© Borgis - Postępy Nauk Medycznych 6/2014, s. 400-404
*Magdalena Rogalska-Płońska, Anna Grzeszczuk, Tomasz Szulżyk, Aldona Kowalczuk-Kot, Robert Flisiak
Hipertriglicerydemia indukowana nadużywaniem alkoholu u chorego z koinfekcją HIV/HCV – opis przypadku
Alcohol induced severe hypertriglyceridemia in HIV/HCV coinfected patient – case report
Department of Infectious Diseases and Hepatology, Medical University of Białystok
Head of Department: prof. Robert Flisiak, MD, PhD
Streszczenie
Zaburzenia lipidowe mogą mieć charakter pierwotny lub wtórny, a nadużywanie alkoholu jest jedną z głównych przyczyn wtórnej dyslipidemii. Wpływ alkoholu na metabolizm lipidów polega między innymi na pobudzeniu syntezy trójglicerydów i lipoprotein o bardzo małej gęstości, jak również stymulacji lipolizy w tkance tłuszczowej. Zakażenie HIV również predysponuje do zaburzeń gospodarki lipidowej na skutek bezpośredniego działania wirusa czy też działania leków antyretrowirusowych. Inhibitory protezy wiążą się z największym ryzykiem hipertriglicerydemii i hipercholesterolemii, spośród wszystkich leków antyretrowirusowych. Natomiast koinfekcja z HCV rozważana jest jako czynnik chroniący przed znaczną dyslipidemią. Hypertriglicerydemia jest czynnikiem ryzyka chorób sercowo-naczyniowych, a jej ciężka postać ze stężeniem trójglicerydów przekraczającym 1000 mg/dL, wiąże się z ryzykiem ostrego zapalenia trzustki. U 36-letniego chorego z koinfekcją HIV/HCV rozpoznano ciężką hipertriglicerydemię przekraczającą 8000 mg/dL. Ustalono, że pacjent okresowo nadużywa alkohol, rozpoczęcie leczenia fenofibratami i zmiana leków antyretrowirusowych nie spowodowały istotnego obniżenia stężenia lipidów. Najwyższe stężenia cholesterolu i trójglicerydów obserwowano w okresach nadużywania alkoholu, co wiązało się z jednoczesnym narastaniem aktywności GGT. Reasumując, nie wydaje się, aby koinfekcja HIV/HCV miała wpływ na przebieg ciężkiej hipertriglicerydemii indukowanej spożywaniem alkoholu.
Summary
Alcohol use is one of the most prominent causes of dyslipidemia. The effects of alcohol on lipid metabolism are diverse: it induces synthesis of triglycerides, formation of very low density cholesterol and stimulates lipolysis in fatty tissue. HIV infection also predisposes to lipid disturbances resulting from direct influence of the virus as well as from the action of antiretroviral drugs, however coinfection with HCV is considered to be protective against significant dyslipidemia in some cases. Hypertriglyceridemia is a risk factor of cardiovascular disease and severe form is considered to be at increased risk for acute pancreatitis. 36-years old HIV/HCV coinfected patient was diagnosed with severe hypertriglyceridemia exciding 8000 mg/dL. Periodic alcohol abuse was stated. Fenofibrate initiation and antiretroviral therapy switch did not influence the lipid levels significantly. Increase in lipids levels occurred simultaneously with periods of alcohol abuse. In conclusion, HIV/HCV coinfection does not seem to influence severe dyslipidemia induced by alcohol use.

Introduction
Lipid disorders seen in human immunodeficiency virus (HIV) infected patients include: elevated concentration of triglycerides (TGs) and total cholesterol (TC), decreased high-density lipoprotein (HDL) and variable changes in low-density lipoprotein (LDL). The mechanism of lipid disturbances accompanying HIV infection is unexplained and probably multifactorial (1). While lipid profile abnormalities have been described before the HAART (Highly Active Antiretroviral Therapy) era, hyperlipidemia associated with antiretroviral (ARV) drugs use is now more common and more severe. Protease inhibitors (PIs) create the greatest risk of hypertriglyceridemia and hypercholesterolemia among the all classes of ARV drugs (2, 3).
In general, lipid disorders can be divided into primary and secondary defects of lipid metabolism. One of the most prominent secondary causes of hypertriglyceridemia is excessive alcohol use. The effects of alcohol on lipid metabolism are diverse and includes stimulation of TG synthesis, very low-density lipoprotein (VLDL) formation, and lipolysis in fatty tissue (4, 5).
Although hypertriglyceridemia is often asymptomatic, it is a risk factor for cardiovascular disease. Patients presenting with severe hypertriglyceridemia are at increased risk for acute pancreatitis (6, 7).
Here we present a case report of severe hypertriglyceridemia in HIV/HCV (hepatitis C virus) co-infected patient with a history of chronic alcohol abuse. Informed consent has been obtained from the patient.
Case report
A 36-years old Caucasian male identified 6 years earlier with HIV and HCV without any other health complaints and who declared good adherence to ARV treatment (saquinavir, ritonavir and abacavir/lamivudine) for the past 2 years, came to the clinic for a routine blood test. He was subsequently diagnosed to have dyslipidemia with severe hypertriglyceridemia (2417 mg/dL) based on current American Association of Clinical Endocrinologists’ (AACE) guidelines (8). The concentrations of TC, HDL and LDL were 537, 44 and 493 mg/dL, respectively. Laboratory tests also revealed elevated activities of alanine (ALT) and aspartate (AST) aminotransferases (80 U/L and 62 U/L respectively), gamma glutamyl transferase (GGT – 416 U/L) and increased mean corpuscular volume (MCV – 101fl). The most recent lymphocyte counts revealed: CD3+ – 1266 cells/μl, CD4+ – 563 cells/μl and CD8+ – 728 cells/μl. HIV-RNA was undetectable. Blood pressure was normal and patient body mass index was 24. It was further revealed that he had been smoking about 20 cigarettes a day and drinking a moderate amount of alcohol over the past weeks. Physical examination was unremarkable. The patient refused admission to the hospital. Fenofibrate was prescribed and he was advised to introduce low-fat diet as well as eliminate alcohol and cigarettes. In the subsequent months the patient remained asymptomatic, yet despite the hypolipidemic treatment, his serum was lipemic with lipid concentrations as follows: TG – 6011 and 8396 mg/dL, TC – 896 and 990 mg/dL. ARV treatment was modified and atazanavir was introduced instead of saquinavir. Four months later, the patient was admitted to the hospital. TG concentration at a day of admission was 7199 mg/dL and total cholesterol – 1018 mg/dL, GGT activity – 795 U/L, amylase activity and CRP concentration were in norm. Abdominal ultrasonography revealed hepatosteatosis, although no changes in the pancreas, gallbladder, bile ducts and other organs were found. Gastroscopy showed slight mucosal inflammation of the antrum. Fenofibrate treatment was maintained and during the subsequent weeks, TG concentration decreased to 475 mg/dL, TC to 320 mg/dL, GGT activity to 338 U/L. After leaving the hospital, the patient made monthly visits to the HIV outpatient center. During the past several months lipid levels and GGT activity were again found to increase. The patient admitted that he periodically consumed excessive amounts of alcohol during this time. He refused both hospital readmission and treatment for alcohol dependence. Successive laboratory test results showed simultaneous increases in the concentrations of TG and TC concentrations as well as GGT activity, a marker of toxic liver damage (fig. 1).
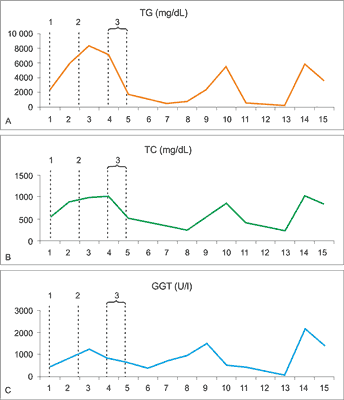
Fig. 1. A. Triglyceride (TG), B. total cholesterol (TC) concentrations and C. gamma glutamyl transpeptidase (GGT) activity during 16 months observation. 1. Initiation of fenofibrate treatment; 2. switch of antiretroviral therapy; 3. hospitalization.
Discussion
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Lake JE, Currier JS: Switching antiretroviral therapy to minimize metabolic complications. HIV Therapy 2010; 4: 693-711.
2. Grunfeld C, Kotler DP, Shigenaga JK: Circulating interferon-alpha levels and hypertriglyceridemia in the acquired immunodeficiency syndrome. American Journal of Medicine 1991; 90: 154-162.
3. Clotet B, van der Valk M, Negredo E, Reiss P: Impact of nevirapine on lipid metabolism. Journal of Acquired Immune Deficiency Syndrome 2003; 34 (suppl. 1): 79-84.
4. Bessembinders K, Wielders J, van de Wiel A: Severe hypertriglyceridemia influenced by alcohol (SHIBA). Alcohol 2011; 46: 113-116.
5. Brunzell JD: Clinical practice. Hypertriglyceridemia. New England Journal of Medicine 2007; 357: 1009-1017.
6. Yuan G, Al-Shali KZ, Hegele RA: Hypertriglyceridemia: its etiology, effects and treatment. Canadian Medical Association Journal 2007; 176: 1113-1120.
7. Ewald N, Kloer HU: Severe hypertriglyceridemia: an indication for apheresis? Atherosclerosis. Supplements 2009; 10: 49-52.
8. Jellinger PS, Smith DA, Mehta AE et al.: American Association of Clinical Endocrinologists’ Guidelines for Management of Dyslipidemia and Prevention of Atherosclerosis: executive summary. Endocrine Practice 2012; 18: 269-293.
9. Baraona E, Lieber CS: Alcohol and lipids. Recent Developments in Alcoholism 1998; 14: 97-134.
10. Schneider J, Liesenfeld A, Mordasini R et al.: Lipoprotein fractions, lipoprotein lipase and hepatic triglyceride lipase during short-term and long-term uptake of ethanol in healthy subjects. Atherosclerosis 1985; 57: 281-291.
11. Hannuksela ML, Rämet ME, Nissinen AE et al.: Effects of ethanol on lipids and atherosclerosis. Pathophysiology 2004; 10: 93-103.
12. Riddler SA, Smit E, Cole SR et al.: Impact of HIV infection and HAART on serum lipids in men. JAMA: the journal of the American Medical Association 2003; 289: 2978-2982.
13. Grinspoon S, Carr A: Cardiovascular risk and body-fat abnormalities in HIV-infected adults. New England journal of medicine 2005; 352: 48-62.
14. Wanke C, Gerrior J, Hendricks K et al.: Alterations in lipid profiles in HIV-infected patients treated with protease inhibitor therapy are not influenced by diet. Nutrition in clinical practice 2005; 20: 668-673.
15. El-Sadr WM, Mullin CM, Carr A et al.: Effects of HIV disease on lipid, glucose and insulin levels: results from a large antiretroviral-naive cohort. HIV Medicine 2005; 6: 114-121.
16. Rasheed S, Yan JS, Lau A, Chan AS: HIV replication enhances production of free fatty acids, low density lipoproteins and many key proteins involved in lipid metabolism: a proteomics study. PLoS One 2008; 3: e3003.
17. Yeni P: Update on HAART in HIV. Journal of hepatology 2006; 44 (suppl. 1): S100-103.
18. Sax PE: Strategies for management and treatment of dyslipidemia in HIV/AIDS. AIDS Care 2006; 18: 149-157.
19. Zhou H, Pandak WM Jr, Lyall V et al.: HIV protease inhibitors activate the unfolded protein response in macrophages: implication for atherosclerosis and cardiovascular disease. Molecular pharmacology 2005; 68: 690-700.
20. Calza L, Manfredi R, Chiodo F: Dyslipidaemia associated with antiretroviral therapy in HIV-infected patients. The journal of antimicrobial chemotherapy 2004; 53: 10-14.
21. Malan N, Krantz E, David N et al.: Efficacy and safety of atazanavir-based therapy in antiretroviral therapy naive HIV-1 infected subjects, both with and without ritonavir: 48 week results for AI424-089. AIDS Research and Therapy 2009; 6: 3.
22. Cooper CL, Mills E, Angel JB: Mitigation of antiretroviral-induced hyperlipidemia by hepatitis C virus co-infection. AIDS 2007; 21: 71-76.
23. Visnegarwala F, Chen L, Raghavan S, Tedaldi E: Prevalence of diabetes mellitus and dyslipidemia among antiretroviral naive patients co-infected with hepatitis C virus (HCV) and HIV-1 compared to patients without co-infection. The Journal of infection 2005; 50: 331-337.
24. Hokanson JE, Austin MA: Plasma triglyceride level is a risk factor for cardiovascular disease independent of high-density lipoprotein cholesterol level: a metaanalysis of population-based prospective studies. Journal of cardiovascular risk 1996; 3: 213-219.
25. Yuan G, Al-Shali KZ, Hegele RA: Hypertriglyceridemia: its etiology, effects and treatment. Canadian Medical Association journal 2007; 176: 1113-1120.
26. Ewald N, Kloer HU: Treatment options for severe hypertrigliceridemia (SHTG): the role of apheresis. Clinical research in cardiology supplements 2012; 7 (suppl. 1): 31-35.
27. Lo J: Dyslipidemia and lipid management in HIV-infected patients. Current opinion in endocrinology, diabetes and obesity 2011; 18: 144-147.
28. Jellinger PS, Smith DA, Mehta AE et al.: American Association of Clinical Endocrinologists’ Guidelines for Management of Dyslipidemia and Prevention of Atherosclerosis. Endocrine practice 2012; 18 (suppl. 1): 1-78.

