© Borgis - New Medicine 4/2013, s. 111-113
*Lidia Zawadzka-Głos, Iwona Krajewska
Treatment of subglottic stenosis in children
Department of Pediatric Otolaryngology, Medical University of Warsaw, Poland
Head of Department: Lidia Zawadzka-Głos, MD, PhD
Summary
Subglottic stenosis of the larynx are currently the most common indication for tracheostomy in children. Treatment of postintubation laryngeal stenosis has been modificated for many years. Methods of treatment of postintubation stenosis of the larynx can be divided into two groups: endoscopic and open surgical methods. Below presented review of these methods of treatment are based on the literature and our own experience.
INTRODUCTION
In 1930, Chevalier Jackson noted that children with chronic stenosis of the larynx did not grow out of their problem while ageing but require treatment. Acquired stenosis of the larynx was the result of trauma caused by the surroundings or infectious diseases such as syphilis, tuberculosis, and typhoid fever (1). These children were treated by tracheotomy. Often too high tracheostomy itself provoked the process of fibrosis and drove to secondary stenosis of the larynx at the cricoid cartilage. During this period, there was no method of reconstruction of the respiratory tract, and many children were treated by expansion, with virtually no major effects, leaving them dependent on a tracheotomy for life.
Acquired subglottic stenosis of the larynx has started to be well recognized since 1965, when McDonald and Stocks initiated prolonged intubation as a treatment for infants requiring respirator (2). The increase in the occurrence of postintubation stenosis of the larynx and the high risk of mortality of children with tracheostomy of as much as 24% resulting in the development of new methods of treatment and reconstruction of the respiratory tract (3, 4).
METHODS FOR OPEN SURGERY
In 1971, the first cut was described by Grahn of Rethie procedure of cricoid cartilage (5). This procedure was based on the intersection of the vertical rear wall of cricoid cartilage and scar excision and stent type of Aboulker. In 1974 Evans and Todd developed the first laryngotracheoplastic by using the characteristic stepped front notches of cricoid cartilage and upper tracheal rings that were left open, and stented it by stent made from silastic for about 6 weeks (6).
In 1980, Cotton and Seid proposed a method of anterior cricoid cartilage called cricoid split (CS) (7). The method has been used for infants with stenosis of the front of the glottis and stenosis of the subglottic region. The concept involved decompression of cricoid cartilage in exchange for a tracheotomy. According to data from 1987 Holinger successfully intubated off 77% of patients in the 138 children without performing a tracheotomy (8).
Severe laryngotracheal stenosisstarted to heal with the use of cartilage grafts. Fearon and Cotton in 1972 described a procedure for the use of cartilage graft to expand the light of the larynx in children (4, 9). The implant used part of the thyroid cartilage. In subsequent studies they used the cartilage of the nasal septum, the cartilage of the ear, the cartilage of the ribs, but also part of the hyoid bone or piece of muscle sterno-clavicular-mastoideus. They also tried to enter the conserved cartilage, but due to the very high risk of resorption of the cartilage, they discontinued its use (10). The largest use of autogenic cartilage has been found in the patient’s ribs. The implants were introduced in the front or rear part of the cricoid cartilage, or in both parts simultaneously. This type of treatment is called laryngo-tracheal reconstruction (LTR). In 1989 Cotton analyzed more than 200 cases that were treated with the use of LTR and acquired 92% decannulation of the trachea after an average of 1.23 procedures for each patient (11). Zalzal passed about 90% of the decannulation of the trachea after the application of the front and back of the cartilage implant (12). All children after medical intervention were stented for a minimum of six weeks.
Quadrilateral cuts cricoid cartilage in the LTR was started in 1992 as one of the treatment options for severe subglottic stenosis of the larynx (13). The procedure provides for cutting not only the front and back wall of cricoid cartilage, but also to the side walls. This technique is often combined with the use of cartilage implant.
Treatments type LTR in the 90’s were divided into two groups: one-step (single-stage) or SS-LTR and two-stage (double-stage) – DS-LTR. SS-LTR technique involves the simultaneous removal of a tracheotomy during reconstructive surgery of the larynx and trachea (14, 15). Currently the procedure is dominated by resection of cricothyroid-tracheal (CTR). Partial cricothyroid-tracheal resection (PCTR) is increasingly being performed by surgery in severe narrowing of the larynx. It involves resection of the narrowed section of the respiratory tract and end-to-end anastomosis. Monnier among surgical patients rated their efficacy of this method as of 93% (16).
METHODS OF ENDOSCOPIC SURGERY
Methods of endoscopic treatment of stenosis of the larynx usually are reserved for benign strictures. This field also has changed a lot. Starting from the methods of widening probes by laser surgery or endoscopic treatment method using argon plasma coagulation. The availability of new technologies in medicine and modern tools in the hands of the surgeon usually makes the treatment more effective.
Endoscopic treatment of postintubation stenosis is still evaluated. Recently main attention has been directed at the local factors that may affect treatment outcomes. In view of the even increasing number of premature births and the need of prolonged intubation in preterm infants increases the risk of postintubation stenosis. New methods to continue treatment with the lowest risk for the patient, yet effective enough to lead to decannulation of the trachea as soon as possible, are still looked for.
Argon plasma coagulation (APC) is a treatment method based on the thermal effect induced in tissues by high-frequency currents. This method is used in surgery since the eighties of the twentieth century. At the beginning, this technique of the electrocoagulation has been used to stop bleeding from the large area of the partial liver resection. Properties APC showed it also that this method was applied in other areas of surgical treatment, becoming a very important tool in the open surgery and endoscopic surgery. Development of a flexible APC applicator allows for the use of APC in endoscopic procedures carried out with the use of flexible endoscopes. At present, APC has become one of the standard methods of treatment in gastroenterology and bronchoscopy. In 1997, for the first time Bergler applied the APC in (17). With the passage of time, new types of applicators of various shapes tailored for specific applications. Argon plasma coagulation is a non-contact method of monopolar electrosurgical coagulation. The basic principle of this method is to provide the working tip applicator with high-frequency alternating power voltage of 4000V and with thenoble gas such as argon. The flow through the argon the high frequency power reaches the ionization of the gas, which allows it to acquires the properties of a good conductor. Ionized argon or argon plasma is the fourth state of matter distinct from the solid, liquid and gas. Argon plasma allows high-frequency power to flow directly into the tissue without the need for contact with the applicator which is non-contact coagulation. The electric circuit in the case of monopolarelectrosurgery device application engineering is closed by the passive electrode, sworn to the patient. Argon plasma has a limited depth of penetration. This effect is due to evaporation phenomena in the tissue by the dehydration leading to a local increase of electrical resistance. Therefore, the electric power changes the direction of the movement by moving to areas with higher conductivity, or leaving the place already coagulated and covering their actions to untreated tissue. It is searching for ionized argon plasma radius areas with good conductivity. Thus it comes to uncontrolled deep penetration of the energy into the tissue. Expected depth of coagulation ranges from 0.5 to 3 mm. As coagulation takes place in an inert gas, a smoke release is negligible. By applying a high frequency current to the tissue reaches to rapid dehydration and shrinkage of collagen contained therein, which leads to the rapid reduction of the volume. On the coagulated superficial surface leading to the result of coagulation necrosis blanching mucosa, wherein it refers only to the superficial layer while maintaining the deeper layers, including the cartilage (18). When treatment is properly conducted treatment on the surface of tissue treated by APC on the surface of the coagulated tissue remains only a thin layer of the fibrin. With such of superficial effect it is very safe tool in the clearing of the tracheobronchial tree, larynx and esophagus and perforation risk is low. Argon plasma coagulation is an alternative technique for Nd-YAG laser in the clearing of the bronchial tree (19). APC has been applied at the Department of Pediatric Otolaryngology, Medical University of Warsaw in the treatment of postintubation subglottic stenosis of the larynx in children with good results thus expanding the possibilities of endoscopic surgery of the larynx of a new method with the use of modern medical technology (20, 21).
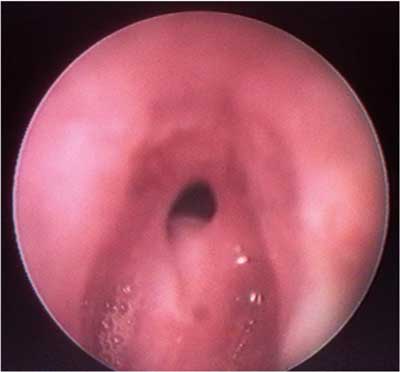
Fig. 1. Subglottic stenosis.

Fig. 2. Subglottic area after reconstruction.
CONCLUSIONS
Postintubation subglottic stenosis of the larynx in children is difficult to treat. There are many methods of treatment for a particular patient. Qualification of a patient to the type of the surgical method is based on the degree of stenosis. Effective, minimally invasive treatments, are still looked for.
Piśmiennictwo
1. Myer CM 3rd, Cotton RT: Historical development of surgery for pediatric laryngeal stenosis. Ear Nose Throat J 1995 Aug; 74(8): 560-562, 564. 2. McDonald IH, Stocks JG: Prolonged nasotracheal intubation. A review of its development in a paediatric hospital. Br J Aneaesth 1965 Mar; 37:161-173. 3. Bailey BJ, Biller HF: Surgery of the larynx. W.B. Saunders Company 1985: 3-14. 4. Fearon B, Cotton R: Surgical correction of subglottic stenosis of the larynx. Prelimenary report of an experimental surgical technique. Ann Otol Rhinol Laryngol 1972 Aug; 81(4): 508-513. 5. Grahne B: Operative treatment of severe chronic traumatic laryngeal stenosis in infants up to three years old. ActaOtolaryngol 1971 Jul-Aug; 72(1): 134-137. 6. Evans JN, Todd GB: Laryngo-tracheoplasty. J Laryngol Otol 1974 Jul; 88(7): 589-597. 7. Cotton RT, Seid AB: Management of the extubation problem in the premature child: anterior cricoid split as an alternative to tracheotomy. Ann Otol 1980; 89: 508-511. 8. LD, Stankiewicz JA, Livingston GL: Anterior cricoid split: the Chicago experience with an alternative to tracheotomy. Laryngoscope 1987 Jan; 97(1): 19-24. 9. Fearon B, Cinnamond M: Surgical correction of subglottic stenosis of the larynx. Clinical results of the Fearon-Cotton operation. J Otolaryngol 1976 Dec; 5(6): 475-478. 10. Hubbell RN, Zalzal G, Cotton RT, McAdams AJ: Irradiated costal cartilage graft in experimental laryngotracheal reconstruction. Int J Pediatr Otorhinolaryngol 1988 Feb; 15(1): 67-72. 11. Cotton RT, Gray SD, Miller RP: Update of the Cincinnati experience in pediatric laryngotracheal reconstruction. Laryngoscope 1989 Nov; 99(11): 1111-1116. 12. Zalzal GH: Treatment of laryngotracheal stenosis with anterior and posterior cartilage grafts. A report of 41 children. Arch Otolaryngol Head Neck Surg 1993 Jan; 119(1): 82-86. 13. Cotton RT, Mortelliti AJ, Myer CM 3rd: Four-quadrant cricoid cartilage division in laryngotracheal reconstruction. Arch Otolaryngol Head Neck Surg 1992 Oct; 118(10): 1023-1027. 14. Cotton RT, Myer CM 3rd, O’Connor DM, Smith ME: Pediatric laryngotracheal reconstruction with cartilage grafts and endotracheal tube stenting: the single-stage approach. Laryngoscope 1995 Aug; 105(8Pt1): 818-821. 15. Seid AB, Godin MS, Pransky SM et al.: The prognostic value of endotracheal tube-air leak following tracheal surgery in children. Arch Otolaryngol Head Neck Surg 1991 Aug; 117(8):880-882. 16. Monnier P, Savary M, Chapuis G: Partial cricoid resection with primary tracheal anastomosis for subglottic stenosis in infants and children. Laryngoscope 1993 Nov; 103(11 Pt 1): 1273-1283. 17. Bergler W Honig M, Gotte K et al.: Treatment of recurrent respiratory papillomatosis with argon plasma coagulation. J. LaryngolOtol 1997 Apr; 111(4): 381-384. 18. Sutedja G, Bollinger CT: Endobronchialelectrocautery and argon plasma coagulation. InterventionalBronchoscopy 2000; 30: 120-132. 19. Grochowski Z, Olechnowicz H: Zastosowanie plazmowej koagulacji argonowej w leczeniu nowotworowych i nienowotworowych zwężeń dróg oddechowych. Kardiochirurgia i TorakochirurgiaPolska 2006; 3(4): 377-382. 20. Zawadzka-Głos L, Chmielik M, Gabryszewska A: The application of argon plasma coagulation in the treatment of laryngeal stenosis in children. New Medicine 2001; 2, vol 4: 23-25. 21. Zawadzka-Głos L, Chmielik M, Gabryszewska A: The endoscopic treatment of postintubation laryngeal stenosis in children, using argon plasma coagulation. Int J Pediatric Otorhinolaryngology 2003, 67: 609-612.

