Antonina Respondek-Bartuś1, Natalia Dziewa1, Radosław Cylke1, *Małgorzata Kołodziejczak1, 2
Hidradenitis suppurativa. Contemporary diagnosis and treatment
Trądzik odwrócony. Współczesna diagnostyka i leczenie
1Department of General and Transplant Surgery Medical University of Warsaw
2Warsaw Proctology Centre, St. Elizabeth’s Hospital in Warsaw
Streszczenie
Trądzik odwrócony jest za rzadką i przewlekłą chorobą zapalną. Znacząco obniża jakość życia pacjentów, powodując cierpienie psychospołeczne. Najistotniejszym problemem wydaje się być dość uboga wiedza na temat tego schorzenia, zarówno wśród lekarzy, jak i społeczeństwa, co wiąże się ze znacznie opóźnioną diagnozą.
Autorzy przedstawiają aktualny przegląd piśmiennictwa dotyczący etiologii i diagnostyki choroby, a także zalecanego postępowania. Szczególną uwagę zwracają na najnowsze trendy w leczeniu, które kładą nacisk na spersonalizowane i interdyscyplinarne podejście terapeutyczne, uwzględniające farmakoterapię, metody chirurgiczne, a także modyfikację stylu życia. Podkreślają, że wczesne wdrożenie leczenia ma kluczowe znaczenie dla zatrzymania postępu choroby i uniknięcia poważnych powikłań.
Summary
Hidradenitis suppurativa, also known as acne inversa, is a rare chronic inflammatory disease. It causes psychosocial distress, significantly reducing patients’ quality of life. The limited knowledge of the condition, both among medical specialists and the general public, appears to be the main issue, often leading to significantly delayed diagnosis.
We present an up-to-date review of the literature on the aetiology and diagnosis of the disorder, along with current therapeutic recommendations. Particular attention was given to the latest treatment trends, which prioritise a personalized and interdisciplinary therapeutic approach encompassing pharmacotherapy, surgical interventions, and lifestyle modifications. It is emphasized that early treatment initiation is crucial for halting disease progression and preventing serious complications.

Hidradenitis suppurativa (HS), also known as acne inversa, is a rare and incurable disorder. However, its early clinical forms are encountered in primary healthcare settings far more often than commonly assumed (1).
The first descriptions of the disorder date back to the 19th century, when the term “hidradenitis” or “Velpeau’s disease” was introduced (2). The term “reverse acne” has been in use since the 1980s (3).
The Polish ICD-10 diagnosis L73.2, which reads multiple abscesses of the apocrine glands of the axilla, is misleading as it fails to account for lesions in other typical locations. Given the disease’s aetiology, it is also suggested that the term “apocrine gland inflammation” (hidradenitis suppurativa) should no longer be used. The term ‘acne inversa’ appears to more accurately reflect the etiopathogenesis of HS, as the pathological process originates in the hair follicle and only later involves the apocrine glands; it can also affect areas entirely lacking these glands (4).
Prevalence
The global prevalence of 0.4% qualifies HS as a rare disease. Higher rates are observed among African-Americans and in mixed ethnicity populations.
In Poland, the prevalence of HS is estimated at 1.6%, including about 400 patients with the most severe forms of the disease.
Women account for 75% of patients, and the average age at diagnosis is 36 years (5, 6).
Population-based studies indicate a higher prevalence of HS among individuals with lower socioeconomic status (6).
Comorbidities
Patients diagnosed with HS have an increased risk of comorbidities, including metabolic syndrome, diabetes mellitus (DM), polycystic ovary syndrome (POS), acne vulgaris, thyroid disease, arthritis, and inflammatory bowel diseases (Crohn’s disease and ulcerative colitis).
HS is also associated with a higher incidence of stroke, myocardial infarction, and a two times higher risk of sudden cardiac death.
Autoinflammatory syndromes (PASH, PAPA) and other conditions, i.e. the coexistence of HS with acne vulgaris, pyoderma gangrenosum, arthritis, and psoriasis, have also been described (6). Patients with HS are also more likely to develop depression and anxiety disorders, as well as have a higher risk of suicide.
Risk factors
Smoking is one of the main risk factors for HS, with smoking patients accounting for 44-70%. Nicotinism most likely causes an increase in interleukin 1 (β), as well as obstruction of hair follicle outlets due to acanthosis (4, 6).
Recent studies also point to dysbiosis of the skin microbiota, characterized by reduced commensal counts, increased levels of opportunistic anaerobic bacteria, and the presence of Staphylococcus aureus. It is believed that nicotinism may promote the growth of this pathogen (6).
A clear correlation has been found between HS and obesity, as well as between increasing BMI and a more severe disease course in women. The association between obesity and HS may be attributed to increased friction in hypertrophied skin folds (7). Friction and mechanical trauma induce activation of metalloproteinases and release of proinflammatory cytokines (6).
Aetiology
Scientific data suggest the involvement of an inflammatory process triggered by an abnormal immune response, blocked hair follicle outlets (proliferation of anaerobic bacteria), and hormonal factors as the main causes of the disease.
About 40% of patients have positive family history. Although the exact inheritance mechanism has not been fully described, research suggests an autosomal dominant pattern. However, environmental factors are known to affect gene expression and contribute to the development of HS symptoms (4).
Symptoms
HS manifests as painful, deep-seated inflammatory lesions involving specific body regions. It can appear as early as in adolescence. Patients often initially present to general practitioners (GPs), reporting painful recurrent furuncles (boils) in multiple locations.
The lesions typically occur in characteristic body regions such as under the arms, beneath the breasts, in the groin, on the inner thighs, and in the suprapubic area.
The armpits, buttocks, and perineal area, often including the perianal region, are most commonly affected in men (fig. 1-3). Less common sites include the neck and the area behind the auricle.
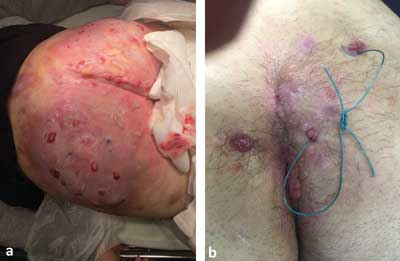
Fig. 1a, b. Extensive HS on the Buttocks

Fig. 2. HS in the groin
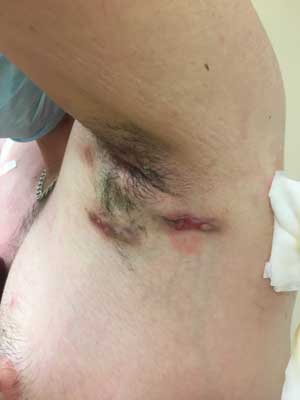
Fig. 3. HS located in the axillary area
The skin lesions take the form of nodules, hair cysts, subcutaneous tunnel structures, hyperpigmentation, and rope-like scarring. Pilonidal (hair) cysts may be a prodromal symptom of HS. These conditions can co-occur, e.g. pilonidal cyst, perianal fistula and HS.
Differential diagnoses (including misdiagnoses) include recurrent boils and acne vulgaris, which often coexist with HS (1, 8). The differential diagnosis should also consider pyoderma gangrenosum (PG), cutaneous manifestations of Crohn’s disease, and deep fungal or tuberculous infections. Lymphogranuloma venereum, granuloma inguinale and actinomycosis may also present with a similar clinical picture (6, 8). The estimated diagnostic delay is up to 12 years (5).
Diagnosis
The diagnosis is based on typical clinical manifestations in the form of characteristic skin lesions involving specific body areas, which persist or recur despite standard treatment for skin infections.
The presence of at least 2 lesions over a period of 6 months or 5 lesions in a lifetime is considered the diagnostic criterion (9).
Additional investigations, such as wound cultures, are generally not helpful for diagnosis or treatment planning; however, negative microbiological results and an additional family history can serve as secondary considerations (6).
It is crucial to note that squamous cell carcinoma, most often in the anogenital region, can progress with tissue necrosis, abscess formation, and fistulas, mimicking the features of HS on physical examination (4, 10).
Scientific reports indicate that chronic inflammation during the course of HS predisposes patients to neoplastic transformation (9, 11). According to Plewig et al., such neoplasms are more aggressive than those arising de novo. They often cause local infiltration, metastasis, and recurrence after surgical excision (4). Therefore, it is essential to obtain tissue samples for histopathology from the involved areas, even though some authors argue that this alone is not necessary to confirm the diagnosis (12).
Given the prevalence of adverse lifestyle factors and comorbidities, special attention is paid to a holistic approach to patient care. It is important to assess blood pressure and cardiovascular risk factors, including smoking history and BMI (12).
It is also advocated to screen the patients for depression, evaluate the disease’s impact on quality of life, and assess pain intensity. It is recommended to use HADS (Hospital Anxiety and Depression Scale) and DLQI (Dermatology Life Quality Index), with HiSQOL modification (Hidradenitis Suppurativa Quality of Life), which measures 17 quality-of-life items in the past 7 days (6, 13).
Diagnostic tools
Inflammatory and non-inflammatory forms of HS have been distinguished, based on the degree of detectable inflammation.
The severity of the inflammatory form can be classified as mild, moderate and severe, using the International Hidradenitis Suppurativa Severity Score System (IHS4). This tool is utilized in both clinical practice and research. The total score is calculated by summing the number of nodules (x 1), abscesses (x 2), and draining tunnels (x 4). A total score of ≤ 3 indicates mild disease, a score of 4 to 10 signifies moderate HS, and a score > 11 indicates severe disease (13).
The decision regarding surgical treatment plan is primarily based on the Hurley staging system (fig. 4). It also depends on the affected location (7), and the frequency of exacerbations (13).
Hurley staging system
Stage I
single or multiple isolated abscesses without sinus tract formation or scarring
Stage II
recurrent abscesses with one or more sinus tracts and scarring widely separated by normal skin
Stage III
diffuse involvement with multiple interconnected sinus tracts or abscesses |
Fig. 4. The Hurley staging system
SAHS assesses the severity of the disease, including the number of areas involved. HASI-R measures the extent of lesions based on the estimated body surface area (BSA) involved. HiSCR is used to evaluate treatment response, defined as an at least 50% reduction in total lesions compared to baseline. It is mainly used in clinical trials to assess treatment response. HiSCR75 and HiSCR90, which include a 75% and 90% reduction, respectively, are modifications of this scale.
The HS-IGA is the latest tool for assessing disease severity and treatment response. It is a score ranging from 0 to 5, based on the total number of lesions, with a treatment response defined as at least a 2-point reduction (13).
Treatment
Scientific data on HS treatment remain limited, with most available information being low quality or based primarily on expert opinion. Since the publication of the S1 guidelines in 2015, the validation of new therapeutic approaches has significantly transformed the understanding of HS treatment (7).
Pharmacotherapy is indicated for patients presenting only with inflammatory lesions, while surgery is needed in cases with tissue destruction (14).
It is the combination of anti-inflammatory pharmacotherapy and surgical resection of irreversibly damaged tissue that is currently considered the most appropriate therapeutic approach (7).
On the other hand, any surgical intervention should be part of a comprehensive treatment plan that always takes pharmacotherapy indications into account (14).
Pharmacotherapy
There is no regular medication officially approved for this indication, except for biological agents such as adalimumab, secukinumab, and bimekizumab, and it is crucial that patients be informed of this from the outset.
Topical treatment with clindamycin 1% emulsion applied twice daily for three months is recommended for Hurley stage I (mild). The treatment may be extended if clinically indicated.
A three-month course of oral tetracyclines, particularly in cases of frequent exacerbations, may also be effective due to their anti-inflammatory properties (1).
Clindamycin is the only antibiotic that has been investigated as a topical treatment for mild to moderate HS. In a randomized, double-blind, controlled trial involving 27 Hurley stage I and II patients, topical clindamycin 0.1% exhibited a 4.5?fold stronger improvement than placebo on superficial lesions (folliculitis, papules and pustules). Its effect on deep lesions, however, was negligible.
Topical 15% resorcinol (or resorcinol-containing cream) has been shown to be well tolerated and effective in reducing the size and number of HS lesions as well as pain. It is a phenolic compound with keratolytic, antipruritic and antiseptic effects. It is used twice daily during the exacerbation phase and once daily as maintenance treatment for up to 16 weeks. There are no available data on the use of resorcinol during pregnancy. Systemic toxicity following topical use of resorcinol is extremely rare (6). A retrospective trial reported HiSCR response in 85.3% of patients receiving resorcinol versus 52% with topical clindamycin (7).
It is also advisable to cleanse wounds with topical antiseptics, such as 4% chlorhexidine emulsion (available in Poland as 1% solution, potentially as a compounded medication) or benzalkonium chloride (currently, there are no skin-use preparations available in Poland) to prevent the development of drug-resistant bacteria (15). However, there is no scientific evidence to support the use of over-the-counter skin cleansers with antibacterial or anti-inflammatory effects, such as chlorhexidine, benzoyl peroxide, zinc pyrithione or triclosan. Patients with stage II (moderate) HS are put on systemic treatment with oral tetracyclines for a full three months. If good treatment response is achieved, it is recommended to continue treatment for an additional 12 weeks. Lymecycline 408 mg or doxycycline 100 mg, taken once or twice daily, are commonly used (1).
It is important to monitor patients for the risk of colitis and severe diarrhoea in the case of prolonged antibiotic therapy. This treatment is contraindicated in pregnant women and children under 9 years of age due to the risk of permanent tooth discoloration (7). Attention should also be paid to phototoxicity reaction and increased hepatotoxicity in patients with renal impairment (6).
Treatment of stage I and II HS can be started and continued in the primary healthcare setting.
Often, therapy can be temporarily discontinued once disease remission is achieved. If the response is inadequate, the patient should be referred to a dermatologist or a specialized centre.
If tetracycline therapy is unsuccessful, second-line treatment in specialized care involves clindamycin 300 mg twice daily combined with rifampicin 300 mg twice daily for 10 weeks. If effective, the entire cycle is recommended to be repeated 2-3 times per year.
It is important to note that rifampicin use can cause discoloration of urine and interacts with most hormonal contraceptives (as an enzyme inducer, it reduces their effectiveness). Before initiating rifampicin therapy, manufacturers recommend running baseline laboratory tests and repeating them during treatment (including complete blood count, urea, electrolytes, and liver function tests), with particular attention paid to patients with a history of alcohol abuse.
During long-term clindamycin therapy, the risk of pseudomembranous colitis caused by Clostridium difficile should be taken into consideration (7).
Combined treatment with rifampicin and clindamycin has been shown to significantly reduce plasma clindamycin levels in patients with HS. However, the clinical significance of this finding remains unknown (7).
The duration of systemic antibiotic therapy may be reduced by administering intravenous clindamycin at a dose of 600 mg twice daily for 5 days. This regimen can also be used before starting combination therapy in case of disease exacerbation.
No significant antimicrobial resistance has been observed in patients treated with tetracyclines or oral clindamycin. However, those put on topical clindamycin were more likely to develop clindamycin-resistant strains of Staphylococcus aureus compared to patients not receiving antibiotic therapy (7).
Patients with stage III HS should be directly referred to specialized care due to the need for complex pharmacological (starting with clindamycin/rifampicin combination therapy) and surgical treatment. It is important to emphasize that a multidisciplinary approach should be used in patients with HS. Limited surgical wound debridement alone is insufficient to prevent the spread of lesions. Disease control with pharmacotherapy before any planned surgical intervention is considered the most effective approach for stages II and III (13).
Biological therapy
Adalimumab, an inhibitor of tumour necrosis factor-alpha (TNF-α); secukinumab, an inhibitor of interleukin-17A (IL-17A); and bimekizumab, an inhibitor of IL-17A/F, all administered subcutaneously, have been approved by the European Medicines Agency for patients with moderate to severe HS not responding to conventional systemic therapies. Adalimumab has been additionally approved (2018) for patients aged ≥ 12 years for this indication.
Such treatment is administered in specialized centres. Clinical experience indicates good drug tolerance and significant improvement in patients with severe forms of the disease. Due to frequent relapses, long-term therapy is recommended to maintain effective disease control (7).
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Collier F, Ingram JR: Holistic assessment key in hidradenitis suppurativa. The Practitioner 2019; 263(1829): 21-24.
2. Jemec GBE: Hidradenitis suppurativa. Clinical practice. N Engl J Med 2012; 366: 158-164.
3. Bergler-Czop B: Trądzik odwrócony (acne inversa) – najczęstsze błędy w postępowaniu diagnostyczno-terapeutycznym. Dermatologia po Dyplomie 2014; 03.
4. Kołodziejczak M, Ciesielski P: Choroby proktologiczne. Diagnostyka i leczenie. Wyd. 1. PZWL, Warszawa 2022: 178-189.
5. Prof. Narbutt: Wywiad, artykuł MP: Na HS cierpi w Polsce około 400 pacjentów. Newseria 2018.
6. Matusiak Ł, Walecka I, Reich A et al.: Polskie wytyczne dotyczące diagnostyki i leczenia hidradenitis suppurativa. Dermatol Rev/Przegl Dermatol 2024; 111: 1-20.
7. Zouboulis CC, Bechara FG, Benhadou F et al.: European S2k guidelines for hidradenitis suppurativa/acne inversa part 2: Treatment. J Eur Acad Dermatol Venereol 2025; 39(5): 899-941.
8. Reddick CL, Singh MN, Chalmers RJG: Successful treatment of superficial pyoderma gangrenosum associated with hidradenitis suppurativa with adalimumab. Dermatol Online J 2010; 16(8): 15.
9. Ingram JR, Collier F, Brown D et al.: British Association of Dermatologists guidelines for the management of hidradenitis suppurativa (acne inversa). Br J Dermatol 2018; 180(5): 1009-1017.
10. Dessinioti C, Plaka M, Zisimou C et al.: Advanced squamous cell carcinoma of the axillae mimicking hidradenitis suppurativa. J Eur Acad Dermatol Venereol 2017; 31(9): 421-423.
11. Caccavale S, Caccavale T, La Montagna M: Hidradenitis suppurativa complicated by squamous cell carcinoma: isoscartopic response. Int Wound J 2017; 14(6): 1397-1398.
12. Fabbrocini G, Ruocco E, De Vita V, Monfrecola G: Squamous cell carcinoma arising in long-standing hidradenitis suppurativa: an overlooked facet of the immunocompromised district. Clin Dermatol 2017; 35: 225-227.
13. Karagiannidis I, Nikolakis G, Sabat R, Zouboulis CC: Hidradenitis suppurativa/Acne in-versa: an endocrine skin disorder? Rev Endocr Metab Disord 2016; 17: 335-341.
14. Hurley H, Roenigh R: Axillary hyperhidrosis, apocrine bromohidrosis, hidradenitis suppurativa, and familial benign pemphigus: surgical approach. Dermatologic surgery. New York 1989: 729-739.
15. Cramer P, Schneider-Burrus S, Kovics M et al.: Acne inversa – opcje chirurgiczne, rekonstrukcje i kombinacje z terapiami lekowymi. Hautarzt 2021; 72(8): 692-699.
16. Tappenden P, Carroll C, Stevens JW et al.: Adalimumab for treating moderate-to-severe hidradenitis suppurativa: An evidence review group perspective of a NICE single technology appraisal. Pharmacoeconomics 2017; 35(8): 805-815.
17. Bechara FG, Podda M, Prens EP et al.: Efficacy and safety of Adalimumab in conjunction with surgery in moderate to severe hidradenitis Suppurativa: the SHARPS randomized clinical trial. JAMA Surg 2021; 156(11): 1001-1009.
18. Masson R, Shih T, Jeong C et al.: Omówienie artykułu: Hormonal Treatments in Hidradenitis Suppurativa: A Systematic Review. J Drugs Dermatol 2023; 22(8): 785-794.
19. Hazen PG, Hazen BP: Hidradenitis suppurativa: successful treatment using carbon dioxide laser excision and marsupialization. Dermatol Surg 2010; 36: 208-213.
20. Tierney E, Mahmoud BH, Hexsel C et al.: Randomized control trial for the treatment of hidradenitis suppurativa with a neodymium-doped yttrium aluminium garnet laser. Dermatol Surg 2009; 35: 1188-1198.
21. Wollina U, Koch A, Heinig B et al.: Acne inversa (Hidradenitis suppurativa): a review with a focus on pathogenesis and treatment. Indian Dermatol Online J 2013; 4: 2-11.
22. Rieger UM, Erba P, Pierer G, Kalbermatten DF: Hidradenitis suppurativa of the groin treated by radical excision and defect closure by medial thigh lift: aesthetic surgery meets reconstructive surgery. J Plast Reconstr Aesthet Surg 2009; 62: 1355-1360.
23. Liy-Wong C, Kim M, Kirkorian Y et al.: Hidradenitis Suppurativa in the Pediatric Population. An International, Multicenter, Retrospective, Cross-sectional Study of 481 Pediatric Patients. JAMA Dermatol 2021; 157(4): 385-391.
24. Seivright J, Villa N, Grogan T et al.: Omówienie artykułu: Impact of Pregnancy on Hidradenitis Suppurativa Disease Course: A Systematic Review and Meta-Analysis. Dermatology 2022; 238(2): 260-266.
25. Fitzpatrick L, Hsiao J, Tannenbaum R et al.: Adverse pregnancy and maternal outcomes in women with hidradenitis suppurativa. J Am Acad Dermatol 2022; 86: 46-54.
26. Xu LY, Wright DR, Mahmoud BH et al.: Histopathologic study of hidradenitis suppurativa following long-pulsed 1064-nm Nd:YAG laser treatment. Dermatol 2011; 147: 21-28.
27. Mahmoud BH, Tierney E, Hexsel CL et al.: Prospective controlled clinical and histopathologic study of hidradenitis suppurativa treated with the long-pulsed neodymium:yttriumaluminium-garnet laser. J Am Acad Dermatol 2010; 62: 637-645.
28. Kołodziejczak M, Sudoł-Szopińska I, Wilczyńska A, Bierca J: Utility of transperineal and anal ultrasonography in the diagnostics of hidradenitis suppurativa and its differentiation from a rectal fistula. Postepy Higieny i Medycyny Doswiadczalnej (Online) 2012; 66: 838-842.




