*Michał Michalik, Adrianna Podbielska-Kubera, Agnieszka Dmowska-Koroblewska
Diode laser-assisted uvulopalatoplasty using palisade technique
Leczenie chrapania metodą korekcji podniebienia miękkiego z wykorzystaniem lasera diodowego w technice palisadowej
Department of Otolaryngology, MML Medical Center, Warsaw, Poland
Head of Department: Michał Michalik, MD, PhD
Streszczenie
Wstęp. Drożność dolnej części górnych dróg oddechowych zależy od stabilnego napięcia mięśni gardła, podniebienia miękkiego i języka. Konsekwencją zmniejszenia napięcia mięśni oddechowych jest powtarzające się całkowite lub częściowe zwężenie dróg oddechowych. Wówczas przepływające powietrze wywołuje drgania tkanek miękkich, które słyszane jest jako chrapanie.
Materiał i metody. W badaniu brało udział 34 pacjentów: 27 mężczyzn i 7 kobiet. Zastosowano leczenie chrapania metodą korekcji podniebienia miękkiego z wykorzystaniem lasera diodowego dwutlenkowęglowego, o długości fali 810 nm, mocy 5 W oraz długości impulsacji 4 sekundy.
Przed wykonaniem badania pacjenci zostali poddani ocenie laryngologicznej, zebrano wywiad dotyczący chorób przewlekłych. Ponadto pacjenci wypełnili 2 ankiety: kwestionariusz do diagnostyki przesiewowej zaburzeń oddychania w czasie snu oraz kwestionariusz Skali Senności Epworth. Dodatkowo wykonano badanie tomografii komputerowej 3D oraz rynometrię akustyczną. Wszystkie badania powtórzono 3 miesiące po zabiegu.
Wyniki. Zabieg wykonano u 34 osób. Ustąpienie dolegliwości zaobserwowano u 21 osób, a zmniejszenie objawów – u 13 osób. W grupie badawczej nie znaleźli się pacjenci, u których nie nastąpiłaby poprawa. Żaden z pacjentów nie zgłosił powikłań.
Wnioski. U pacjentów uzyskano dobry efekt usztywnienia struktur podniebienia, co przyczyniło się do ustąpienia chrapania. Do zalet stosowania lasera diodowego w technice palisadowej zaliczamy bezpieczeństwo stosowania, małą inwazyjność, krótki czas gojenia, minimalne ryzyko powikłań. Zabieg przeprowadzany jest w trybie ambulatoryjnym, w znieczuleniu miejscowym.
Summary
Introduction. The patency of the lower part of the upper respiratory tract depends on the muscle tone of pharynx, soft palate and tongue muscles. The consequence of the lowered tone of these muscles is recurrent total or partial narrowing of the airways. In this case, the turbulent airflow causes soft tissue vibration, which is heard as snoring.
Material and Methods. The study included 34 patients: 27 men and 7 women. Carbon dioxide diode laser-assisted uvulopalatoplasty was employed. The diode laser with a wavelength of 810 mm, power of 5 W, and pulse length of 4 seconds was used.
Before the procedure, all the patients underwent laryngological assessment. Medical history of the patients was also collected. In addition, the patients completed 2 questionnaires: a sleep disorder screening questionnaire and Ephworth Sleepiness Scale. Moreover, a 3D CT scan and acoustic rhinometry were performed. All the tests were repeated 3 months after the procedure.
Results. The procedure was performed in 34 patients. Complete clinical response was observed in 21 cases, and a partial response was seen in 13 cases. There were no patients in the study group who had not experienced at least a partial improvement of symptoms. None of the patients reported any complications.
Conclusions. Good clinical outcome was obtained in all the patients after obtaining palatal stiffening, which contributed to the resolution of symptoms. The advantages of diode laser-assisted uvulopalatoplasty using palisade technique include the safety of the procedure, minimal invasiveness, short healing time, and a low risk of complications. The procedure is performed on an out-patient basis under local anesthesia.
Introduction
Sleep breathing disorders
Sleep breathing disorders include upper airway resistance syndrome, obstructive sleep apnea, and primary snoring (1).
The patency of the lower part of the upper respiratory tract (i.e. not including nasal cavity) depends on the muscle tone of pharynx, soft palate and tongue muscles (1). The major part of the upper and lower airways are supported with osseous (nose, larynx) or cartillagenous structures, thus maintaining their patency (1). Pharyngeal walls, on the other hand, consist entirely of soft tissues. The pharyngeal patency is maintained by the muscle tone of muscles consisting the anterior and lateral pharyngeal walls (1).
The consequence of the lowered tone of these muscles is recurrent total or partial narrowing of the airways (1). In this case, upper airway negative pressure during inspiration is higher than the tone of the stabilizing muscles. As a consequence, oropharyngeal isthmus narrows, and the turbulent airflow causes soft tissue vibration, which is heard as snoring (fig. 1) (1). The air velocity increases at the narrowest part of the airways (Bernouilli effect) (2). When critical closing pressure is reached, airway narrowing or closure ensues (2). Narrowing or closure of upper airways may occur along their entire length, but most frequently, oropharynx and laryngopharynx are affected (1). The following hypoxia and hypercapnia cause increased respiratory effort and awakening, which contributes to an increase of the muscle tone and restoring pharyngeal patency. The resulting short-term hyperventilation allows to fall back to sleep (2).
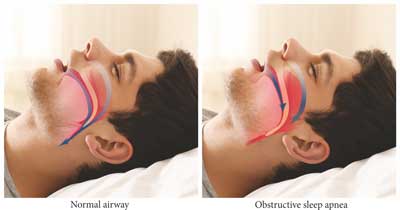
Fig. 1. Comparison of normal airway vs. airways in obstructive sleep apnea
Snoring
Snoring is the vibration of upper respiratory structures and the resulting sound due to obstructed air movement during breathing (3). In the initial stage, snoring can go unnoticed by the patient, however, it is burdensome for the patient’s family. The results of a poll performed by TNS Polska in 2016 commissioned by MML Medical Center show that the number of patients diagnosed with snoring has been growing in the recent years, which is a result of a more universal access to diagnostics, as well as of the increased awareness of the patients (4). Snoring directly affects the quality of sleep and, in the opinion of the respondents, may result in a number of health problems (4). Snoring is observed in ca. 20% of men and 5% of women aged 30–35 (3). The prevalence increases with age. In the population of over 60 years of age, the prevalence of persons snoring is estimated at 60% in men and 40% in women (3).
The most serious complication of snoring is obstructive sleep apnea (5). Obstructive sleep apnea (OSA) is characterized by repetitive episodes of shallow or paused breathing during sleep, associated with a reduction in blood oxygen saturation and awakening (5). Sleep apnea is observed in the majority of patients who snore (3). Main complications of sleep breathing disorders include constant tiredness, xerostomia, headaches, daytime sleepiness, cognitive impairment, as well as depression (4). OSA is also associated with an increased risk of serious comorbidities, such as cardiovascular diseases, cerebrovascular accidents, diabetes mellitus, hypertension, and post-operative cardiovascular and respiratory complications (5). Cardiovascular Health Study (6) confirmed the relationship between OSA symptoms, including daytime sleepiness, apnea and snoring with higher fasting glucose and 2-hour postprandial glucose, as well as decreased insulin sensitivity.
Snoring may be a result of pathologies in the nasal cavity, pharynx, larynx, or all of these structures (5). The pathologies of the nasal cavity include nasal turbinate hypertrophy, deviated septum, and nasal polyps (5). The main pharyngeal anomalies that may be responsible for snoring comprise: uvular hypertrophy, macroglossia, flaccid or hypertrophied soft palate, tonsillar hypertrophy, and drooping lateral pharyngeal walls (5).
Figure 2 presents a computed tomography scan of a patient with restored normal airway patency after the surgery. Figure 3 presents a 3 dimensional computed tomography reconstruction that allows to determine the level of the obstruction.
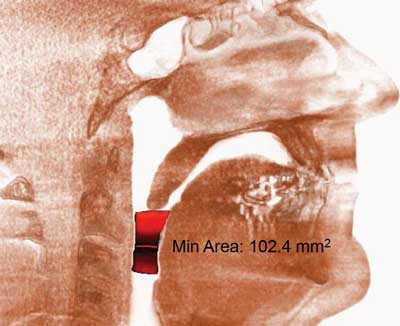
Fig. 2. Computed tomography scan presenting a restored airway patency after surgery
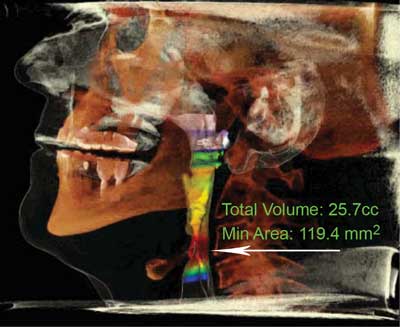
Fig. 3. Determining obstruction level (marked with an arrow) in 3D CT reconstruction
Diagnostics and treatment of snoring
Snoring is an indication for a laryngological consultation and diagnostics. The first stage of diagnostics comprises of a detailed interview and physical examination. Next stages include qualification for nose, palate, and tongue surgery (5).
Several questionnaires have been developed to identify patients with an increased risk of snoring.
Questionnaires consist of questions concerning the signs of sleep breathing disorders (e.g. daytime fatigue or sleepiness), comorbidities (e.g. hypertension, obesity), as well as individual risk factors of the patient (age, sex, neck size) (7).
Among the most frequently used questionnaires is the STOP-BANG questionnaire. The questionnaire focuses on the following signs and risk factors: snoring (S), tiredness (T), observed stop of breathing (O), blood pressure (P), BMI over 35 (B), age over 50 (A), large neck size (N), and gender – male (G) (1). Another option is to use the Berlin Questionnaire (BQ), which assesses the presence and frequency of snoring, presence of apnea, daytime sleepiness, hypertension, and obesity (1). BQ a has high sensitivity (69–86%) and specificity (56–95%) in OSA diagnosis (5).
Epworth Sleepiness Scale is a useful tool as well. It is used to assess daytime sleepiness. The patient is asked to assess his chance of falling asleep in 8 different settings (8). Scored of 0–24 points are obtained. A result of above 10 points suggests sleep breathing disorder, as opposed to generalized fatigue (4).
The questionnaires alone are considered to be insufficient for screening (5). In addition to questionnaires, polysomnography, drug-induced sleep endoscopy, 3D computed tomography, laboratory tests, and home sleep apnea testing are used in the diagnostic process.
Polysomnography is a standard for the diagnosis of sleep breathing disorders (2, 7). This method employs multiple sensors that allow to simultaneously record the air flow, blood oxygen saturation, respiratory effort, and electrical activity of heart, brain, eyes, and muscle. Additional sensors include sensors of body position, chest, abdomen, and limb movements, as well as cameras and microphones (9). Polysomnography is expensive, time-consuming, and requires specialized equipment in a sleep laboratory and qualified personnel (9).
Polygraphy is a simplified version of polysomnography. It can be used as a screening test and performed in a hospital or at home (10). Polygraphy does not include electroencephalography, electrooculography, and muscle tone measurement. It is less precise than polysomnography, however, it is usually enough to make the right diagnosis (10). In some cases, it may be necessary to perform full polysomnography (10).
Three-dimensional computed tomography (CT) reveals sites of obstruction in the upper respiratory tract, including many abnormalities associated with snoring, including deviated nasal septum, nasal turbinate hypertrophy, pharyngeal and tonsillar hypertrophy, as well as macroglossia or hypertrophied soft palate (11). The advantages of 3D CT include being painless, non-invasive, and quick.
It is possible to perform a home sleep apnea testing. This testing is indicated in adults aged from 18 to 65 with a high probability of moderate to severe OSA, as well as in persons in which it is not possible to perform polysomnography for safety reasons or due to the need to remain immobile, moderate to severe respiratory disease, neuromuscular disease, and cardiac insufficiency (8). Home sleep apnea testing are a useful supplement to the diagnostics of obstructive sleep apnea, as it offers a simple, objective and cost-effective tool to assess the symptoms of OSA. Home sleep apnea testing may, however, underestimate the severity of sleep breathing disorders. Moreover, it is not known to what extent the test time registered by the device represents the actual sleep (12).
Choosing the right treatment method depends on many factors that affect the anatomy of the head and neck region. These factors include, among others, age, sex, body weight, and race (5). In addition, symptoms reported by the patient should be taken into account (5). It is crucial to determine the obstruction site that is the cause of apnea and snoring. In parallel with the surgical treatment, lifestyle changes are necessary. These include: weight loss, smoking cessation, avoiding intake of sedatives, caffeine restriction, avoiding alcohol consumption in the evening, reducing noise levels in bedroom and its adequate ventilation, comfortable bed, appropriate amount of sleep, and maintaining correct body position during sleep (2, 5).
In the treatment of snoring and apnea, it may be useful to introduce devices and techniques that facilitate breathing (CPAP – continous positive airway pressure) (13). Oral appliances (OAs) are frequently used, as they improve the patency of upper respiratory tract by changing the position of the tongue and related structures of the upper respiratory tract (13).
Sublingual nerve stimulation is effective in patients with multilevel obstruction in the upper respiratory tract (14).
Nasal and sinus surgery improve upper respiratory tract patency and increase the efficacy of CPAP and oral appliances (2). In case of pathologies of the nasal septum, septoplasty is performed (2). Another cause of difficulty in nasal breathing is turbinate hypertrophy. In these cases, conchoplasty is performed. Tonsillectomy is currently a standard surgical treatment for sleep breathing disorders in children caused by tonsillar hypertrophy (6, 15). Surgical procedures in patients with sleep apnea also include baratric surgery facilitating weight loss, as well as tracheostomy (16). Tracheostomy is indicated in patients who refused CPAP treatment and in patients in whom other surgical approaches failed (17).
Advanced surgical treatment methods for apnea and snoring include multilevel surgery – uvulopalatopharyngoplasty, tongue surgery, mandibular surgery, etc.
Uvulopalatopharyngoplasty consists of surgical removal of uvula and part of soft palate in order to widen oropharyngeal isthmus and eliminate obstruction present in the oropharynx (1). Maxillomandibular advancement surgery (MMA) moves mandible forward.
The use of minimally invasive surgical techniques enables the patient to return home at the day of the surgery. What is more, minimally invasive surgical techniques significantly reduce the risk of bleeding and post-surgical complications. These procedures are indicated in patients with deviated nasal septum, macroglossia, and tonsillar hypertrophy. In the MML Medical Centre, there are many techniques used for treatment of sleep breathing disorders. The most commonly used include: radiofrequency induced thermotherapy (RFITT), plasma surgery, harmonic scalpel assisted surgery, introducing polyethylene (PET) implants, and diode laser assisted surgery.
Radiofrequency induced thermotherapy consists of the change in proportion of the structures of the soft palate and tongue using electromagnetic waves (17). In case of soft palate hypertrophy, the aim of the procedure is to create small scarring within the palate that, previously drooping, becomes more rigid (18). This technique is characterized by fast wound healing (19).
Harmonic scalpel assisted surgery uses ultrasonographic vibrations for cutting and instant tissue coagulation (20). The scalpel begins to operate only in motion, by changing electrical energy into mechanical energy. The use of the scalpel reduces the duration, as well as intraoperative blood loss, of many otolaryngological surgical procedures (20).
A method of treatment for snoring and sleep apnea that has been introduced by the MML Medical Centre is the minimally invasive diode laser-assisted uvulopalatoplasty using palisade technique. The uses of laser-assisted surgery include palatoplasty, surgery of palatoglossal and palatopharyngeal arch, and uvuloplasty. During the procedure, the diode laser fiber is introduced into the soft palate, which results in the formation of linear intra-parenchymal adhesions that stiffen the palate and shift it in the vertical plane (fig. 3). This prevents the tissue from vibrating during sleep, which, in turn, leads to increased sleeping comfort and maximally widened airways. This technique allows a fast and efficient airflow improvement, eliminating the symptoms of sleep breathing disorders. The procedure lasts about 30 minutes and is performed on an out-patient basis under local anesthesia or under sedation. The patients are able to return to normal activity after a short period of recovery. This is undoubtfully a significant advantage of the diode laser-assisted uvulopalatoplasty using palisade technique over other types of procedures.
Aim
The aim of this paper was to present the efficacy of diode laser-assisted uvulopalatoplasty using palisade technique in treatment of drooping or elongated soft palate in patients suffering from sleep breathing disorders.
Material and methods
This retrospective study included 34 patients: 27 men and 7 women. Carbon dioxide diode laser-assisted uvulopalatoplasty was employed. The diode laser with a wavelength of 810 mm, power of 5 W, and pulse length of 4 seconds was used.
A medical history of chronic diseases was collected in all the patients. In addition, the patients were asked to fill in 2 questionnaires: a screening questionnaire for sleep breathing disorders (extended, original questionnaire based on the Berlin Questionnaire) (fig. 4) and Epworth Sleepiness Scale prepared by British Snoring & Sleep Apnea Association (fig. 5), used to assess sleepiness and probability of falling asleep in everyday situations (21).



Fig. 4. Original questionnaire for diagnosing breathing disorders during sleep

Fig. 5. Epworth Sleepiness Scale
The patients underwent laryngological examination including external nose, anterior nares, nasal septum, and inferior nasal turbinates. The soft palate was also assessed with Mallampati Score (tab. 1), and the assessment of tonsils was performed with the help of the Pirquet Tonsillar Hypertrophy Grading Scale (tab. 2). The examination was performed on an out-patient basis.
Tab. 1. Mallampati score
| Grade | Description |
| 1. | soft palate, uvula, fauces, pillars visible |
| 2. | soft palate, uvula, fauces visible |
| 3. | soft palate, base of uvula visible |
| 4. | soft palate not visible |
Tab. 2. Pirquet score
| Grade | Description |
| 1. | Tonsils hidden behind tonsillar pillars |
| 2. | Tonsils extend to pillars |
| 3. | Tonsils visible beyond pillars |
| 4. | Tonsils covering 50% of space between pillars |
| 5. | Tonsils extend to midline |
In all the patients, a 3D computed tomography scan was performed in order to reveal the pathological structures that were responsible for snoring. In case of our study group, sites of obstruction in the upper respiratory tract included deviated nasal septum, nasal turbinate hypertrophy, pharyngeal and tonsillar hypertrophy, as well as macroglossia or hypertrophied soft palate. In every patient, acoustic rhinometry was also performed.
During the procedure, supine position on the operating table was used. Standard intraoperative monitoring included ECG, heart rate, blood pressure, and oxygen saturation. Regional surface anesthesia with 10% lidocaine and infiltrative anesthesia with Dentocaine (40 mg artcaine hydrochloride and 0.01 mg adrenaline) were used.
All the patients were prescribed analgesics. Directly after the procedure, the patients were allowed to leave the clinic, and within 2–3 hours, they could ingest food and beverages. The control visits were planned 1 week and 6 weeks after the procedure. The control included questionnaires, medical interview and endoscopic examination.
Treatment outcomes were assessed 3 months after the procedure. Laryngological examination, as well as endoscopic examination, , 3D computed tomography, and acoustic rhinometry were performed. The patients were also asked to complete the questionnaires again.
If soft palate was sufficiently rigid and the symptoms persisted, the differential diagnosis was extended to other procedures mentioned above, depending on the clinical evaluation of the patient.
Results
Between the years 2007 and 2017, 12,911 procedures for snoring and sleep apnea have been performed in the MML Medical Centre. The patients included 3,228 women and 9,683 men. Mean age of the patients was 46 years in case of men and 35 years in case of women. Mean disease duration was 4 years.
Thirty-four diode laser-assisted uvulopalatoplasty procedures using palisade technique were performed. Complete clinical response was observed in 21 cases, and a partial response was seen in 13 cases. There were no patients in the study group who had not experienced at least a partial improvement of symptoms. There were no complications, such as postoperative bleeding, hyperthermia, or dyspnea.
Treatment outcomes were assessed 3 months after the procedure. A patient satisfaction survey concerning treatment outcomes revealed that 26 patients (76%) were very satisfied and 8 patients (24%) were rather satisfied with the outcomes. The quality of sleep also improved – 3 months after the procedure, 30 out of 34 patients (88%) felt less pressure in the pharynx and declared that they could breathe better. Moreover, a decrease of the snoring volume was confirmed in the subjective assessment of the partners of the patients, as well as a reduced frequency of awakenings due to snoring, and decreased xerostomia, were confirmed. The full results are currently undergoing analysis.
Discussion
Considering the complexity of the patophysiology of sleep apnea, the management of this disease should be discussed in multidisciplinary teams including sleep disorder specialists, orthodontists, maxillofacial surgeons, and otolaryngologists (2).
Treatment of apnea and snoring can be performed on an out-patient or in-patient basis. Patients with minor lesions are referred for out-patient treatment. Out-patient procedures are indicated in cases of turbinate hypertrophy, drooping soft palate, and uvular hypertrophy. This type of treatment is minimally invasive, and requires no special preparation. The procedure is short, usually up to 30 minutes, and is performed under local anesthesia. Bleeding and tissue irritation are minimal.
Surgical procedures may be used as a first-line treatment in selected patients in all stages of OSA, in whom there are anatomical abnormalities contributing to collapsing of the upper respiratory tract during sleep (2).
Multilevel surgery is performed in adult patients with OSA in whom conventional and alternative treatment methods have failed (22). Success rates for these procedures vary widely and depend on the surgical approach, as well as surgeon’s experience (12). Lower age of the patient (< 60 years of age), lower BMI (< 30 kg/m2) and lack of comorbidities are associated with higher success rates (22).
Currently, minimally invasive surgical techniques have become increasingly popular. Their main use is the treatment of breathing disorders caused by the obstruction within the upper respiratory tract. In studies on the effectiveness of laser-assisted base of tongue reduction and epiglottis surgery, promising results and a significant improvement in the AHI (apnea-hypopnea index) were obtained (17).
An approach that has been developed by the MML Medical Centre is diode laser-assisted uvulopalatoplasty using palisade technique. Based on our observations, it is a method that is safe and minimally invasive, and enables short healing time, minimal risk of complications, and minimal pain. The advantage of diode laser-assisted uvulopalatoplasty using palisade technique over other surgical approaches include primarily the possibility of performing the procedure in a short time (30–40 minutes) under local anesthesia on an out-patient basis. Immediately after the procedure, the patient can return to their daily activities. This is an unquestionable advantage when compared with radiofrequency induced thermotherapy, in which recovery time is several days, and postoperative pain may require the use of analgesics.
Conclusions
1. The introduced surgical approach makes it possible to obtain a subjective symptom relief in OSA patients.
2. This technique results in stiffening of drooping soft palate in the vertical plane by creating intraparenchymal adhesions.
3. Diode-assisted uvulopalatoplasty is an important alternative to other methods of treatment for sleep breathing disorders.
Piśmiennictwo
1. Slowik JM, Collen JF: Apnea, Obstructive Sleep Apnea. In: StatPearls. Treasure Island (FL): StatPearls Publishing. 2017; Available from: https://www.ncbi.nlm.nih.gov/books/NBK459252/.
2. Memon J, Manganaro SN: Apnea, Snoring And Obstructive Sleep, CPAP. In: StatPearls. Treasure Island (FL): StatPearls Publishing. 2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441909/.
3. Chruściel-Nogalska M, Kozak M, Ey-Chmielewska H: Zespół obturacyjnego bezdechu podczas snu – podstawy diagnostyki i leczenia 2015; Dent For 2015; 43: 65-69.
4. Badanie wśród ogółu Polaków na temat chrapania. Raport TNS Polska dla Centrum Medycznego MML. 2016.
5. Chung JW, Kim N, Wee JH et al.: Clinical features of snoring patients during sedative endoscopy. Korean J Intern Med 2017; Available from: http://europepmc.org/abstract/med/29132198.
6. Muraki I, Wada H, Tanigawa T: Sleep apnea and type 2 diabetes. J Diabetes Investig 2018; 18. Available from: https://onlinelibrary.wiley.com/doi/abs/10.1111/jdi.12823.
7. Corso R, Rusotto V, Gregoretti C, Cattano D: Perioperative management of obstructive sleep apnea: a systematic review. Minerva Anestesiol 2017; 84(1): 81-93.
8. Foldvary-Schaefer NR, Waters TE: Sleep-Disordered Breathing. Continuum (Minneap Minn) 2017; 23(4): 1093-1116.
9. Sutherland K, Almeida FR, de Chazal F, Cistulli PA: Prediction in obstructive sleep apnoea: diagnosis. Expert Rev Respir Med 2018; 12(4): 293-307.
10. Cundrle I Jr, Belehrad M, Jelinek M et al.: The utility of perioperative polygraphy in the diagnosis of obstructive sleep apnea. Sleep Med 2016; 25: 151-155.
11. Chousangsuntorn K, Bhongmakapat T, Apirakkittikul N et al.: Computed Tomography Characterization and Comparison With Polysomnography for Obstructive Sleep Apnea Evaluation. J Oral Maxillofac Surg 2017; 17: 31170-31179.
12. Aurora RN, Putcha N, Swartz R, Punjabi NM: Agreement Between Results of Home Sleep Testing for Obstructive Sleep Apnea with and Without a Sleep Specialist. Am J Med 2016; 129(7): 725-730.
13. Dicus Brookes CC, Boyd SB: Controversies in Obstructive Sleep Apnea Surgery. Oral Maxillofac Surg Clin North Am 2017; 29(4): 503-513.
14. Wray CM, Thaler ER: Hypoglossal nerve stimulation for obstructive sleep apnea: A review of the literature. World J Otorhinolaryngol Head Neck Surg 2016; 2(4): 230-233.
15. Zhang LY, Zhong L, David M, Cervin A: Tonsillectomy or tonsillotomy? A systematic review for paediatric sleep-disordered breathing. Int J Pediatr Otorhinolaryngol 2017; 103: 41-50.
16. Soose RJ: Novel Surgical Approaches for the Treatment of Obstructive Sleep Apnea. Sleep Med Clin 2016; 11(2): 189-202.
17. Ferguson MS, Magill JC, Kotecha BT: Narrative review of contemporary treatment options in the care of patients with obstructive sleep apnoea. Ther Adv Respir Dis 2017; 11(11): 411-423.
18. Olszewska E, Rutkowska J, Czajkowska A, Rogowski M: Selected surgical managements in snoring and obstructive sleep apnea patients. Med Sci Monit 2012; 18(1): CR13–CR18.
19. Hwang SY, Jefferson N, Mohorikar A, Jacobson I: Radiofrequency Coblation of Congenital Nasopharyngeal Teratoma: A Novel Technique. Case Rep Otolaryngol 2015; 2015: 634958.
20. Polacco MA, Pintea AM, Gosselin BJ, Paydarfar JA: Parotidectomy using the Harmonic scalpel: ten years of experience at a rural academic health center. Head Face Med 2017; 13: 8.
21. Johns MW. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep 1991; 14: 540-545.
22. Cao MT, Sternbach JM, Guilleminault C: Continuous positive airway pressure therapy in obstuctive sleep apnea: benefits and alternatives. Expert Rev Respir Med 2017; 11(4): 259-272.
otrzymano: 2018-05-15
zaakceptowano do druku: 2018-06-21
Adres do korespondencji:
*Michał Michalik
Centrum Medyczne MML ul. Bagno 2, 00-112 Warszawa
Tel.: + 48 608-381-781
e-mail: m.michalik@mml.com.pl
New Medicine 2/2018Strona internetowa
czasopisma New MedicinePozostałe artykuły z numeru 2/2018:






