© Borgis - Postępy Nauk Medycznych 6/2014, s. 376-383
*Barbara Sobala-Szczygieł1, Brygida Adamek2, Michał Kukla3, Anna Żurek1, Jarosław Szczygieł4, Lucjan Kępa1, Barbara Oczko-Grzesik1, Grażyna Spausta2, Andrzej Wiczkowski2, Marek Hartleb3
Wpływ interferonu alfa-2b na regenerację hepatocytów w regionie wrotnym i okołocentralnym u szczurów, po częściowej hepatektomii
Influence of interferon-alpha2b on hepatocytes regeneration in periportal and perivenular zones of liver lobules after partial hepatectomy in rats
1Department of Infection Diseases, Medical University of Silesia, Katowice
Head of Department: prof. Anna Boroń-Kaczmarska, MD, PhD
2Department of General Biology, Medical University of Silesia, Katowice
Head of Department: prof. Andrzej Wiczkowski, MD, PhD
3Department of Gastroenterology and Hepatology, Medical University of Silesia, Katowice
Head of Department: prof. Marek Hartleb, MD, PhD
4Upper Silesian Rehabilitation Center “Repty”, Tarnowskie Góry
Head of Center: Krystian Oleszczyk, MD, PhD
Streszczenie
Wstęp. Wątroba jest jednym z niewielu narządów ssaków zdolnym do regeneracji. Stwierdzono, że w zależności od położenia hepatocytów względem naczyń krwionośnych istnieją istotne różnice morfologiczne i funkcjonalne oraz różna wrażliwość tych komórek na działanie czynników toksycznych. Interferony ( IFN ) są cytokinami o działaniu antagonistycznym w stosunku do czynników wzrostu dla komórek somatycznych i krwiotwórczych. Interakcje między poszczególnymi elementami morfotycznymi oraz wpływ czynników chemicznych na regenerujące komórki są na tyle skomplikowane, że nie można użyć modelu in vitro do badania procesu regeneracji. Celem pracy jest ocena wpływu dwóch kolejnych dawek IFN na proliferujące hepatocyty w heterogennych obszarach okołonaczyniowych i okołowrotnych regenerującej wątroby szczura.
Cel pracy. Celem pracy była ocena wpływu interferonu podawanego w dłuższym okresie na proliferację hepatocytów przestrzeni okołowrotnej i okołocentralnej regenerującej wątroby szczura.
Materiał i metody. Sześćdziesiąt samców szczura Wistar podzielono na trzy grupy po 20 zwierząt. Zwierzętom grupy A podano 0,9% NaCl, po 24 h przeprowadzono hepatektomię, a po kolejnych 24 h podano kolejną dawkę NaCl. W grupie B postąpiono analogicznie, podając jednak interferon (IFN)-a2b. W grupie C podano IFN-a2b i przeprowadzono zabieg pozorowany. Następnie zwierzęta wszystkich grup były uśmiercane w czasie 48, 72, 96 i 120 h po zabiegu. Preparaty wybarwiono hematoksyliną i eozyną w celu przeprowadzenia oceny morfologicznej, morfometrycznej oraz poddano procedurze srebrzenia białek argyrofilnych (AgNOR).
Wyniki. Wykazano hamujący wpływ interferonu na regenerujące hepatocyty po zabiegu częściowej hepatektomii. Efekt ten jest najsilniej wyrażony po 48 godzinach. Analiza średnich wartości powierzchni jąder hepatocytów wykazuje większe jądra w komórkach przestrzeni okołowrotnej w 48 godzinie po hepatektomii, w kolejnych przedziałach czasowych (w 96 godzinie) większe jądra obserwowano w komórkach okołocentralnych. Zanotowano wyższe wartości sumarycznej powierzchni AgNOR jąder komórek proliferujących przestrzeni okołowrotnej we wszystkich przedziałach czasowych w porównaniu z komórkami nieproliferującymi.
Wnioski. Interferon α hamuje aktywność proliferacyjną hepatocytów do 72 godzin po PH. Regeneracja hepatocytów strefy okołocentralnej pod wpływem interferonu jest opóźniona o 48 godzin w porównaniu do hepatocytów strefy okołowrotnej.
Summary
Introduction. The liver is one of few organs in mammals with the ability to regenerate. However, there are morphological and functional differences and also different susceptibility of hepatocytes to harmful factors depending on their location in relation to blood vessels. Interferons (IFNs) are antagonists of growth factors for somatic and hematopoietic cells. The system of interactions among particular morphotic elements and chemical factors is very complex and that is why it is impossible to use in vivo pattern of the process of regeneration under in vitro conditions.The aim of the study is the evaluation of the long-term influence of two successive doses of IFN on proliferating hepatocytes in heterogenic perivenous and periportal areas of liver lobules in rats.
Aim. The aim of the study was the evaluation of the long-term interferon influence on proliferating hepatocytes with respect to periportal and perivenular lobular zone in rats.
Material and methods. Sixty male Wistar rats, three-month-old were divided into three groups of 20 animals each. The first group (group A) was injected with normal saline, 24 hours later 70% hepatectomy was performed and after the next 24 hours the second dose of normal saline was administered. A similar schedule was applied in the second group (group B) injected with interferon (INF)-α2b instead. The third group (group C) was injected with IFN-α2b and sham operated. Then the rats were killed at 48, 72, 96 and 120 hours after the surgery. The liver sections were stained with hematoxylin and eosin in order to perform a morphological and morphometric assessment and silver-stained for nucleolar organizer regions (AgNORs). Nucleolar organizer regions are segments of DNA that transcribe to ribosomal RNA and are situated on short arms of the acrocentric chromosomes 13, 14, 15, 21 and 22. The number of NORs is related to the cell cycle and the quantity of interphase NORs increases in cycling cells from the early G1 phase to the late S phase.
Results. The results showed the inhibitory influence of interferon on proliferating hepatocytes after partial hepatectomy, with the maximum after 48 hours. Analysis of mean values of the hepatocyte nuclear area for cells shows larger nuclei in periportal cells at 48 hours after PH, but larger nuclei in perivenous cells at 96 hours after PH. Higher values of the total area of AgNOR per nucleus were shown in periportal hepatocytes in all time intervals for proliferating cells and compared to non-proliferating cells.
Conclusions. Interferon α inhibits the proliferative activity of hepatocytes up to 72 hours after PH. The reaction of perivenous hepatocytes to interferon is delayed by 48 hours compared to periportal hepatocytes.

INTRODUCTION
The liver is one of few organs in mammals with the ability to regenerate. However, there are morphological and functional differences and also different susceptibility of hepatocytes to harmful factors depending on their location in relation to blood vessels. The process of regeneration starts in periportal regions and then gradually spreads towards perivenous hepatocytes.
The system of interactions among cytokines, growth factors, hormones, hepatocytes and non-parenchymal cells (1-3) is very complex and that is why it is impossible to use in vivo pattern of the process of regeneration under in vitro conditions. A partial hepatectomy (PH) by the method of Higgins and Anderson is a pattern of regeneration, which is universally recognized and used in the empirical studies. A removal of 70% of the liver stimulates the process of regeneration and one-third of the liver that is left after PH is sufficient to sustain life after the surgery. In the empirical studies it is assumed as a normally regenerating liver. The system of interactions among particular morphotic elements and chemical factors is very complex and that is why it is impossible to use in vivo pattern of the process of regeneration under in vitro conditions (4).
Interferons (IFNs) are antagonists of growth factors for somatic and hematopoietic cells. In cell cultures exposed to INF, disturbances in the mitotic cycle are observed – a longer time of transition from G0 and G1 to S phase and prolongation of S phase. A complete inhibition of mitosis was also seen, resulting from inhibition of ornithine decarboxylase synthesis, which is a key enzyme for polyamine and DNA synthesis (3, 4). Administration of two doses of interferon prior to and during PH influences cell proliferation through inhibition of polyamine and liver enzyme synthesis (e.g. ornithine decarboxylase), which are key factors in the process of regeneration (1, 2).
Taking above into consideration, in our study we divided liver cells according to their location in the liver lobule, treating periportal and perivenous hepatocytes as two different population of cells.
AIM
The aim of the study is the evaluation of the long-term influence of two successive doses of IFN on proliferating hepatocytes in heterogenic perivenous and periportal areas of liver lobules in rats.
MATERIAL AND METHODS
The study was performed on 60 three-month-old Wistar rats of mean weight 300 g. The animals were randomly assigned to one of 3 groups, 20 rats each. The rats were maintained on rat chow and water under standard conditions. During PH the left and middle lobe of the liver were removed. The study protocol was approved by the Local Ethics Committee for Animal Experiments of the Medical University of Silesia (No. 1/02; 19.02.2002). In all groups the animals were killed in subgroups of 5 at 48, 72, 96 and 120 hours after the surgery or sham operations (fig. 1).
Group A1 (Hepatectomy + NS)
Each rat was injected subcutaneously with normal saline (NS) in the shoulder girdle region. After 24 hours PH under general anaesthesia was performed. After the next 24 hours the second dose of normal saline was administered. Liver tissue samples were collected during PH and at autopsy.
Group B (Hepatectomy + Interferon α)
The animals were injected subcutaneously with the first dose of interferon (Intron A, Shering-Plough, 5 MU/100 ml NS) in the shoulder girdle region. After 24 hours PH under general anaesthesia was performed. After the next 24 hours the second dose of interferon was administered.
Group C (Laparotomy + Interferon α)
The animals were injected subcutaneously with the first dose of interferon (Intron A, Shering-Plough, 5 MU/100 ml NS) in the shoulder girdle region. After 24 hours the rats were anaesthetised with ketamine and sham operated. Sham operations consisted of midline laparotomy and gentle manipulation of the liver in the same manner as resected animals. After the next 24 hours the second dose of interferon was administered.
60 liver tissue samples obtained at autopsy and 40 samples obtained during PH were fixed in 4% formaldehyde solution and embedded in paraffin. At least 6 slides, each containing 6 sections, were prepared from each paraffin block. The slides were stained with hematoxylin and eosin in order to perform a morphological and morphometric assessment by evaluating the area of cells and cell nuclei. The obtained sections underwent also one-step silver-staining for nucleolar organizer regions (AgNOR) by Ploton and colleagues with modifications recommended by the Committee on AgNOR Quantitation within the European Society of Pathology (5). NORs are segments of DNA that encode ribosomal RNA directly related to protein synthesis and cellular proliferation. Active NORs are associated with a subset of specific proteins (C23 and B23), that reacts with silver nitrate (AgNORs), appearing as secondary constrictions in acrocentric chromosomes during mitosis or as black dots in interphase nuclei. Being more numerous in actively proliferating cells, AgNORs quantity increases during G1 phase to reach a maximum during S phase. The amount of AgNOR proteins can be a marker of proliferation, because this amount is related to cell cycle phases the quantification of the nucleolar organizer regions (NORs) reflecting the proliferative capacity of cells or indicating the degree of malignancy.
The data were collected in MS Excel 2000 and analyzed using Statistica 7.1 PL software pack. For each parameter, measures of descriptive statistics were calculated (mean, standard deviation). Then, the Shapiro-Wilk test for normality was performed. Comparison between groups of parameters with normal distribution and parameters with non-normal distribution were performed using Student’s t-test and Mann-Whitney U test, respectively.
A comparison of frequency of occurrence of the given classes between groups was performed using a chi-square test. When the assumptions of chi-square test were not met, comparison of contingency tables was performed using Fisher’s exact test.
RESULTS
Testing the significance of differences between mean values of measured parameters for perivenous and periportal cells, higher values of the surface area of hepatocytes regenerating under the influence of interferon were found in perivenous cells compared to periportal cells at 48, 72 and 96 hours after the surgery (fig. 1, 2).
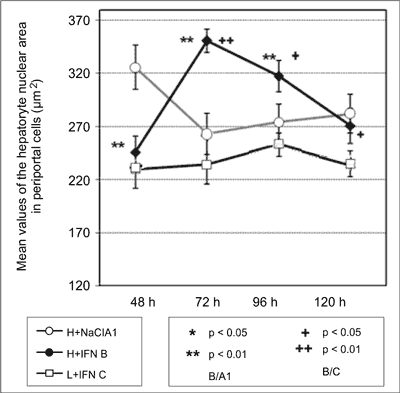
Fig. 1. Values of the hepatocyte nucleat area in periportal cells.
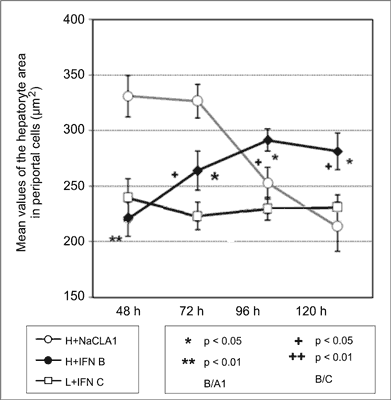
Fig. 2. Values of the hepatocyte area in periportal cells.
The analysis of mean values of the hepatocyte nuclear area in perivenous and periportal cells shows a significant difference between these two groups. Perivenous cells in group A have smaller nuclei at 48, 72 and 96 hours after PH compared to periportal cells. Comparison of the mean values of the estimated parameter for cells in group B shows larger nuclei in periportal cells at 48 hours after PH, but larger nuclei in perivenous cells at 96 hours after PH (fig. 3, 4).

Fig. 3. Values of the hepatocyte nucleat area in perivenous cells.
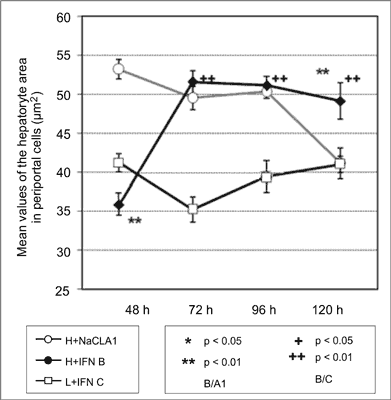
Fig. 4. Values of the hepatocyte nucleat area in periportal cells.
Higher values of the total area of AgNOR per nucleus were shown in periportal hepatocytes in all time intervals for proliferating cells in group A and B and compared to non-proliferating cells in group A0 and C. No differences between non-proliferating cells were found (fig. 5, 6).
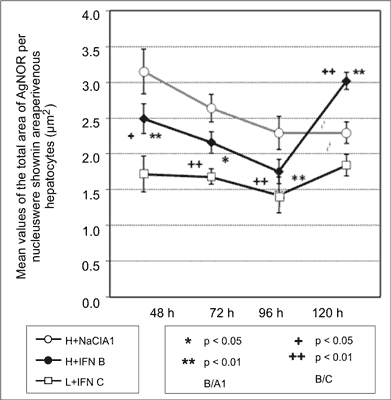
Fig. 5. Values of the total area of AgNOR per nucleus were shown in perivenous.

Fig. 6. Values of the total area of AgNOR per nucleus in periportal hepatocytes.
DISCUSSION
The results of the observation of liver regeneration suggest that the process of cell division in regenerating liver depends not only on time but also on the location of cells in the liver. A higher proliferative capacity is observed in zone 1 compared to zone 3 of the liver acinus (6). Studies by Segal et al. and by Arber suggest constant proliferation of cells located where the canals of Hering connect with bile ductules. The authors used tritiated thymidine to prove that cells migrate toward the central vein and simultaneously differentiate into mature hepatocytes or cholangiocytes (6, 7). Availability of nutritional and regulatory substances like growth factors, hormones and cytokines, is an additional factor differentiating regenerative capacity of liver cells in particular zones of the liver acinus (8, 9). Taking the above into consideration, in our study we divided liver cells depending on their location, treating periportal and perivenous hepatocytes as 2 different population of cells.
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Favre C, Carnovale CE, Monti JA, Carrillo MC: Inhibition by interferon α-2b of rat liver regeneration: effect on ornithine decarboxylase and total protein synthesis. Biochem Pharmacol 2001; 61: 1587-1593.
2. Wong S, Gauthier T, Kaita KD, Minuk GY: The differential effects of three forms of interferon alfa on hepatic regeneration after partial hepatectomy in the rat. Hepatology 1995; 21: 883-886.
3. Kukla M, Berdowska A, Stygar D et al.: Serum FGF21 and RGP4 levels in patient with chronic hepatitis C. Scand J Gastroenterol 2012; 47(8-9): 1037-1047.
4. Higgins GM, Anderson RM: Experimental pathology of the liver. I. Restoration of the liver of the white rat following partial surgical removal. Arch Pathol 1931; 12: 186-202.
5. Ploton D, Menager M, Jeannesson P et al.: Improvement in the staining and in the visualization of the argyrophilic proteins of the nucleolar organizer region at the optical level. Histochem J 1986; 18: 5-14.
6. Diehl AM, Rai RM: Liver regeneration 3: Regulation of signal transduction during liver regeneration. The FASEB Journal 1996; 10: 215-227.
7. Zajicek G: Hepatocytes and intrahepatic bile duct epithelium originate from a common stem cell. Gastroenterology 1991; 100: 582-583.
8. Fausto N: Liver regeneration. J Hepatol 2000; 32: 19-31.
9. Diehl AM: The role of cytokines in hepatic regeneration. Curr Opin Gastroenterol 1997; 13: 525-533.
10. Lim R, Knight B, Patel K et al.: Antiproliferative effects of interferon alpha on hepatic progenitor cells in vitro and in vivo. Hepatology 2006; 43(5): 1074-1083.
11. Theocharis SE, Margeli AP, Tsokos MG: α2b-interferon inhibits rat liver regeneration after partial hepatectomy without affecting thymidine kinase activity. J Lab Clin Med 1995; 125: 588-596.
12. Theocharis SE, Margeli AP, Skaltsas SD et al.: Effect of interferon-α2b administration on rat liver regeneration after partial hepatectomy. Dig Dis Sci 1997; 42: 1981-1986.
13. Kocić G, Kocić R, Vlahović P et al.: Different responses of rat liver adenosine metabolizing enzymes during in vivo and in vitro treatment with interferon-α2b. J Viral Hepat 1998; 5: 353-356.
14. Gabriel A: Ocena wybranych czynników prognostycznych z zakresu epidemiologii, histopatologii, immunohistochemii oraz cytofluorometrii w przewidywaniu dynamiki i postępu włóknienia u chorych na przewlekłe zapalenie wątroby typu C. Rozprawa habilitacyjna. Śląska Akademia Medyczna, Katowice 2002.
15. Murray AB, Strecker W, Silz S: Ultrastructural changes in rat hepatocytes after partial hepatctomy and comparison with biochemical results. J Cell Sci 1981; 50: 433-448.
16. Vizzotto L, Vartemati M, Degna CT, Asenia P: Liver transplantation in man: morphometric analysis of the parenchymal alterations following cold ischaemia and warm ischaemia/reperfusion. J Anat 2002; 198: 603-610.
17. Spausta G, Ciarkowska J, Wiczkowski A, Adamek B: Wpływ Intronu A na parametry morfometryczne hepatocytów regenerującej wątroby szczura. Ann Acad Med Siles 2002; 50: 50-51.
18. Sirri V, Roussel P, Hernandez-Verdun D: The AgNOR proteins qualitative and quantitative changes during the cell cycle. Micron 2000; 31: 121-126.
19. Trere D, Farabegali F, Cancellieri A et al.: AgNOR area in interphase nuclei of human tumors correlates with the proliferative activity evaluated by bromodeoxyuridine labeling and Ki-67 immunostaining. J Pathol 1991; 165: 53-59.
20. Azzaroli F, Accogli E, Nigro G et al.: Interferon plus ribavirin and interferon alone in preventing hepatocellular carcinoma: A prospective study on patients with HCV related cirrhosis. World J Gastroenterol 2004 Nov 1; 10(21): 3099-3102.
21. Kukla M, Beredowska A, Gabriel A et al.: Association between hepatic angiogenesis and serum adipokine profile in non-obese chronic hepatitis C patients. Pol J Pathol 2011; 62(4): 218-228.
22. Kukla M: Angiogenesis: a phenomen chich aggravates chronic liver disease progression. Hepatol Int 2013; 7(1): 4-12.
23. Skonieczka K, Mucha B, Soszyńska K et al.: Analiza regionów jąderkotwórczych (AgNOR) w białaczkach. Acta Haemat Pol 2004; 35: 537-548.
24. Lorand-Metze I, Carvalho MA, Metze K: Relationship between morphometric analysis of nucleolar organizer regions and cell proliferations in acute leukemia. Cytometry 1998; 32: 51-56.
25. Martin CA, Surur JM, Garcia MN et al. : Comparative study of DNA synthesis and nucleolar organizer regions of sinusoid litoral cells in maus regenerating liver. Biocell 2005, 29: 33-38.






