© Borgis - Postępy Nauk Medycznych 12/2014, s. 828-834
*Jarosław Kozakowski, Renata Kapuścińska, Wojciech Zgliczyński
Rozpoznawanie niealkoholowego stłuszczenia wątroby u kobiet z nadwagą i otyłością z zespołem policystycznych jajników. Współzależności metaboliczne między oboma zespołami – badanie wstępne
Diagnosis of non-alcoholic fatty liver disease (NAFLD) in overweight and obese women with polycystic ovary syndrome (PCOS). Metabolic associations between NAFLD and PCOS – preliminary study
Department of Endocrinology, Center of Postgraduate Medical Education, Bielański Hospital, Warszawa
Head of Department: prof. Wojciech Zgliczyński, MD, PhD
Streszczenie
Wstęp. Badania wskazują, że zespół policystycznych jajników (PCOS) może zwiększać ryzyko rozwoju niealkoholowego stłuszczenia wątroby (NAFLD), i odwrotnie: stłuszczenie wątroby być czynnikiem ryzyka PCOS.
Cel pracy. Ocena częstości występowania niealkoholowego stłuszczenia wątroby (NAFLD) rozpoznawanego nieinwazyjnie u kobiet z zespołem policystycznych jajników (PCOS), a także zbadanie metabolicznych zależności między NAFLD i PCOS.
Materiał i metody. U 35 kobiet z nadwagą i otyłością z PCOS (19-49 lat, BMI 26,8-60,2 kg/m2) oznaczono aktywność aminotransferaz i GGTP oraz przeprowadzono badanie ultrasonograficzne.
Wyniki. U 17 z badanych (48,6%) rozpoznano NAFLD. Aktywność enzymów wskaźnikowych – AlAT i GGTP – była znamiennie wyższa, a wskaźnik de Ritisa (AspAT/AlAT) niższy u badanych z NAFLD w porównaniu do kobiet bez stłuszczenia wątroby. Wskaźniki te korelowały z miernikami otyłości. U kobiet z NAFLD stężenie glukozy, insuliny i wartość HOMA były wyższe niż u badanych bez stłuszczenia wątroby. AlAT i wskaźnik de Ritisa korelowały ze stężeniem LDL-cholesterolu. AlAT i GGTP korelowały także ze stężeniem triglicerydów. Wskaźnik LH/FSH był niższy u kobiet z NAFLD w porównaniu do badanych bez stłuszczenia wątroby.
Wnioski. Niealkoholowe stłuszczenie wątroby rozpoznano metodami nieinwazyjnymi niemal u połowy badanych kobiet z nadwagą i otyłością z zespołem policystycznych jajników. Nieinwazyjne wskaźniki diagnostyczne wykazywały korelację z miernikami otyłości oraz metabolizmu glukozy i lipidów. Niższe wartości wskaźnika LH/FSH u kobiet z NAFLD wskazują na możliwość dodatkowego wpływu zaburzeń związanych ze stłuszczeniem wątroby na regulację hormonalną kobiet z PCOS.
Summary
Introduction. Recent findings suggest that women with polycystic ovary syndrome (PCOS) may be at risk for developing non-alcoholic fatty liver disease (NAFLD) and conversely, hepatic steatosis may be a risk factor for PCOS.
Aim. To estimate noninvasively the prevalence of non-alcoholic fatty liver disease in overweight and obese women with polycystic ovary syndrome and to investigate potential metabolic associations between NAFLD and PCOS.
Material and methods. In 35 overweight and obese women with PCOS (19-49 years, BMI 26.8-60.2 kg/m2) aminotransferases activity was estimated and ultrasonography imaging was performed.
Results. In 17 of all subjects (48.6%) NAFLD was diagnosed. ALT and GGTP were significantly higher, and de Ritis ratio (ATP/ALT) was lower in women with NAFLD in comparison to subjects without liver steatosis, and correlated with indices of obesity. In women with NAFLD glucose, insulin and HOMA were higher than in patients without liver disease. In NAFLD patients ALT and de Ritis ratio correlated with LDL-cholesterol. ALT and GGTP correlated also with triglyceride levels. LH/FSH ratio was lower in women with NAFLD in comparison to patients without liver steatosis.
Conclusions. Non-alcoholic liver disease was diagnosed non-invasively in nearly half of overweight/obese subjects with PCOS. Non-invasive markers of liver diseases correlated with indices of obesity, glucose and lipid metabolism. Lower LH/FSH ratio in women with NAFLD may suggest, that abnormalities associated with liver steatosis additionally contribute to hormonal dysregulation in women with PCOS.

Introduction
Polycystic ovary syndrome (PCOS) affects as many as 1 out of 10 women in the reproductive age, and therefore is one of the most frequent endocrine disorders in this population (1). The main signs and symptoms of PCOS are irregular, but one of the important features are polycystic ovaries, usually recognize by ultrasound imaging.
Approximately 50% of the women with PCOS characterize by overweight or obesity (2). Abdominal type of obesity that is dominating in this syndrome leads to insulin resistance in patients and is a well-recognized risk factor for further metabolic and hormonal disturbances.
Non-alcoholic fatty liver disease (NAFLD) is a chronic condition also associated with insulin resistance. It is defined by detection of ectopic fat accumulation in liver, either by imaging or by histology in the absence of other identifiable causes of liver steatosis, in particular in the absence of excessive alcohol consumption (3). Patients with liver steatosis have 3-fold higher prevalence of prediabetes and type 2 diabetes mellitus than healthy individuals (4). Moreover, disease in this advanced state may lead to cirrhosis in 10-15% of patients.
Regarding relationships between NAFLD and PCOS elevated alanine aminotransferase (ALT) activity, one of the non-invasive markers of liver diseases is a common finding in PCOS (5).
Features of NAFLD were found in 41% of patients with PCOS (6), and it is suspected that in obese women with PCOS number of liver steatosis is even higher (7). On the other hand, signs and symptoms of PCOS in women with diagnosed NAFLD reached 71% (8). Recent findings suggest that women with polycystic ovaries may be at risk for developing non-alcoholic fatty liver disease and conversely, hepatic steatosis may be a risk factor for polycystic ovary syndrome (9).
Aim
The goal of our study was to estimate the prevalence of non-alcoholic liver disease in overweight and obese women with PCOS. We also investigated potential associations between the some non-invasive markers of liver diseases and metabolic indices as well as hormones in our patients with PCOS.
Material and methods
We evaluated 35 overweight and obese women with PCOS aged 19-49 years, mean 28.7 ± 7.6 (x ± SD) with BMI 26.8-60.2 kg/m2 (36.9 ± 8.0). In all subjects polycystic ovary syndrome was diagnosed according to the Rotterdam consensus criteria (10). Other conditions with similar signs were ruled out. Clinical hyperandrogenaemia was defined as presence of hirsutism and/or acne. Biochemical hyperandrogenaemia was defined as serum testosterone levels greater than 3.1 nmol/L, dehydroepiandrosterone-sulfate levels (DHEA-S) greater than 2000-4100 ng/ml depending on age, or free androgen index (FAI) > 5. Ovaries in US were defined as polycystic when they included either 10 or more follicles measuring 2-9 mm in diameter or their volume was greater than 10 cm3 (fig. 1). Similar conditions: hyperprolactinemia, Cushing’s syndrome or nonclassical congenital adrenal hyperplasia were excluded. Also use of oral contraceptives and other hormonal drugs excluded from the study.
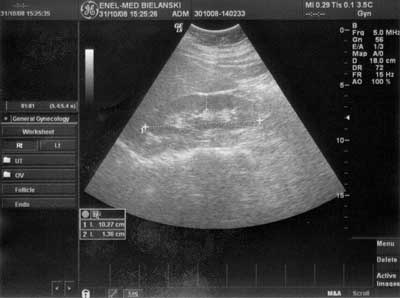
Fig. 1. Example of ultrasound imaging of polycystic ovary in one of our patients (K. M.).
Non-alcoholic fatty liver disease was diagnosed according to actual criteria of American Association for the Study of Liver Diseases (AASLD) (3) which include (1) evidence of liver steatosis by imaging or by histology, and (2) no causes for secondary hepatic fat accumulation such as significant alcohol consumption, use of steatogenic medication or hereditary disorders. On the basis of US imaging concomitant with data form medical history all patients were divided into group I including subjects without fatty liver disease and group II of women with diagnosed NAFLD. In agreement with recommendations of AASLD (3) we did not performed liver biopsy in our polycystic ovaries suffering, but otherwise healthy patients.
All subjects were studied after an overnight fast. As a part of physical examination body height and weight were measured, and then body mass index (BMI) was calculated. Blood was collected at about 0800 h for glucose, lipids (total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol and triglycerides), high sensitive C-reactive protein (hsCRP), alanine and aspartate aminotransferases (ALT and AST), gamma glutamyl transpeptidase (GGTP), insulin, LH, FSH, testosterone, dehydroepiandrosterone-sulfate (DHEA-S), 17-hydroxyprogesterone, sex hormone-binding globulin and TSH, through an iv catheter placed in the forearm. The liver enzymes have traditionally been used as surrogate markers of liver disease; however, their accuracy is limited. Then, we additionally calculated de Ritis ratio, as AST (IU/L)/ALT (IU/L) activity (11). Homeostasis Model of Assessment – Insulin Resistance (HOMA) was calculated by the formula: fasting plasma insulin (microinternational units per milliliter) x fasting plasma glucose (millimoles per liter)/22.4. Subjects were considered as insulin resistant when HOMA index was > 2.5. Free androgen index (FAI) was calculated as testosterone (nmol/l)/SHBG (nmol/l) levels. FAI > 5 indicated hyperandrogenemia.
All of the subjects underwent transvaginal ultrasonography (TV-US) and US of abdomen to estimate liver echogenity and to exclude adrenal pathology. In ultrasound evaluation of hepatic steatosis usually four criteria are used: parenchymal brightness, liver to kidney contrast, deep beam attenuation, bright vessel walls (12). We based on descriptions from our US department, in which hepatic steatosis or at least liver hyperechogenity were featured. Body composition was determined by dual-energy absorptiometry method (DEXA, fig. 2).
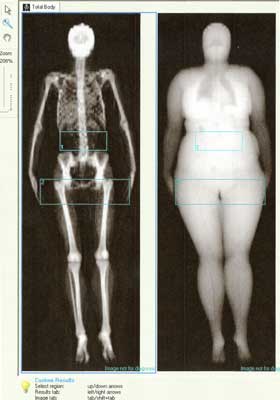
Fig. 2. Example of imaging of body composition by the DEXA method in one of our patients (M. P.).
The local ethical committee approved the study and informed consent was obtained from all of the participants.
Assays
Glucose was measured with glucose hexokinase reagent set with sensitivity 2.16 mg/dL. An enzymatic colorimetric method was used to measure total cholesterol in the presence of cholesterol oxidase and esterase. The sensitivity was 0.116 mg/dL. HDL-cholesterol was measured with enzymatic colorimetric method; sensitivity was 3 mg/dL. Triglycerides were also measured with enzymatic colorimetric method with sensitivity 0.85 mg/dL. All mentioned enzymatic (ALT, AST, GGTP) and biochemical measurements were performed using Roche Cobas Integra 400 chemistry analyzer (Roche Diagnostics). Insulin was measured by immunoradiometric method (Insulin IRMA – Immunotech SA, France); sensitivity was 2.0 mIU/ml. TSH, LH, FSH and estradiol were measured by immunochemiluminescence method with IMMULITE 2000 (Siemens Healthcare Diagnostics, Inc); sensitivity for estradiol was 15 pg/ml. Also total testosterone was measured by immunochemiluminescence method with IMMULITE 2000 (Siemens Healthcare Diagnostics, Inc); sensitivity was 0.15 ng/ml (= 0.5 nmol/l). Results were then multiplied by factor 3.46 to obtain nmol/l. Dehydroepiandrosterone-sulfate was measured by immunochemiluminescence method with IMMULITE 2000 (Siemens Healthcare Diagnostics, Inc); sensitivity of this method was 30 ng/ml. 17-hydroxyprogesterone was measured by 17OH-RIA-CT Kit (DIAsource ImmunoAssays SA, Belgium); detection limit: 0.02 ng/ml.
Body mass index was calculated as a body weight (kg)/height (m2). To perform measurements of body composition we used Lunar Prodigy (GE Lunar, Madison, WI, USA) device, which was calibrated each day with a standardized phantom and serviced regularly. The coefficient of variation for measurements with this method is about 2%.
Statistical analysis
All the data are presented as the mean ± SD. The distribution of continuous variables was tested for normality by the Kolmogorov-Smirnov test. To examine bivariate relationships between data Pearson correlation or linear Spearman’s rank correlation analyses were used. Comparisons between groups with normal distribution of the data were performed by unpaired Student’s t-test, in other cases comparisons were performed by Kolmogorov-Smirnov test for two samples. For all analysis, a two-tailed P ≤ 0.05 was considered to indicate statistical significance. All calculations were performed with the Statistica 8.0 software package (StatSoft Inc, Tusla, OK, USA).
Results
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. March WA, Moore VM, Willson KJ et al.: The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod 2010; 25: 544-551.
2. Gambineri A, Pelusi C, Vicennati V et al.: Obesity and the polycystic ovary syndrome. Int J Obes Relat Metab Disord 2002; 26: 883-896.
3. Chalasani N, Younossi Z, Lavine JE et al.: The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology 2012; 142: 1592-1609.
4. Ortiz-Lopez C, Lomonaco R, Orsak B et al.: Prevalence of prediabetes and diabetes and metabolic profile of patients with nonalcoholic fatty liver disease (NAFLD). Diabetes Care 2012; 35: 873-878.
5. Vassilatou E, Lafoyianni S, Vryonidou A et al.: Increased androgen bioavailability is associated with non-alcoholic liver disease in women with polycystic ovary syndrome. Hum Reprod 2010; 25: 212-220.
6. Cerda C, Pèrez-Ayuso RM, Riquelme A et al.: Nonalcoholic fatty liver disease in women with polycystic ovary syndrome. J Hepatol 2007; 47: 412-417.
7. Setji TL, Holland ND, Sanders LL et al.: Nonalcoholic steatohepatitis and nonalcoholic fatty liver disease in young women with polycystic ovary syndrome. J Clin Endocrinol Metab 2006; 91: 1741-1747.
8. Brzozowska MM, Ostapowicz G, Weltman MD: An association between nonalcoholic fatty liver disease and polycystic ovarian syndrome. J Gastroenterol Hepatol 2009; 24: 243-247.
9. Baranova A, Tran TP, Birerdinc A, Younossi ZM: Systemic review: association of polycystic ovary syndrome with metabolic syndrome and non-alcoholic fatty liver disease. Aliment Pharmacol Ther 2001; 33: 801-814.
10. Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group 2004 Revised 2003 consensus on diagnostic criteria and long-term health risk related to polycystic ovary syndrome Fertil Steril 81: 19-25.
11. Hall P, Cash J: What is the Real Function of the Liver ‘Function’ Tests? Ulster Med J 2012; 81: 30-36.
12. Saadeh S, Younossi ZM, Remer EM et al.: The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002; 123: 745-750.
13. Tharger G, Day CP, Bonora E: Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 2010; 363: 1341-1350.
14. Teede H, Deeks A, Moran L: Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med 2010: 8: 41.
15. Legro RS, Castrance VD, Kauffman RP: Detecting insulin resistance in polycystic ovary syndrome: purposes and pitfalls. Obstet Gynecol Surv 2004; 59: 141-154.
16. Karoli R, Fatima J, Chandra A et al.: Prevalence of hepatic steatosis in women with polycystic ovary syndrome. J Hum Reprod Sci 2013; 6: 9-14.
17. Vernon G, Baranova A, Younossi ZM: Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 2011; 34: 274-285.
18. Sorbi D, Boynton J, Lindor KD: The ratio of aspartate aminotransferase to alanine aminotransferase: potential value in differentiating nonalcoholic steatohepatitis from alcoholic liver disease. Am J Gastroenterol 1999; 94: 1018-1022.
19. Fracanzani AL, Valenti L, Bugianesi E et al.: Risk of severe liver disease in nonalcoholic fatty liver disease with normal aminotransferase levels: a role for insulin resistance and diabetes. Hepatology 2008; 48: 792-798.
20. Collantes RS, Ong JP, Younossi ZM: The metabolic syndrome and nonalcoholic fatty liver disease. Panminerva Med 2006; 48: 41-48.
21. Kotronen A, Westerbacka J, Bergholm R et al.: Liver fat in the metabolic syndrome. J Clin Endocrnol Metab 2007; 92: 3490-3497.
22. Jornayvaz FR, Samuel VT, Shulman GI: The role of muscle insulin resistance in the patoghenesis of atherogenetic dyslipidemia and nonalcoholic fatty liver disease associated with the metabolic syndrome. Annu Rev Nutr 2010; 30: 273-290.
23. Petersen KT, Dufour S, Savage DB et al.: The role of skeletal muscle insulin resistance in the pathogenesis of the metabolic syndrome. Proc Natl Acad Sci USA 2007; 104: 12587-12594.
24. Adiels M, Taskinen M-R, Boren J: Fatty liver, insulin resistance, and dyslipdemia. Curr Diab Rep 2008; 8: 60-64.
25. Browning JD: Statins and hepatic steatosis: perspectives from the Dallas Heart Study. Hepatology 2006; 44: 466-471.
26. Morales AJ, Laughlin GA, Butzow T et al.: Insulin, somatotropic, and luteinizing hormone axes in lean and obese women with polycystic ovary syndrome: Common and distinct features. J Clin Endocrinol Metab 1996; 81: 2854-2864.


