© Borgis - Postępy Fitoterapii 4/2014, s. 195-199
Radosław J. Ekiert1*, Jan Krzek1, Magdalena Lenartowicz1, Halina Ekiert2
Analiza stewiozydu oraz rebaudiozydu A w słodzikach
Analysis of stevioside and rebaudioside A in sweeteners
1Department of Inorganic and Analytical Chemistry, Jagiellonian University, Medical College, Cracow, Poland
Head of Department: prof. dr hab. Jan Krzek
2Department of Pharmaceutical Botany, Jagiellonian University, Medical College, Cracow, Poland
Head of Department: prof. dr hab. Halina Ekiert
Streszczenie
Produkty z rośliny stewia mają coraz większe znaczenie, ponieważ wykazują korzystne właściwości żywieniowe i lecz-nicze. Głównymi związkami aktywnymi biologicznie, zawartymi w tego typu produktach, są dwa glikozydy stewiolowe: stewiozyd i rebaudiozyd A. Są one około 300 razy słodsze od sacharozy. Produkty obecne na rynku mogą istotnie różnić się jakością. W celu weryfikacji jakości słodzików ze stewii, opracowano nową procedurę analityczną. Wykorzystano metodę wysokosprawnej chromatografii cienkowarstwowej z detekcją densytometryczną, która zapewniła odpowiednią zdolność rozdzielczą, możliwość identyfikacji oraz oznaczania ilościowego. Proponowana procedura umożliwia dokonywanie oznaczeń w sposób prosty i bezpośredni, dając wiarygodne rezultaty. Celem pracy była ocena jakości kilku słodzików z uwzględnieniem zawartości stewiozydu i rebaudiozydu A, czasu rozpadu, masy i czystości tabletek. Rezultaty wskazują na znaczące różnice we właściwościach fizykochemicznych słodzików, co wpływa na ich właściwości sensoryczne i biologiczne.

Introduction
Stevioside and rebaudioside are ent-kaurene glycosides becoming form leaves of Stevia rebaudiana (Bertoni), the plant native to Paraguay and Brazil. Stevia has been used by Indians for centuries with tea or yerba mate to make the beverage sweet. The glycosides exert strong sweetness and are used in many low calorie sweeteners of natural origin. Nowadays, the high-scale usage of these compounds was initiated in Japan over 40 years ago, then it was cultivated in many countries all over the world (1-3). The leaves contain 4-20% diterpene glycosides (3, 4).
The Food and Drug Administration classified rebaudioside A as generally recognized as safe (5). The Joint FAO/WHO Expert Committee on Food Additives defined the admissible daily intake of stevioside as 0-10 mg/kg of body weight (6). In 2011 glycosides were accepted for usage in whole European Union as food additive E960 (7). The above mentioned substances have also many other beneficial values, they act as antihyperglycemic, hypotensive, antihyperlipidemic, antiinflammatory, antioxidant, antiviral, DNA-protective and chemopreventive agents (1, 4, 8-10). The further studies are required to justify that effects other than sweetness are clinically significant.
The glycosides from stevia are heat stable up to 200°C, so they can be used in variety of pastries. They are proper for cooking and baking. They also tolerate low pH values (were stable for 2 hours at 60°C in pH range 1-10) and do not undergo fermentation (3, 11). It is believed that they do not pose any allergic risk (3, 9). Chemical structure of stevioside and rebaudioside A are presented on figure 1.
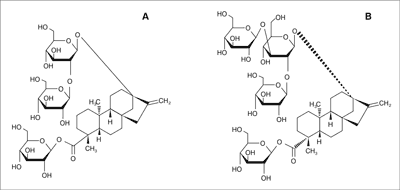
Fig. 1. Chemical structure of stevioside (A) and rebaudioside A (B).
Many chromatographic procedures, due to separation potential, were proposed for determination of steviol glycosides, including especially high-performance liquid chromatography HPLC (12-16) and thin-layer chromatography TLC (12, 15, 17-22).
TLC is a very useful and efficient tool in plant material analyses. It do not require high purification of analyzed samples, what is a necessity in column separation techniques. All hitherto published TLC procedures are laborious and require use of dying reagents. Because of lack of any simple, direct and effective high performance TLC procedure with densitometric detection, authors find it reasonable and valuable to develop a new procedure fulfilling these criteria.
Materials and methods
Reagents
All chemicals were of analytical grade and purchased from Chempur (Piekary Śląskie, Poland). Reference standards: stevioside hydrate (purity ≥ 98% by HPLC) and rebaudioside A (purity ≥ 96% by HPLC) were from Sigma-Aldrich (St. Louis, USA). Standard solutions, prepared with water-methanol mixture (1+4, v/v) with concentration of 2 mg/ml were used.
Material
Five sweeteners coming from European Union market: Sweetiva Stevia, Süssina Stevia, Stevia Green Leaf, Stewia Planta Dulce and Stevija were investigated. The water-methanolic solutions were made. Milled tablet mass in amount 500 mg was extracted with mixture composed of 1 ml of bidistilled water and 4 ml of methanol for 15 min. The turbid solutions need to be filtered once (Stewia Planta Dulce trice) by filtration paper No 2.
Storage: the reference standards, standard and sample solutions were kept in a refrigerator at approx. 2-8°C protected from light. The sample solutions should be kept for no longer than 3 days. The sweeteners were stored in room temperature 15-25°C not exposed to the direct radiation.
Apparatus
An 18 cm x 9 cm x 18 cm, Sigma Aldrich (St. Louis, USA) vertical chromatographic chamber, Linomat 5 band applying module and Scanner 3 densitometer were used. Additionally, a fluorescence detector at a wavelength of 254 nm was used in order to observe the quenching of fluorescence by the investigated substances what enable assessment of application correctness and preliminary separation efficiency. All three pieces of equipment were from Camag (Muttenz, Switzerland).
Method parameters
Application
The standard solutions and sample solutions were applied onto HPTLC silica gel 60 plates of 10 cm x 10 cm in size with F254 fluorescence factor (Merck, Darmstadt, Germany). The solutions were applied with a Linomat module with rate equal 200 nl/s. The bands were formed 10 mm from the bottom of the plate edge, while the front of chromatograms was fixed at 5 mm from the upper edge of the plate. There were six tracks on each plate. Band width was 7 mm and space between bands was 8 mm. The development distance equaled 85 mm.
The standard solutions were applied on chromatographic plate in volumes from 1.0 μl to 50.0 μl in order to establish method’s analytical range which was determined as 7.5-22.5 μl for stevioside and 5.0-25.0 μl for rebaudioside A. In the case of sample solutions the application procedure was adjusted to method’s range and required separate analysis of two analytes. The analyses of stevioside required bigger volums of sweeteners’ solutions comparing with rebaudioside A. In case of Sweetiva Stevia, Süssina Stevia and Stewia Planta Dulce, there was applied 10 μl of solutions in order to determine stevioside and 5 μl for determination of rebaudioside A. Stevia Green Leaf and Stevija required 3 μl and 2 μl respectively.
Separation
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Buszewski B, Noga S. Hydrophilic interaction liquid chromatography (HILIC)–a powerful separation technique. Anal Bioanal Chem 2012; 402:231-47. 2. Cardello HMAB, da Silva MAPA, Damasio MH. Measurement of the relative sweetness of stevia extract, aspartame and cyclamate/saccharin blend as compared to sucrose at different concentrations. Plant Foods Human Nutr 1999; 54:119-30. 3. Lemus-Mondaca R, Vega-Gálvez A, Zura-Bravo L et al. Stevia rebaudiana Bertoni, source of a high-potency natural sweetener: a comprehensive review on the biochemical, nutritional and functional aspects. Food Chem 2012; 132:1121-32. 4. Pawar RS, Krynitsky AJ, Rader JI. Sweeteners from plants – with emphasis on Stevia rebaudiana (Bertoni) and Siraitia grosvenorii (Swingle). Anal Bioanal Chem 2013; 405:4397-407. 5. FDA. GRAS notice for rebaudioside A (REB A) No. 252, 2008. 6. JECFA.
Joint FAO/WHO Expert Committee on Food Additives Monographs 5, Compendium of Food Additive Specifications from 69th JEFCA Meeting, 2008. 7. EU. Commission Regulation No 1131/2011, 2011. 8. Baechler BJ, Nita F, Jones L et al. A novel liquid multi-phytonutrient supplement demonstrates DNA-protective effects. Plant Foods Human Nutr 2009; 64:81-5. 9. Goyal SK, Samsher, Goyal RK. Stevia (Stevia rebaudiana) a bio-sweetener: a review. Int J Food Sci Nutr 2010; 61:1-10. 10. Madan S, Ahmad S, Singh GN et al. Stevia rebaudiana (Bert.) Bertoni – a review. Indian J Nat Prod Resour 2010; 1:267-86. 11. Šic-Žlabur J, Voća S, Dobričević N et al. Stevia rebaudiana Bertoni – a review of nutritional and biochemical properties of natural sweetener. Agric Consp Sci 2013; 78:25-30. 12. Dacome AS, da Silva CC, da Costa CEM et al. Sweet diterpenic glycosides balance of a new cultivar of Stevia rebaudiana (Bert.) Bertoni: Isolation and quantitative distribution by chromatographic, spectroscopic, and electrophoretic methods. Process Biochem 2005; 40:3587-94. 13. Gardana C, Scaglianti M, Simonetti P. Evaluation of steviol and its glycosides in Stevia rebaudiana leaves and commercial sweetener by ultra-high-performance liquid chromatography-mass spectrometry. J Chromatogr A 2010; 1217:1463-70. 14. Kolb N, Herrera JL, Ferreyra DJ et al. Analysis of sweet diterpene glycosides from Stevia rebaudiana: improved HPLC method. J Agric Food Chem 2001; 49:4538-41. 15. Nikolova-Damyanova B, Bankova V, Popov S. Separation and quantitation of stevioside and rebaudioside A in plant extracts by normal-phase high performance liquid chromatography and thin-layer chromatography: a comparison. Phytochem Anal 1994; 5:81-5. 16. Woelwer-Rieck U, Lankes C, Wawrzun A et al.: Improved HPLC method for the evaluation of the major steviol glycosides in leaves of Stevia rebaudiana. Europ Food Res Tech 2010; 231:581-8. 17. Chester K, Tamboli ET, Singh M et al. Simultaneous quantification of stevioside and rebaudioside A in different stevia samples collected from the Indian subcontinent. J Pharm Bioall Sci 2012; 4:276-81. 18. Fullas F, Kim J, Compadre CM et al. Separation of natural product sweetening agents using over pressured layer chromatography. J Chromatogr 1989; 464:213-9. 19. Jaitak V, Gupta AP, Kaul VK et al. Validated high-performance thin-layer chromatography method for steviol glycosides in Stevia rebaudiana. J Pharm Biomed Anal 2008; 47:790-4. 20. Kedik SA, Fedorov SV, Yanul’ NA et al. Medicinal plants. Chromatographic determination of stevioside in raw plant material. Pharm Chem J 2003; 37:19-22. 21. Londhe SV, Nanaware SM. HPTLC method for simultaneous analysis of stevioside and rebaudioside-A in Stevia rebaudiana. J AOAC Int 2013; 96:24-6. 22. Shirwaikar A, Parmar V, Bhagat J et al. Identification and estimation of stevioside in the commercial samples of stevia leaf and powder by HPTLC and HPLC. Int J Pharm Life Sci 2011; 2:1050-8. 23. The fitness for purpose of analytical methods: A laboratory guide to method validation and related topics. Eurachem 1998. 24. Harmonised Tripartite Guideline. Validation of analytical procedures: Text and methodology, Q2(R1). ICH 2005. 25. Komsta Ł. Chemometric and statistical evaluation of calibration curves in pharmaceutical analysis – a short review on trends and recommendations. J AOAC Int 2012; 95:669-72.

