© Borgis - New Medicine 1/2003, s. 44-47
Paweł Bernat, Radosław Pietura, Małgorzata Szczerbo-Trojanowska
Application of T1- and T2-weighted magnetic resonance imaging (MRI) in assessment of uterine leiomyoma morphology after uterine artery embolisation
Department of Interventional Radiology and Neuroradiology Medical University of Lublin
Summary
Objective: To evaluate the application of T1- and T2-weighted magnetic resonance imaging (MRI) in assessment of uterine leiomyoma morphology after uterine artery embolisation.
Methods: Between November 2001 and January 2003 MR examination was performed on 110 patients. T1-weighted and T2-weighted spin-echo MR images were made through the pelvis in sagittal, tranverse, coronal planes before the procedure, and 3 months after. Fifty-eight % of leiomyomas gave an iso- or hyperintensive signal in T2-weighted imaging. The mean uterine volume was 496 cm3 and the mean volume of the dominant fibroid was 198 cm3. Leiomyoma was submucosal in 63% of patients, transmural in 13%, and subserosal in 24%.
Results: After 3 months, MRI showed the mean volume reduction of the dominant fibroid to be 57%, ranging from 36% to 89%, and of the uterus to be 41%, ranging from 30% to 73%. In T1-weighted MR images submucosal-expelling fibroids showed hyperintensive signal, and only long-term changes in morphology could have been observed. T2-weighted MR images were used in evaluation of volume reduction and the detailed morphology of the fibroids.
Conclusions: A T2-weighted MR sequence is an effective method for the assessment of the morphology of uterine leiomyoma, the T1-weighted sequence being used in imaging of submucosal – expelling fibroids.
INTRODUCTION
Uterine artery embolisation is a less invasive treatment for symptomatic uterine leiomyoma, and is increasingly popular. Indications for the therapy include abnormal uterine bleeding (menorrhagia and metrorhagia), and bulk-related syndromes (pain, increased urinary frequency, nocturia, constipation, patient discomfort) caused by one or more fibroids (6). Since 1995, when Ravine first described uterine artery embolisation, plenty of subsequent published studies have shown embolisation to be effective in the treatment of heavy menstrual bleeding (90%, 92%) and of bulk symptoms (93%, 92%) – data are based on analyses of 200-400 patients treated with uterine artery embolisation and observed over a minimum of 12 months (8, 11, 17, 18).
In Poland, uterine artery embolisation was performed for the first time in November 2001 at the Medical University of Lublin (16). A decrease in, or lack of, symptoms as a result is long-lasting (21). The most dangerous side-effect of embolisation is uterine abscess, which indicates definitive hysterectomy (15). Its frequency has been estimated as 1 per 100 and as 1 per 700 treatments (18, 20). Sometimes, from one to four months after treatment, parts of submucosal leiomyoma may be expelled into the uterine cavity and progress to the vagina. This condition is associated with pain and a slight temperature. No case has been published describing an enlarged leiomyoma or the appearance of a new leiomyoma after embolisation. The only method used for qualification for the procedure is magnetic resonance imaging (MRI). MR imaging is much more sensitive and specific than transvaginal ultrasound examination in diagnosing adenomyosis, which may occur at the same time in as many as 20% of patients with leiomyoma (2). MR examination also allows the exclusion of other disorders, such as sarcoma or tumours of ovary. The aim of this paper is to evaluate the application of T1- and T2-weighted magnetic resonance imaging (MRI) in the assessment of the morphology of uterine leiomyoma after uterine artery embolisation.
MATERIALS AND METHODS
Between November 2001 and January 2003, MR 1,5T examination was carried out on 180 patients, but complete data is only available for 110 patients aged of 32-51. Pre-procedure imaging studies were made to confirm the diagnosis of leiomyoma, determine the size and position, and to identify adenomyosis or other concurrent diseases. T1-weighted and T2-weighted spin-echo MR images were obtained through the pelvis in sagittal, tranverse and coronal planes before the procedure, and 3 months afterwards. The volumes of the dominant leiomyoma and uterus, both before and after treatment, were calculated using the formula for a prolate ellipse (L×W×D×0.523) – W, L, D being the avarages of the dimensions of the leiomyoma in three different planes. Main indication for the procedure was abnormal uterine bleeding. MR imaging clearly defines the position of the leiomyoma relative to the endometrium. An endometrial biopsy, a recent vaginal culture, and a pregnancy test (βHCG) were also prudent for all patients. Pre-procedure evaluation consists of a consultation with the gynaecologist and the radiologist, with the case history and a physical examination. Blood tests include blood count, CRP, blood urea, nitrogen/creatinine, pro-thrombin time, and partial thromboplastin time. Histological examinations were performed in order to exclude a disordered proliferative endometrium or a malignant endometrial tumour as the couse of abnormal bleeding. After institutional review board approval, we began our study in consecutive patients undergoing uterine artery embolisation for leiomyoma, informed written consent having been obtained in each case. Uterine artery embolisation procedures were performed in the Department of Interventional Radiology under fluoroscopy control and under local anaesthesia (1% lignocaine). During the procedure patients were given 0-6 mg midazolam for sedation. Vascular access was effected via the left axillary artery. A4 Fr (Ř 1.35 mm) sheath and then a 4 Fr pigtail catheter were introduced with the tip sited in the distal aorta below the renal arteries. Flush aortography allowed definition of the pelvic arterial anatomy. Using a selective catheter (4Fr Vertebral) and angled hydrophilic wire, both uterine arteries were cannulated. The catheter tip was positioned at or beyond the junction of the descending and the horizontal portions of the uterine artery. Polyvinyl alcohol particles, at a size of 350-500 and 500-710 microns, were deployed until cessation of antegrade flow occurred. Final angiography showed the occlusion of both uterine arteries and their branches. After the procedure the patient returned to the gynaecology department for further observation. Post-procedure pain was managed using morphine, administered by a patient-controlled analgesia (PCA) pump. Patients normally left the hospital after 24 hours.
RESULTS
Uterine artery embolisation was technically successful in all cases. The duration of the procedure was from 45 to 120 minutes (mean 61 minutes). The number of leiomyomas among patients examined with MR imaging ranged from 1 to 23 (mean 2). Before treatment the mean volume of the uterus was 496 cm3, and the mean volume of the dominant fibroid was 198cm3. Leiomyoma was submucosal in 63% of patients, transmural in 13% of patients, and subserosal in 24% of patients. At 3 months follow-up T2-weighted MR images were used in evaluation of volume reduction and the detailed morphology of fibroids. After therapy, the mean volume of the dominant fibroid decreased to 85 cm3, the percentage reduction being a mean of 57%, ranging from 36% to 89%. The uterus decreased in volume to 292 cm3, a mean percentage reduction of 41%, on a range of from 30% to 73%. In T1- and T2-weighted MR imaging in 94 patients, leiomyomas showed hypointensive signal. In 12 patients, with symptoms of fibroids expelling into the uterine cavity, the signals were hyperintensive both in T1- and T2-weighted images. In 4 patients leiomyomas were hypointensive in T1-weighted imaging and hyperintensive in T2-weighted imaging at the same time. This group of fibroids showed a hyperintensive signal in T2-weighted imaging before embolisation, representing the cellular histologic subtype of leiomyomas. No hysterectomy as a result of infection had to be performed in any of 110 patients treated.
DISCUSSION
The imaging studies at 3 months after embolisation revealed a progressive reduction in uterine volume (mean 41% ) and, to a greater extent, in the dominant leiomyoma volume ( mean 57% ). These data are similar to those described by other authors. T1- and T2- -weighted MR images are used in detailed assessment of morphology and volume reduction after therapy. Most leiomyomas we treated, as an outcome of embolisation, showed a hypointensive signal both in T1 and T2 relaxation times as a result of hyalinisation and fibrosis. In 4 patients in T2-weighted MR imaging, the fibroids were hyperintensive before and after therapy, which was caused by a paricular histologic subtype of leiomyoma, the cellular. In 12 patients showing pain and a slight temperature, MR examination allowed diagnosis of expelling fibroids, and excluded the possibility of a uterine abscess. In this group of patients, in T1- and T2-weighted images, hyperintensive signals were observed. MRI, due to its high effectiveness and sensitivity, shows the number, size, precise location and extent of leiomyomas, and recently has become more important in obstetrics and pelvic radiological examination (10). MRI is also accurate in distinguishing adenomyosis from leiomyoma (18). We used MRI as a primary method both before and after embolisation in assessing fibroids. We observed no cases of leiomyosarcoma among our group of patients. There is a case described, when a patient was given uterine artery embolisation after excluding leiomyosarcoma, as a result of an incorrect USG diagnosis (5). Additional analysis is needed to determine whether the imaging characteristics at the outset or during follow-up most accurately predict the outcome (18). On the basis of the results of our study, we found no patients with fibroids for whom MRI was certainly contraindicated. To date, no studies have compared the outcome of uterine artery embolisation with other leiomyoma therapies.
CONCLUSIONS
Magnetic resonance imaging is the method of choice in assessing leiomyoma before uterine artery embolisation. A T1-weighted sequence is used in imaging the course of submucosal – expelling fibroids into the uterine cavity. T2-weighted MR images are used in evaluation of the volume reduction and detailed morphology of fibroids, as well as for distinguishing between haemorrhagic and cellular leiomyoma, and to exclude leiomyosarcoma. MRI is sensitive and specific for the assessment of leiomyoma morphology after uterine artery embolisation.
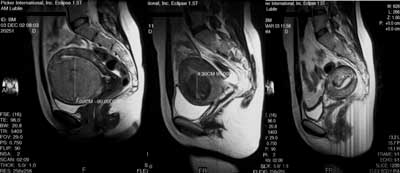
Fig. 1. Uterine artery embolisation. Sagittal T2-weighted images demonstrate the same patient before (left), 3 months after (middle) and four months after (right) procedure.
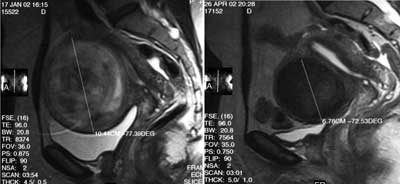
Fig. 2. Uterine artery embolisation. Sagittal T2-weighted images, the same leiomyoma-decreased coronary diameter and signal intensity after procedure.
Piśmiennictwo
1. Andrews R.T. et al.: Successful embolisation of collateral from the ovarian artery during uterine artery embolization for fibroids: a case report. J. Vasc. Interv. Radiol. 2000; 11:607-610. 2. Ascher S.M. et al.: Adenomyosis: prospective comparison of MR imaging and transvaginal sonography. Radiology 1994; 190:803-806. 3. Berkowitz R. et al.: Vaginal expulsion of submucosal fibroids after uterine artery embolization: a report of three cases. J. Reproduct. Med. 1999; 44:373-376. 4. Broder M.S. et al.: The Appropriateness of Recommendations for Hysterectomy. Obstet Gynecol. 2000; 95:199-205. 5. Common A.A. et al.: Therapeutic failure of uterine fibroid embolisation caused by underlying leiomyosarcoma. J. Vasc. Interv. Radiol. 2001; 12:1449-1452. 6. Goodwin S.C. et al.: Uterine artery embolization for the treatment of uterine leiomyomata, midterm results. J.Vasc. Interv. Radiol. 1999; 10:1159-1165. 7. Goto A. et al.: Usefulness of Gd-DTPA contrast-enhanced dynamic MRI and serum determination of LDH and its izosymes in the differential diagnosis of leiomyosarcoma from degenerated leiomyoma of the uterus. Intern. J. Gynecol. Cancer 2002; 12(4):354-358. 8. Hutchins F. et al.: Selective uterine artery embolization as primary treatment for symptomatic leiomyomata uteri. J. Am. Assoc. Gynecol. Laparosc. 1999; 6:279-284. 9. Leibsohn S. et al.: Leiomyosarcoma in a series of hysterectomies performed for presumed uterine leiomyomatas. Am. J. Obstet. Gynecol. 1990; 162:968-976. 10. Levine D. et al.: Obstetrics MR imaging Radiology 1999; 211:609-617. 11. McLucas B. et al.: Uterine Fibroid Embolization: Nonsurgical treatment for symptomatic fibroids. J. Am. Coll. Surg. 2001; 192:95-105. 12. Omary R.A. et al.: The effect of pelvic MR imaging on the diagnosis and treatment of women with presumed symptomatic uterine fibroids. J. Vasc. Interv. Radiol. 2002; 13:1149-1153. 13. Outwater E.K. et al.: Adenomyosis: current concepts and imaging considerations. AJR 1998; 214:437-441. 14. Pelage J. et al.: Fibroid-related menorrhagia: treatment with superselective embolization of the uterine arteries and midterm follow-up. Radiology 2000; 215:428-431. 15. Pietura R. et al.: Zabieg embolizacji tętnic macicznych jako alternatywna metoda leczenia mięśniaków macicy. Ginekologia Polska 2003; 1,79-84. 16. Pietura R. i wsp.: Opis przypadku chorej z objawowym mięśniakiem macicy leczonej metodą embolizacji tętnic macicznych. Ginekol. Pol. 2003; 1:69-72. 17.Ravina J.H. et al.: Arterial embolisation to treat uterine myomata. Lancet 1995; 364:71-72. 18. Spies J. et al.: Uterine Artery Embolization for Leiomyomata. Obstet Gynec. 2001; 98:29-34. 19. Spies J. et al.: Complications after uterine artery embolization for Leiomyomas. Obstet Gynec. 2002; 100:873-880. 20. Walker W.J., Pelage P.: Uterine artery embolization for symptomatic fibroids: clinical result in 400 women with imaging follow-up. Brit. J. Obstet. Gynaecol. 2002; 109:1262-1272. 21.Worthington-Kirsch R.L. et al.: Uterine arterial embolization for the management of leiomyomas: quality of life assessment and clinical response. Radiology 1998; 208:625-629. 22. Zawin M. et al.: Does pelvic magnetic resonance imaging differentiate among the histologic subtypes of uterine leiomyomata? Fertility and Sterility 1998; Vol 70, NO.3.
Pozostałe artykuły z numeru 1/2003:

