). More than 7-fold increase in risk of falling has been described in stroke patients unaided while dressing (9).
BONE LOSS
Bone loss following stroke was found to contribute significantly to increased fracture risk. It starts within a few days following vascular brain injury and progresses until the 3rd-4th month after stroke. In the first year following stroke bone loss in the paretic arm can be the equivalent of more than 20 years of physiological bone loss in healthy individuals of comparable age (10).
Prospective studies determining biochemical markers of bone turnover revealed that bone resorption in hemiplegic patients increased as early as 7 days after stroke. It was accompanied by a decrease in bone formation suggesting significant remodeling imbalance at the bone multicellular unit level (11).
Increased bone resorption results in a rapid mobilization of bone calcium and immobilization-induced hypercalcemia, which is usually mild and can be detected by measurement of serum ionized calcium concentration. Significant positive correlation has been found between the degree of immobilization and serum ionized calcium concentration within the first and next years following stroke (10).
Several studies have reported a marked reduction in bone mineral density (BMD) of the paretic side after hemiplegic stroke (8). Measurements with dual-energy X-ray absorptiometry (DXA) scanning revealed that both upper and lower limbs appeared to be vulnerable to localized bone loss but a decrease in BMD of upper extremities was more evident than that of lower ones (8). It was found that within 1 year following stroke BMD in the paretic lower limb decreased by more than 10%, while BMD in the upper limb without paresis could actually increase, probably due to increased habitual use of the nonparetic hand (12) (fig. 1).
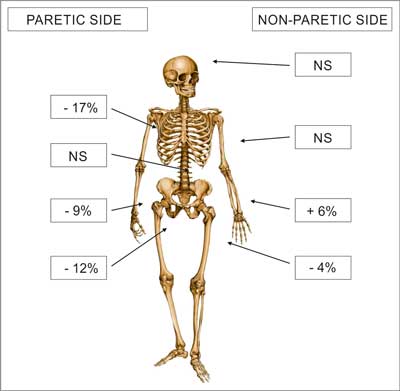
Fig. 1. Regional changes in bone density between 1 and 12 months after stroke in 18 individuals with pronounced paresis from unilateral stroke. Percentages are based on data from Ramnemark et al. (3) NS – non statistically significant
Immobility seems to be a major factor responsible for generalized bone loss. It was found that decrease in BMD correlated with the duration of hemiplegia-induced immobilization, time and degree of functional recovery, and severity of functional deficits (8, 13). Everyday capacity, muscle mass and strength as well as physical fitness of the paretic leg were found to influence BMD of the proximal femur (8, 10, 12).
Apart from reduced physical activity and immobilization the key mechanisms of bone loss in patients following stroke include: endocrine, nutritional, and pharmacological factors.
An important role in the development of osteoporosis seem to play disturbances in the vitamin D – parathyroid hormone (PTH) axis. In elderly individuals higher risk of stroke is often combined with vitamin D deficit, dramatically widespread in this population (14). It was shown that up to 83% of inpatients were vitamin D deficient and significant part of them had extremely low serum 25-hydroxyvitamin D concentrations, below 10 ng/mL (10, 15).
In most elderly patients with hemiplegic stroke even severe vitamin D deficiency is not accompanied by secondary hyperparathyroidism. It was suggested that elevated calcium serum levels, being the result of immobilization, reduced PTH secretion, and inhibited renal synthesis of 1,25-dihydroxyvitamin D. Consequently, stroke patients may show low serum concentrations of both 25OH vitamin D and 1,25(OH)2 vitamin D together with normal or low levels of PTH (10, 16-18).
Another factor that can be implied in post-stroke osteoporosis is vitamin K deficiency. It was found that vitamin K2 is essential for γ-carboxylation of bone Gla-protein, which is indispensable to set up bone matrix. In hemiplegic persons vitamin K deficiency was associated with low BMD and increased incidence of hip fractures on the paretic side (10).
Older age, mobility impairment, dysphagia, cognitive deficits and social isolation are important factors for poor nutrition frequently found in stroke patients. Apart from reduced vitamins D and K supply malnutrition may result in vitamin B12 and folate deficiency, and secondary hyperhomocysteinemia. It was shown that elevated serum homocysteine concentration influenced enzymatic collagen crosslinking in the posttranslational modification of collagen molecules, decreased bone tissue quality, and played important role in bone fragility associated with aging (10, 19, 20).
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Mozaffarian D, Benjamin EJ, Go AS et al.: Heart disease and stroke statistics – 2015 update: a report from the American Heart Association. Circulation 2015; 131(4): 229-322.
2. Quinn TJ, Paolucci S, Sunnerhagen KS et al.; European Stroke Organisation (ESO) Executive Committee; ESO Writing Committee: Evidence-based stroke rehabilitation: an expanded guidance document from the european stroke organisation (ESO) guidelines for management of ischaemic stroke and transient ischaemic attack. J Rehabil Med 2008; 41(2): 99-111.
3. Ramnemark A, Nilsson M, Borssen B et al.: Stroke, a major and increasing risk factor for femoral neck fracture. Stroke 2000; 31: 1572-1577.
4. Sato Y, Iwamoto J, Kanoko T et al.: Low-dose vitamin D prevents muscular atrophy and reduces falls and hip fractures in women after stroke: a randomized controlled trial. Cerebrovas Dis 2005; 20: 187-192.
5. Kannus P, Niemi S, Parkkari J et al.: Why is the age-standardized incidence of low-trauma fractures rising in many elderly populations? JBMR 2002; 17(8): 1363-1367.
6. Robinovitch SN, Inkster L, Maurer J et al.: Strategies for avoiding hip impact during sideways falls. JBMR 2003; 18(7): 1267-1273.
7. Dennis MS, Lo KM, McDowell M et al.: Fractures after stroke: frequency, types, and associations. Stroke 2002; 33: 728-734.
8. Myint PK, Poole KES, Warburton EA et al.: Hip fractures after stroke and their prevention. Q J Med 2007; 100: 539-545.
9. Lamb SE, Ferucci L, Volapto S et al.: Risk factors for falling in home-dwelling older womenwith stroke: the Women’s Health and Aging Study. Stroke 2003; 34: 494-501.
10. Carda S, Cisari C, Invernizzi M et al.: Osteoporosis after stroke: a review of the causes and potential treatments. Cerebrovasc Dis 2009; 28: 191-200.
11. Sato Y, Kuno H, Kaji M et al.: Influence of immobilization upon calcium metabolism in the week following hemiplegic stroke. J Neurol Sci 2000; 175: 135-139.
12. Beaupre GS, Lew HL: Bone-density changes after stroke. Am J Phys Med Rehabil 2006; 85: 464-472.
13. Demirbag D, Ozdemir F, Kokino S et al.: The relationship between bone mineral density and immobilization duration in hemiplegic limbs. Ann Nucl Med 2005; 19: 695-700.
14. Holick MF: Vitamin D deficiency. N Engl J Med 2007; 357: 266-281.
15. Shinchuk LM, Morse L, Huancahuari N et al.: Vitamin D deficiency and osteoporosis in rehabilitation inpatients. Arch Phys Med Rehabil 2006; 87(7): 904-908.
16. Sato Y, Fujimatsu Y, Honda Y et al.: Accelerated bone remodeling in patients with poststroke hemiplegia. J Stroke Cerebrovasc Dis 1998; 7(1): 58-62.
17. Sato Y, Kuno H, Kaji M et al.: Increased bone resorption during the first year after stroke. Stroke 1998; 29(7): 1373-1377.
18. Fujimatsu Y: Role of the parathyroid gland on bone mass and metabolism in immobilized stroke patients. Kurume Med J 1998; 45(3): 265-270.
19. van Meurs JB, Dhonukshe-Rutten RAM, Pluijm SMF et al.: Homocysteine levels and the risk of osteoporotic fractire. N Engl J Med 2004; 350: 2033-2041.
20. Sato Y, Honda Y, Iwamoto J et al.: Effect of folate and mecobalamin on hip fractures in patients with stroke. JAMA 2005; 293: 1082-1088.
21. Sato Y, Honda Y, Kunoh H et al.: Long-term oral anticoagulation reduces bone mass in patients with previous hemispheric infarction and nonrheumatic atrial fibrillation. Stroke 1997; 28: 2390-2394.
22. Panday K, Gona A, Humphrey MB: Medication-induced osteoporosis: screening and treatment strategies. Ther Adv Musculoskel Dis 2014; 6: 185-202.
23. Farhat G, Yamout B, Mikati MA et al.: Effect of antiepileptic drugs on bone density in ambulatory patients. Neurology 2002; 58: 1348-1353.
24. Ramnemark A, Nyberg L, Borssen B et al.: Fractures after stroke. Osteoporos Int 1998; 8: 92-95.
25. Kanis J, Oden A, Johnell O et al.: Acute and long-term increase in fracture risk after hospitalisation for stroke. Stroke 2001; 32: 702-706.
26. Andersson AG, Seiger A, Appelros P: Hip fractures in persons with stroke. Stroke Res Treat 2013; 2013 (2013): Article ID 954279.
27. Kim HW, Kang E, Im S et al.: Prevalence of pre-stroke low bone mineral density and vertebral fracture in first stroke patients. Bone 2008; 43: 183-186.
28. Batchelor FK, Hill K, MacKintosh S et al.: What works in falls prevention after stroke? A systematic review and meta-analysis. Stroke 2010; 41(8): 1715-1722.
29. Kerse N, Parag V, Feigin VL et al.: Falls after stroke: results from the Auckland Regional Community Stroke (ARCOS) study, 2002 to 2003. Stroke 2008; 39(6): 1890-1893.
30. Forster A, Young J: Incidence and consequences of falls due to stroke: a systematic inquiry. BMJ 1995; 311: 83-86.
31. Pouwels S, Lalmohamed A, Leufkens B et al.: Risk of hip/femur fracture after stroke. A population-based case-control study. Stroke 2009; 40: 3281-3285.
32. Parker MJ, Gillespie WJ, Gillespie LD: Hip protectors for preventing hip fractures in older people. Cochrane Database Syst Rev 2005; 3: CD001255.
33. Cameron ID, Stafford B, Cummings RG et al.: Hip protectors improve falls self-efficacy. Age Ageing 2000; 29: 57-62.
34. Parkkari J, Heikkilä J, Kannus IP: Acceptability and compliance with wearing energy-shunting hip protectors: a 6-month prospective follow-up in a Finnish nursing home. Age Ageing 1998; 27: 225-229.
35. Forsen L, Sandvig S, Schuller A et al.: Compliance with external hip protectors in nursing homes in Norway. Inj Prev 2004; 10: 344-349.
36. Pang MY, Eng JJ, Dawson AS et al.: A community-based fitness and mobility exercise program for older adults with chronic stroke: a randomized controlled trial. J Am Geriatr Soc 2005; 53: 1667-1674.
37. Cheng PT, Wu SH, Liaw MY et al.: Symmetrical body-weight distribution training in stroke patients and its effect on fall prevention. Arch Phys Med Rehabil 2001; 82: 1650-1654.
38. Fisher A, Srikusalanukul W, Davis M et al.: Poststroke hip fracture: prevalence, clinical characteristics, mineral-bone metabolism, outcomes, and gaps in prevention. Stroke Res Treat 2013; 2013: 641943. 10.1155/2013/641943.
39. Sato Y, Iwamoto J, Kanoko T et al.: Risedronate therapy for prevention of hip fracture after stroke in elderly women. Neurology 2005; 64: 811-816.
40. Poole KE, Loveridge N, Rose CM et al.: A single infusion of zoledronate prevents bone loss after stroke. Stroke 2007; 38: 1519-1525.
otrzymano: 2016-12-07
zaakceptowano do druku: 2016-12-28
Adres do korespondencji:
*Michał Wąsowski
Department of Geriatrics, Internal Medicine and Metabolic Bone Diseases Centre of Postgraduate Medical Education
Czerniakowska 231, 00-416 Warsaw
tel. +48 (22) 584-11-47
kl.geriatrii@szpital-orlowskiego.pl
Postępy Nauk Medycznych 1/2017Strona internetowa
czasopisma Postępy Nauk MedycznychPozostałe artykuły z numeru 1/2017:

