© Borgis - Postępy Nauk Medycznych 2/2011, s. 94-103
*Barbara Bobek-Billewicz
Diagnostic imaging in contemporary oncology
Diagnostyka obrazowa we współczesnej onkologii
Zakład Radiodiagnostyki Centrum Onkologii – Instytut im. M. Skłodowskiej-Curie, Oddział w Gliwicach
Kierownik Zakładu: doc. dr hab. med. Barbara Bobek-Billewicz
Streszczenie
Radiologia i diagnostyka obrazowa odgrywają istotną rolę na wszystkich etapach leczenia chorych na nowotwory złośliwe. Właściwe wykorzystanie metod diagnostyki obrazowej nie tylko poprawia wyniki leczenia ale także ma pozytywny wpływ na koszty leczenia.
W wyborze metody diagnostycznej optymalnej dla chorego i problemu klinicznego decydujący głos powinien mieć radiolog.
Przez wiele lat badania radiologiczne oraz ultrasonograficzne były obrazowaniem strukturalnym, przedstawiającym cechy morfologiczne tkanek prawidłowych i rozwijających się w nich procesów patologicznych. Nowe metody radiologii i diagnostyki obrazowej pozwalają na obrazowanie strukturalne i czynnościowe także nowotworów. Obrazowanie czynnościowe dostarcza danych ważnych dla leczenia chorych na nowotwory złośliwe. MR – najlepiej z dostępnych obecnie metod – łączy obrazowanie morfologiczne z czynnościowym i w pewnym zakresie umożliwia obrazowanie molekularne.
Do głównych zastosowań radiologii i diagnostyki obrazowej we współczesnej onkologii należą:
1. wykrycie i rozpoznanie nowotworu
2. określenie stopnia zaawansowania choroby nowotworowej
3. pomoc w wyborze i wskazanie miejsca pobrania materiału do badania cytologicznego/histopatologicznego
4. wsparcie leczenia (np. planowanie radioterapii, miejscowe leczenie nowotworów pierwotnych i wtórnych)
5. monitorowanie odpowiedzi na leczenie
6. ocena skuteczności leczenia
7. monitorowanie po zakończeniu leczenia umożliwiające wczesne wykrycie wznowy/rozsiewu
8. wykrywanie powikłań po leczeniu
9. udział w ocenie działania i skuteczności nowych leków
Summary
Diagnostic imaging has an important role in all stages of management of oncologic patients and its proper application leads not only to improved treatment results, but also to increased cost-effectiveness.
The great number of currently available imaging modalities causes that this just the radiologist is to have decisive opinion in selection of the diagnostic method of choice for the given patient, diagnostic problem or clinical issue.
For many years, radiologic studies were structural imaging, presenting morphological features of normal tissues and pathologic processes, including neoplasms in these structures. New techniques of radiology and diagnostic imaging allow for structural and functional imaging of a neoplasm. Functional diagnostic imaging provides important data for treatment of patients with malignancies. MRI best combines – among the currently available methods – possibilities of morphological and functional imaging and – to some extent – it offers a possibilities of molecular imaging
Main applications of diagnostic imaging in the modern oncology are:
1. detection and diagnosis of the neoplasm (at the earliest stage of the disease)
2. tumor staging
3. biopsy (image guided biopsy)
4. treatment support
5. monitoring and prediction of treatment response
6. assessment of treatment efficiency
7. follow-up
8. detection and monitoring of early and late complications after treatment
9. supporting clinical trials of new therapeutic agents or in oncology drug development

Diagnostic imaging has an important role in all stages of management of oncologic patients and its proper application leads not only to improved treatment results, but also to increased cost-effectiveness.
Main applications of diagnostic imaging in oncology are:
Detection and diagnosis of the neoplasm (at the earliest stage of the disease)
Detection, i.e. visualisation and noticing of a lesion is the first step. Detection of a neoplasm depending on the quality of the device and perception by a radiologist has recently sometimes been aided by CAD (computer aided detection) systems. Clinically, CAD systems are used in mammography, computed tomography of the lungs to detect nodules and in CT-colonography. CAD reduces the perception error, but it cannot affect interpretation errors.
The next one is interpretation, characterisation the lesion – differential diagnosis of benign and malignant lesions.
There is a special role in neoplasm detection to be played by screening. Diagnostic imaging is used mainly in breast cancer screening.
Breast cancer screening by X-ray mammography is a procedure of well-documented effectiveness in reducing breast cancer mortality. Mammography?s sensitivity ranges – depending especially on the breast structure – from 68 to 93% and it is higher for diagnostic mammography than in the case of screening mammography.
About 10% of cancers are mammographically occult, even if they are palpably detectable (1).
The most frequently, cancer remains undetected in dense, glandular breasts, and this is why it is more and more commonly argued that another screening method is needed. This method cannot be hand-held ultrasound. Tomosynthesis and automated volume breast US are currently tested for their application in breast cancer screening. Early detection of lung cancer by low-dose CT is still rather the subject of clinical studies.
Detection of a neoplasm, interpretation of the image and characteristics of the found lesion requires usually a multimodality approach and even then, biopsy is often necessary for the final diagnosis.
It can be probably said that many modern methods of diagnostic imaging are more sensitive than specific, e.g. only 25-45% of lesions qualified mammographically for biopsy are confirmed to be cancers (2).
Tumor staging
A. Size of the tumour
Traditionally used uni- and bidimensional methods are insufficient and currently volume of the tumour is calculated more and more frequently. Regardless of the applied method, it is necessary to delineate the tumour in order to calculate its size. Delineating of neoplastic infiltration in diagnostic imaging may be difficult and location of the tumour-normal tissue border changes depending on the used modality (3).
Differences in lesion size between morphological and metabolic imaging (1HMRS) have been reported in brain tumours (4). This is also essential for planning surgical treatment as well as radiotherapy.
B. delineation of the relationship between the tumor and adjacent organs
C. metastases to lymph nodes and distant metastases. Apart from the classical 18FDG PET/CT whole body imaging, whole body MRI is applied more and more often. It must be remembered that metastases < 2 mm are currently impossible to visualise in diagnostic imaging.
Biopsy: selection of the biopsy site and guiding (imaging guided biopsy).
Presently, all types of biopsy are guided by imaging modalities. Imaging is especially important in the process of selecting the sampling site in brain tumours considering their histopathologic heterogeneity and difficulties in obtaining larger specimens.
Such an examination should include classical MRI, but also DWI-MR (diffusion weighted imaging), 1HMRS, angiogenesis imaging.
Treatment support – imaging guided treatment
4A. Radiotherapy planning
No other method of oncologic treatment is equally dependant on imaging as radiotherapy which is impossible without x-ray and/or CT images. MRI is especially valuable in IMRT or radiosurgery, because it allows for better assessment of borders of the tumour, adjacent structures and critical organs
4B. Surgery planning
Imaging studies have a special role in surgical treatment of brain tumours. Defining the tumour?s location and planning access route require structural studies, but also mapping of:
– eloquent cortex area (fMRI) – nowadays by the BOLD method (Blood Oxygenation Level Dependent),
– white matter tracts – DTI method (Diffusion Tensor Imaging).
Neuronavigation using MRI and CT sections improves markedly precision of the surgical procedure and is a more and more common tool.
As Nimsky wrote in 2005: “Despite decreasing non-eloquent space for resection, it is out of doubt that today tumours are operated, which we thought we should not operate on only 10 years ago” (5).
4C. Local treatment of primary and metastatic tumours intra-vessel embolisation of malignancies, ablation, radiosurgery.
MRI-guided (high-intensity) focused ultrasound (FUS, HIFU) – method based on delivering big quantity of acoustic energy to the selected structure (neoplastic tumour) which causes heating, acoustic cavitation and mechanical force (6). In this procedure, MRI serves not only planning (delineating of the tumour) and monitoring of treatment effects, but it also provides real-time in vivo body temperature maps. On one hand, this is crucial for efficient ablation and on the other hand – to prevent damage to normal tissues. This method is applied in clinical practice to treat uterine fibroids, while its application in management of other tumours (brain, liver, breast, kidneys, prostate) and in pain palliation in bone metastases is under investigation.
Monitoring and prediction of treatment response (fig. 1, 2).
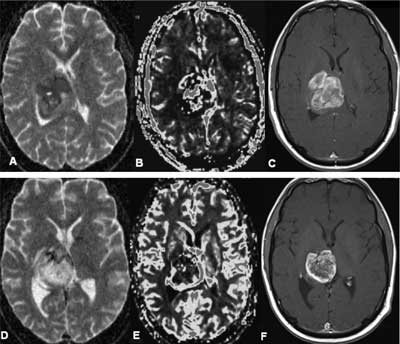
Fig. 1. PNET – treatment response A, B, C: before radio-chemotherapy (May 2009): (A) – ADC map (mean ADC 0,4-0,5 x 10–3 mm2/s), (B) – blood volume map; rCBV > 3,5; (C) – T1-weighted CE image D, E, F: after radio- chemotherapy (August 2009): (D) – ADC map – mean ADC 0,9-1,1 x 10–3 mm2/s is higher in comparison with previous examination, (E) – blood volume map; rCBV 1,5-2 is lower than in previous examination, (F) – T1-weighted CE image.
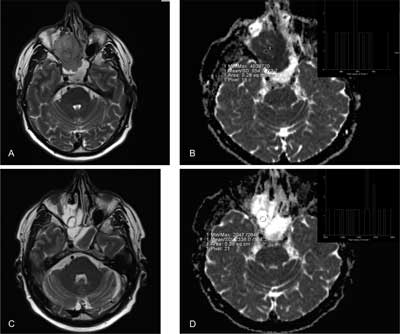
Fig. 2. Sinonasal undifferentiated cancer – treatment response A, B before chemotherapy: A – T2-weighted imaging, B – ADC map (mean ADC = 0,5 x 10–3 mm2/s) C, D during chemotherapy: A – T2-weighted imaging, B – ADC map: ADC had increased significantly (mean ADC = 1,8-2 x 10–3 mm2/s).
Assessment of treatment efficiency – diagnosis/exclusion of presence of a persistent neoplasm after completed therapy. The radiologist?s knowledge of treatment date and treatment methods applied in the patient is necessary to make this assessment. As in the case of early detection of a recurrence, it is necessary for the radiologists to know the image of lesions expected after the applied treatment and of complications.
Follow-up
A. Identification of relapse, recurrence or metastases as early as possible.
B. Diagnosis of WHO II gliomas? malignant transformation as early as possibleMajority of (all?) WHO II gliomas will progress to high-grade tumours and it is oneof the crucial tasks of the contemporary radiology to identify the transformation asearly as possible. The most efficient modalities for predicting the transformation are: PWI and 1HMRS. Transformers reveal a significant increase of rCBV about a dozen months before transformation is visible clinically and in conventional diagnosticimaging (before contrast enhancement is apparent onT1-weighted images) (7). As reported in the paper by Law M et al. in 2006 “Lesions with relative CBV less than 1.75 had a median time to progression of 4620 days ? 433 (standard deviation), and lesions with relative CBV more than 1.75 had a median time to progression of 245 days ? 62” (8).
Detection and monitoring of early and late complications after treatment.
Supporting clinical trials of new therapeutic agents or in oncology drug development
The great number of currently available imaging modalities causes that this just the radiologist is to have decisive opinion in selection of the diagnostic method of choice for the given patient, diagnostic problem or clinical issue.
For many years, radiologic studies were structural imaging, presenting morphological features of normal tissues and pathologic processes, including neoplasms in these structures.
Diagnosing based on classical (morphological) modalities: radiography (x-ray imaging), CT, ultrasound and conventional MRI (T2-weighted, PD, T1-weighted without and with contrast enhancement) is based on detection of structural/anatomical abnormalities and/or abnormal contrast enhancement.
X-ray mammography remains the key and basic method of breast examination, both in screening and in women with clinically suspected breast cancer. The main development of x-ray mammography in the recent years concerns introduction to everyday clinical practice of full-field digital mammography. The main advantages of the digital mammography are: higher contrast resolution and improved dynamic range, elimination of many artefacts, possibility to apply CAD systems (computer aided detection) and PACS (Picture Archival and Communications System) systems. The latest x-ray mammography technique is tomosynthesis. Its result is presented as a series of high-resolution thin slices. Among the possible advantages (this is a new method and it is still intensively investigated) of application of breast tomosynthesis there are: improved cancer detection, reduced number of recalls and biopsies.
For a dozen or so years functional and molecular imaging defined by Hoffman JM and Gambhir SS (9) as “the in vivo characterization and measurement of biologic processes at the cellular and molecular level” have become a part of radiologic imaging examinations, especially MR. A combination of morphologic and functional examinations allows above all for a better answer on the basic question in cancer patient diagnosis: is the visible lesion a malignant tumour (primary, persistent, recurrence, metastasis), what is its size, is the applied therapy successful. Discussion of the main functional imaging modalities used nowadays (PET and SPECT) goes far beyond this paper.
All imaging modalities used in radiology and diagnostic imaging have developed significantly lately.
Computer tomography is currently the most frequently applied imaging modality in oncology.
MDCT (Multidetector Computer Tomography) combines very good spatial (< 1 mm) and time resolution. This offers a possibility to obtain reconstructions in any plane of the same quality as in the case of primary images, and to perform angiography/coronarography, dynamic examinations and CT perfusion. An important limitation of dynamic studies and CT perfusion concern the dose of ionising radiation.
Computer tomography added to the PET system (PET-CT) has markedly improved specificity of positron emission tomography.
From the oncologic point of view, the most important developments in ultrasound in recent years include introduction of contrast agents and elastography. The latter is used most frequently for differential diagnosis of breast lesions, it may be applied also in examinations of the liver. It is important that it has changed from a qualitative into a quantitative method.
But the greatest improvement has been observed in the recent years in magnetic resonance. MRI best combines – among the currently available methods – possibilities of morphological and functional imaging and – to some extent – it offers a possibilities of molecular imaging (9). New techniques of MRI have not only improved morphological imaging, combining excellent tissue resolution with very good spatial (< 1 mm) and time resolution, but they have also made functional studies possible. Especially intensively investigated MRI techniques concerned in oncologic diagnostics are: spectroscopy, diffusion imaging (DWI – diffusion weighted imaging), tumour vascularisation imaging (dynamic imaging and perfusion weighted imaging PWI), susceptibility-weighted imaging.
SNR (signal-to-noise ratio) grows with the increase of magnetic field in MR imaging , may be used to improve spatial resolution, reduce acquisition time, but there is no evidence that a 3T MR is more efficient in detecting or staging of a neoplasm as compared to a 1,5T (10, 11, 12).
Meanwhile, 7T MR are currently used in scientific research and not in clinical practice.
Functional imaging modalities go beyond morphological/anatomical assessment to quantitative and qualitative assessment of physiologic and biochemical processes in vivo. This is important because functional and metabolic changes of tissue/tumour, both on the molecular and functional level, are present much before structural lesions possible to be visualised in classical imaging. Besides, they allow for more precise characterisation of the pathologic process, including differentiation between benign and malignant lesions, which often look very much alike in structural imaging studies.
Functional imaging modalities in oncology can be divided into two types: those which assess physiology of the tumour and those which examine molecular processes in the tumour itself.(13)
1 HMRS (proton magnetic resonance spectroscopy)
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Dee KE, Sickles EA: Medical audit of diagnostic mammography examinations: comparison with screening outcomes obtained concurrently. AJRAm J Roentgenol 2001; 176 (3): 729-33.
2. Bartella L, Smith CS, Dershaw DD, Loberman L: Imaging Brest cancer Radiol Clin N Am 2007; 45: (45-67).
3. Stadlbauer A, Moser E, Gruber S et al.: Improved delineation of brain tumors: an automated method for segmentation based on pathologic changes of 1H-MRSI methablites in gliomas. Neuroimage 2004; 23 (2): 454-61.
4. Pirzkall A, McKnight TR, Grave EE et al.: MR spectroscopy guided target delineation for high-grade gliomas. Int J Radiat Oncol Biol Phys 2001; 50: 915-928.
5. Nimsky C, Grummich P, Sorensen AG et al.: Visualization of the pyramidal tract in glioma surgery by integrating diffusion tensor imaging in functional neuronavigation. Zentralbl Neurochir 2005; 66 (3): 133-41.
6. Kwee TC, Takahara T, Klomp DWJ, Luijten: Cancer imaging: novel concepts in clinical magnetic resonance imaging J Intern Med 2010; 268:120-132.
7. Danchaivijitr N, Waldman AD, Tozer DJ: Low-grade gliomas: Do changes in rCBV measurements atlongitudinal perfusion-weighted MR imaging predict malignant transformation. Radiology 2008; 247: 170-178.
8. Law M, Oh S, Babb JS et al.: Lowa-grade gliomas:dynamic susceptibility – weighted contrast-enhanced perfusion MR imaging-prediction of patient clinical response.Radiology 2006; 238 (2): 658-67.
9. Hoffman JM, Gambir SS Molecular imaging: The Visio and opportunity for radiology in the future. Radiology 2007; 44: 39-47.
10. Krautmacher C, Willinek WA, Tschampa HJ et al.: Brain tumors: full-and half-dose contrast enhanced MR imaging at 3T compared with 1,5T – initial experience. Radiology 2005; 237: 1014-9.
11. Kuhl CK, JostP, Morakkabati N et al.: Contrast enhanced MR imaging of the breast at 3,0 and 1,5T in the same patients: initial experience. Radiology 2006; 239: 666-76.
12. Van Falkenhausen MM, Lutterbey G, Morakkabati-Spitz N et al.: High-field strength MR imaging of the liver at 3.0T: intraindividualcomparative study with MR imaging 1,5T. Radiology 2006; 241: 156-66.
13. Torigian DA, Huang SS, Houseni M, Alami A: Functional imaging of cancer with emphasis on molecular techniques. CA Cancer J Clin 2007; 57: 206-24.
14. Swindle P, McCredie S, Russel P et al.: Pathologic characterization of human prostate tissue with proton MR spectroscopy. Radiology 2003; 228: 144-151.
15. Zakian KL, Sircar K, Hricak H et al.: Correlation of proton MR spectroscopic imaging with gleason score based on step-section pathologic analysis after radical prostatectomy. Radiology 2005; 234 (3): 804-14.
16. Padhani AR: Dynamic contrast-enhancement MR imaging. Cancer Imaging 2000; 1: 52-63.
17. Hylton N: Dynamic contrast-enhanced magnetic resonance imaging as an imaging biomarker. J Clin Oncol 2006; 24: 3293-8.
18. Aksoy FG, Lev MH: Dynamic contrast – enhanced brain perfusion imaging: techniques and clinical applications. SeminUltrasound CT MR 2000; 21: 462-477.
19. Miles KA: Tumor angiogenesis and its relation to contrast enhancement on computed tomography: a review. Rur J Radiol 1999; 30: 198-205.
20. Padhani AR, Leach MO: Antivascular cancer treatments: functional assessments by dynamic contrast enhanced magnetic resonance imaging. Abdominal Imaging 2005; 30: 324-341.
21. Daldrup HE, Shames DM, Husseini W et al.: Quantification of the extraction fraction for gadopentate across breast cancer capillaries. Magn Reson Med 1998; 40: 537-543.
22. Buckley DL, Roberts C, Parker GJ et al.: Prostate cancer: evaluation of vascular characteristics with dynamic contras-enhanced T1-weighted imaging – initial experience. Radiology 2004; 233: 709-15.
23. Stejskal EO, Tanner JE: Spin diffusion measurements : spin-echo In the presence of a time dependent field gradient. J Chem Phys 1965; 42: 288-292.
24. Provenzale JM, Mukundan S, Barboriak DP: Diffusion-weighted and perfusion MR imaging for brain tumor charakterization and assessment of treatment response. Radiology 2006; 239: 632-49.
25. Padhani AR, Liu G, Koh DM et al.: Diffusion-weighted magnetic resonance imaging as a cancer biomarker :consensus and recommendations. Neoplasia 2009; 11: 102-25.
26. Koh DM, Collins DJ: Diffusion weighted MRI in the body: applications and challenges in oncology. AJR Am J Roentgenol 2007; 188 (6): 1622-35.
27. Fuerlein S, Pauls S, Juchems MS et al.: Pitfalls in abdominal diffusion-weighted imaging : how predictive is restricted water diffusion for malignancy. AJR Am J Roentgenol 2009; 193: 1070-6.
28. Taouli B, Koh DM: Diffusion-weighted MR imaging of the liver Radiology 2010; 254 (1): 47-67.
29. Qi LP, Zhang XP, Tang L et al.: Using diffusion-weighted MR imaging for tumor detection in the collapsed lung: a preliminary study. Eur Radiol 2009; 19 (2): 333-41.
30. Mori T, Nomori H, Ikeda K et al.: Diffusion-weighted magnetic resonance imaging for diagnosing malignant pulmonary nodules/masses: comparison woth positron emission tomography. J Thorac Oncol 2008; 3: 358-64.
31. Ohba Y, Nomori H, Mori T et al.: Is diffusion-weighted magnetic resonance imaging superior to positron emission tomography with fludeoxyglucose F18 in imaging non-small cell lung cancer? J Thorac Cardiovasc Surg 2009; 138: 439-45.
32. Sato Ch, Naganawa S, Nakamara T et al.: Differentiation of noncancerous tissue and cancer lesions by apparent diffusion coefficient values in transition and peripheral zone of the prostate. J Mag Reson Imaging 2005; 21: 258-262.
33. Razek AAA, Kandeel AY, Soliman N et al.: Role of diffusion-weighted echo planar MR imaging in differentiation of residual or recurrent head and neck tumors and posttreatment changes. AJNR 2007; 28: 1146-1152.
34. Razek AAA, Soliman NY, Elkhamary A et al.: Role of diffusion-weighted MR imaging in cervical lymphadenopathy. Eur Radiol 2006; 16 (7): 1468-77.
35. Vandecaveye V, De Keyzer F, Vander Poorten V et al.: Head and neck squamous cell carcinoma: value of diffusion-weighted MR imaging for nodal staging Radiology 2009; 251 (1): 134-46.
36. Hasegawa I, Boiselle PM, Kubawara K et al.: Mediastinal lymph nodes in patients non-small cell lung cancer: preliminary experience with diffusion weighted MR imaging. J Thorac Imaging 2008; 23: 157-161.
37. Takahara T, Imai Y, Yamashita T et al.: Diffusion weighted whole body imaging with background body signal suppression (DWIBS): technical improvement using free breathing, STIR and high resolution 3D display. Radiat Med 2004; 22: 275-82.
38. Takenaka D, Ohno Y, Matsumoto K et al.: Detection of bone metastases In non-small cell lung cancer patients: comparison of whole body diffusion-weighted imaging (DWI) whole body MR imaging without and with DWI, whole body FDG-PET/CT and bone scintygraphy. JMagn Reson Imaging 2009; 30: 298-308.
39. Nomori H, Mori T, Ikeda K et al.: Diffusion-weighted magnetic resonance imaging can be use in place of positron emission tomography for N staging of non-small cell lung cancer with fewer false-positive results. J Thorac Cardiovasc Surg 2008; 135: 816-22.
40. Ohno Y, Koyama H, Onishi Y et al.: Non-small cell lung cancer: whole-body MR examination for M-stage assessment – utility for whole-body diffusion-weighted imaging compared with integratedFDG PET/CT. Radiology 2008; 248: 643-54.
41. Steinborn M M, Heuck AF, Tiling R et al.: Whole body bone marrow MRI in patients with metastatic disease to the skeletal system. J Comput Assist Tomogr 1999; 23: 123-129.
42. Daldrup-Link HE, Franzius C, Link TM et al.: Whole-body MR imaging for detection of bone metastases in children and young adults: comparison with skeletal scintigraphy and FDG PET. AJR Am J Roentgenol 2001; 177: 229-236.
43. Antoch G, Saoudi N, Kuehl H et al.: Accuracy of whole-body dual-modality fluorine-18-2-fluoro-2-deoxy-D-glucose positron emission tomography and computed tomography (FDG-PET/CT) for tumor staging in solid tumors: comparison with CT and PET. J Clin Oncol 2004; 22: 4357-4368.
44. Schmidt GP, Schoenberg SO, Schmid R et al.: Screening for bone metastases: whole-body MRI using a 32-channel system versus dual-modality PET-CT. Eur Radiol 2007; 17 (4): 939-949.
45. Vandecaveye V, de Keyzer F, Vander Poorten V et al.: Evaluation of the larynx for tumour recurrence by diffusion-weighted MRI after radiotherapy: initial experience in four cases. Br J Radiol 2006; 79: 681-687.
46. Padhani AR: MRI for assessing antivascular cancer treatments. British Journal of Radiology 2003; 76: S60-S80.
47. Moffat BA, Hall DE, Stojanowska J et al.: Diffusion imaging for evaluation of tumor therapies in preclinical animal models. MAGMA 2004; 17: 249-259.
48. Chen CY, Li CW, Kuo YT et al.: Early response of hepatocellular carcinoma to transcatheter arterial chemoembolization: choline levels and MR diffusion constatnts – initial experience. Radiology 2006; 239: 448-456.
49. Hamstra DA, Galban CJ, Meyer CR et al.: Functional diffusion map as an early imaging biomarker for high-grade glioma: correlation with conventional radiologic response and overall survival. JClin Oncol 2008; 26: 3387-95.
50. Hamstra DA, Rehemtulla A, Ross BD: Diffusion magnetic resonance imaging: a biomarker for treatment response in oncology. J Clin Oncol 2007; 25: 4104-9.
51. Meisamy S, Bolan PJ, Baker EH et al.: Neoadjuvant chemotherapy of locally advanced breast cancer: predicting response with in vivo (1) HMR spectroscopy – a pilot study at 4T. Radiology 2004; 233: 424-31.
52. Dzik-Jurasz A, Domening C, George M et al.: Diffusion MR for prediction of response of rectal cancer to chemoradiation. Lancet 2002; 360: 307-308.
53. De Vries AF, Kresmer C, Hein AP et al.: Tumor microcirculation and diffusion predict therapy outcome for primary rectal carcinoma Int J Radiat Oncol Biol Phys 2003; 56: 958-965.
54. Mardor Y, Roth Y, Ochershvilli A et al.: Pretreatment prediction of brain tumor?s response to radiation therapy using high b-value diffusion weighted MRI. Neoplasia 2004; 6: 136-142.
55. Koh DM, Scurr E, Collins DJ et al.: Colorectalhepatic metastases: quantitative measurements using single-shot echo-planar diffusion-weighted MR imaging Eur Radiol 2006; 16: 1898-1905.
56. George ML, Dzik-Jurasz AS, Padhani AR et al.: Non-invasive methods of assessing angiogenesis and their value in predicting response to treatment in colorectal cancer. Br J Surg 2001; 88: 1628-1636.


