© Borgis - Postępy Nauk Medycznych 7/2013, s. 506-510
*Teresa Jackowska1, 2, Małgorzata Nieścior2, Monika Grzelczyk1, 2
Zatrucie tlenkiem węgla u dzieci – własne obserwacje
Carbon monoxide poisoning in children – own observations**
1Department of Paediatrics, Medical Centre for Postgraduate Education, Warszawa
Head of Department: prof. Teresa Jackowska, MD, PhD
2Department of Pediatrics, Bielanski Hospital, Warszawa
Head of Department: prof. Teresa Jackowska, MD, PhD
Streszczenie
Wstęp. Tlenek węgla (CO) jest bezbarwnym gazem bez zapachu wytwarzanym z niepełnego spalania związków węglowych. Wdychanie CO jest najczęstszą przyczyną zatrucia w krajach uprzemysłowionych.
Cel. Analiza danych epidemiologicznych i klinicznych dzieci hospitalizowanych z powodu zatrucia tlenkiem węgla.
Materiał i metody. Poddano analizie dokumentację medyczną 36 dzieci w wieku od 8 miesięcy do 18 lat (średnio 7 lat 8 mies.), hospitalizowanych w Klinice Pediatrii w Warszawie od stycznia 2003 do października 2012 roku z powodu ostrego zatrucia CO.
Wyniki. Spośród 36 dzieci u 22 (61%) przyczyną zatrucia CO były uszkodzone grzejniki gazowe, u 7 (19,5%) pożar, u 6 (16,7%) uszkodzona wentylacja, a w jednym (2,8%) przypadku nie udało się określić przyczyny. U 12 (33,3%) dzieci nie stwierdzono żadnych objawów, zaś u niektórych występowało kilka. Najczęściej, bo aż w połowie przypadków, była to senność i ospałość, następnie ból głowy, omdlenia, wymioty i nudności. Poziom hemoglobiny tlenkowęglowej (COHb) u dzieci wynosił od 2,0 do 33,1% (średnio 13,3%). U 9 (25%) dzieci konieczne było zastosowanie leczenia w komorze hiperbarycznej (HBO). W tej grupie poziom COHb był od 11,0 do 33,1% (średni 25,3%). Odchylenia w badaniach laboratoryjnych występowały u trójki dzieci z poziomem COHb 11%, 20% i 30,6%. U wszystkich było to podwyższenie CK-MB odpowiednio 2,38, 11,94 i 2,8 ng/ml.
Wnioski. Do zatruć tlenkiem węgla dochodzi w okresie jesienno-zimowym. Badaniem potwierdzającym zatrucie jest oznaczenie karboksyhemoglobiny. Zakres poziomów COHb był dość szeroki i nie wykazywał związku z objawami. W leczeniu najistotniejsze jest jak najszybsze zapewnienie poszkodowanemu odpowiedniego leczenia, pod postacią tlenoterapii biernej lub w komorze hiperbarycznej.
Summary
Introduction. Carbon monoxide (CO) is a colorless, odorless gas produced at an incomplete combustion of carbonaceous compounds. CO inhalation is the most common cause of poisoning in the industrialized world.
Aim. Analysis of epidemiological and clinical data of children hospitalized due to carbon monoxide poisoning.
Material and methods. The medical records of 36 consecutive children aged between 8 months and 18 years (average 7 years 8 months) admitted to the Department of Pediatrics in Warsaw from January 2003 through October 2012 with CO poisoning were reviewed.
Results. Of these 36 cases, 22 (61%) were caused by a faulty gas heater, 7 (19.5%) by a fire, 6 (16.7%) by faulty ventilation, and in one case (2.8%) it was not possible to determine the cause. In 12 (33.3%) children no symptoms were observed, while in certain children a number of symptoms was present. Most frequently, in as many as half of the cases, the symptoms were drowsiness/lethargy, followed by headache, syncope, vomiting and nausea. The carboksyhemoglobin (COHb) levels in all children ranged from 2.0 to 33.1% (average 13.3%). In 9 (25%) children it was necessary to administer HBO treatment. In this group, the COHb levels ranged from 11.0 to 33.1% (average 25.3%). Abnormal results of laboratory tests occurred in 3 children with COHb levels of 11%, 20% and 30.6%. In all of them it was an elevation of CK-MB, to the level of 2.38, 11.94 and 2.8 ng/ml respectively.
Conclusions. Carbon monoxide poisonings are most common in the autumn-winter period. This type of poisoning can be confirmed by determining the carboksyhemoglobin level. Overall, the range of the COHb levels was relatively broad and showed no relationship with the symptoms. The most important intervention is to provide the victim adequate treatment, in the form normobaric oxygen or hyperbaric therapy.

Introduction
Carbon monoxide (CO) is a colorless, odorless gas produced at an incomplete combustion of carbonaceous compounds. CO inhalation is the most common cause of poisoning in the industrialized world. Severe CO poisoning may cause multi-organ dysfunction, frequently necessitating an admission to intensive care units. CO poisoning is frequently unrecognized, because the signs and symptoms are relatively nonspecific (1). In the United States National Poison Data System (NPDS), the reported mortality rates caused by this type of poisoning range between 1% and 31% of all deaths. Most CO exposures occurred at home and most often involved females, children aged ≤ 17 and adults aged 18-44. Clinical symptoms were reported for 68.1% of the total exposures, with headache, nausea and dizziness observed most commonly (2).
The administration of supplemental O2 is the cornerstone in the treatment of CO poisoning. Oxygen inhalation will accelerate the dissociation of CO from hemoglobin, as well as provide enhanced tissue oxygenation. Hyperbaric oxygen therapy (HBOT) is a treatment modality in which a person breathes 100% O2 while exposed to an increased atmospheric pressure (3).
In our study, we have reviewed the symptoms and the results of hyperbaric oxygen therapy (HBOT) in the treatment of 36 children with CO poisoning.
Aim
Analysis of epidemiological and clinical data of children hospitalized due to carbon monoxide poisoning.
Material and methods
The medical records of 36 consecutive children, 12 (33.3%) girls and 24 (66.7%) boys, aged between 8 months and 18 years (average 7 years 8 months), admitted to the Department of Pediatrics in Warsaw from January 2003 through October 2012 due to CO poisoning, were reviewed. Every year, one (2004, 2008, 2010) to eight children (2007, 2012) were hospitalized. Most of the children, as many as 29 (80.5%), were hospitalized during the autumn-winter months (from October to March) (fig. 1). No patients were excluded from the study. Nine of them (25%) were referred to the hyperbaric unit for treatment.
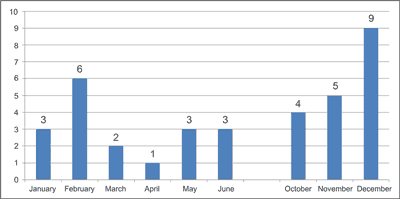
Fig. 1. Hospitalizations due to CO poisoning depending on the date of admittance.
The COHb levels were measured at the local laboratory from venous blood.
Results
Of these 36 cases, 22 (61%) were caused by a faulty gas heater, 7 (19.5%) by a fire, 6 (16.7%) by faulty ventilation, and in one case (2.8%) it was not possible to determine the cause. Table 1 presents the symptoms of the children exposed to carbon monoxide. In 12 (33.3%) children no symptoms were observed, while in certain children a number of symptoms occurred. In most cases, that is in as many as half of the cases, the symptoms were drowsiness/lethargy, followed by headache, syncope, vomiting and nausea.
Table 1. Symptoms in the children exposed to carbon monoxide.
Symptoms (n = 59)
Children (n = 36) | No. | (%) |
| Observed symptoms of poisoning | 24 | 66.7% |
| Drowsiness/lethargy | 12 | 50.0 |
| Headache | 10 | 41.7 |
| Syncope | 8 | 33.3 |
| Dizziness/vertigo | 7 | 29.2 |
| Vomiting | 7 | 29.2 |
| Nausea | 3 | 16.7 |
| Abdominal pain | 2 | 8.3 |
| Redness of the skin | 2 | 8.3 |
| Balance disorders | 2 | 8.3 |
| Confusion | 1 | 4.2 |
| Chest pain | 1 | 4.2 |
| Visual disturbances | 1 | 4.2 |
| Other | 3 | 12.5 |
| Without symptoms of poisoning | 12 | 33.3% |
In five (13.9%) children an elevated level of white blood cells was observed, in four cases occurred an elevated level of cardiac creatine kinase (CK-MB, 2.38-11.94 ng/ml), in one case an increase of troponin was observed, and in one case an elevated level of glucose occurred. In one child the electrocardiogram (ECG) recorded a increase of the ST in the V2-V3 leads. In 27 (75%) cases no abnormalities were reported in laboratory tests and in ECG.
The carboksyhemoglobin (COHb) levels in all children in the study were from 2.0 to 33.1% (average 13.3%). In 9 (25%) children it was necessary to administer/introduce HBO treatment. All the children referred for HBO were treated in the chamber. In this group, the COHb levels ranged from 11.0 to 33.1% (average 25.3%). Two children received one HBOT treatment, two children – two, and five children – three treatments. In 24 children (66.7%) the exclusive treatment was passive oxygen therapy. In this group, the COHb levels ranged from 2.0 to 22% (average 16.5%). In three children whose COHb levels were 3% no treatment was administered.
In three (8.3%) children with COHb levels above 30% (moderate-severe poisoning) the dominant symptom was syncope. In one of them, also vomiting and major convulsions occurred. After the return of consciousness, all children suffered from headaches, and in individual cases the above symptoms were accompanied by confusion, drowsiness and weakness. All of these patients were administered hyperbaric chamber treatment (two to three times), which was followed by passive oxygen therapy.
In 15 (41.7%) children, the COHb levels did not rise above 10%, and the symptoms of poisoning were mild. The most frequently reported symptoms were vomiting, abdominal pain, headache and excessive drowsiness. However, considering the fact that most of the poisoning occurred at night, the drowsiness was misinterpreted by the adults. Also vomiting and abdominal pain suggested an acute gastroenteritis to the family. An example here may be the CO poisoning of two sisters who were moved to another room after their mother was observed to have lost her consciousness. Only after a certain time it was noticed that they did not wake up despite the chaos and noise in the room. The COHb levels in these girls were 9.4% and 8.4% respectively. They were administered passive oxygen therapy treatment.
In the children who were reported to show no symptoms of carbon monoxide poisoning the suspicion of this condition was made based on the clinical history. Six out of the seven children were victims of a fire, and the remaining one was a witness of his father’s carbon monoxide intoxication.
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Prockop LD, Chichkova RI: Carbon monoxide intoxication: An updated review. J Neurol Sci 2007; 262: 122-130.
2. Centers for Disease Control and Prevention (CDC). Carbon monoxide exposures – United States, 2000-2009. Morb Mortal Wkly Rep 2011 (MMWR); 60(30): 1014-1017.
3. Stephen R: Thom, Carbon Monoxide Pathophysiology and Treatment; Physiology and Medicine of Hyperbaric Oxygen Therapy. Elsevier Inc., Philadelphia 2008: 321-347.
4. Heckerling PS, Leikin JB, Maturen A et al.: Predictors of occult carbon monoxide poisoning in patients with headache and dizziness. Ann Intern Med 1987; 107: 174-176.
5. Heckerling PS, Leikin JB, Maturen A: Occult carbon monoxide poisoning: Validation of a prediction model. Am J Med 1988; 84: 251-256.
6. Rucker J, Fisher JA: Carbon Monoxide Poisoning. Clinical Critical Care Medicine. Elsevier Inc., Philadelphia 2006; 63: 679-683.
7. Guzman JA: Carbon Monoxide Poisoning. Crit Care Clin 2012; 28: 537-548.
8. Buckley NA, Juurlink DN, Isbister G et al.: Hyperbaric oxygen for carbon monoxide poisoning. Cochrane Database Syst Rev 2011; 4: CD002041.
9. Baum CR: What’s New in Pediatric Carbon Monoxide Poisoning? Clin Ped Emerg Med 2008; 9: 43-46.

