© Borgis - Postępy Nauk Medycznych 12/2014, s. 852-859
*Karolina M. Nowak, Lucyna Papierska
Zapobieganie i monitorowanie działań niepożądanych przewlekłej steroidoterapii
Prevention and monitoring of the side effects of chronic corticosteroid therapy
Department of Endocrinology, Centre of Postgraduate Medical Education, Bielański Hospital, Warszawa
Head of Department: prof. Wojciech Zgliczyński, MD, PhD
Streszczenie
Glikokortykosteroidy (GKS) wykorzystywane są w leczeniu chorób autoimmunologicznych, hematologicznych, alergicznych i innych o podłożu zapalnym. Przez lata tworzono kolejne syntetyczne związki, mając na uwadze, aby wywierały jak najsilniejszy efekt przeciwzapalny i jak najmniejsze działania niepożądane. Podczas krótkotrwałego leczenia glikokortykoidami korzyści przewyższają zazwyczaj efekty uboczne, z czasem jednak powikłania terapii mogą stać się dla pacjenta obciążeniem równie poważnym jak choroba podstawowa. Zwykle im wyższa dawka i dłuższy czas terapii, tym liczniejsze i groźniejsze działania niepożądane. Należy pamiętać o tym, że w większości przypadków zaniechanie kortykoterapii nie jest możliwe – nie unikniemy więc działań ubocznych GKS, ale przez odpowiednio prowadzoną profilaktykę możemy je zminimalizować.
W poniższej pracy dokonaliśmy przeglądu badań dotyczących monitorowania leczenia glikokortykosteroidami oraz zapobiegania najczęściej przez nie wywoływanym działaniom niepożądanym. Zaproponowaliśmy też schemat postępowania z pacjentem poddanym przewlekłej kortykoterapii.
Summary
Glucocorticoids are used in the treatment of autoimmune, hematological, allergic and other inflammatory diseases. Over the years many synthetic compounds were created in order to exert the strongest anti-inflammatory effect with the least side effects. While short--term treatment with glucocorticoids benefits usually outweigh the adverse events, with time, however, complications of therapy may become a burden for the patient as serious as the underlying disease. Typically the higher the dose and time of treatment, the more numerous and more serious side effects are. In most cases we are not able to give up treatment with corticosteroids and for that reason it is not possible to avoid the side effects. However, with proper prevention methods we can minimize them.
In the paper we present the literature review concerning monitoring the treatment and prevention of the most common side effects of glucocorticoids. We also propose the scheme of the management with patient during chronic glucocorticoid treatment.

Introduction
For the first time glucocorticoids (GCS) were used in medicine over 60 years ago in the treatment of rheumatoid arthritis and Addison’s disease which was then called a „cortisone miracle”. Due to their anti--inflammatory and immunomodulative action GCS are widely used in the treatment of many diseases. Despite unquestionable beneficial effects of this group of compounds, these medications are characterized by a large number of adverse events. The disorders caused by GCS, dependent on dose, the duration of treatment and route of administration, range from iatrogenic Cushing’s syndrome through hypothalamic-pituitary-adrenal axis suppression to neurological and psychiatrical diseases. Even inhaled glucocorticoids (ICS), intraarticularly injected or topically used are not free from systemic side effects. Due to the fact that GCS are one of the most used group of medicines, the important task facing modern medicine is to prevent the numerous adverse effects that they cause. The problem of serious side effects caused by glucocorticoids is the subject of studies for many years, however apart from the prevention of osteoporosis and gastrointestinal bleeding (with simultaneous use of nonsteroidal anti-inflamatory drugs), there are no Evidence Based Medicine recommendations concerning prevention of adverse events during treatment.
The chemical structure, pharmacokinetics and metabolism of GCS
Obtaining glucocorticosteroids with stronger anti--inflammatory activity can be achieved by modification of cortisol structure. These changes include the formation of a double bond between the 1st and 2nd position, which increases anti-inflammatory effect with reduction of binding with mineralocorticoid receptor (prednisolone). Other modifications include the introduction of fluoro group in 9th position (Dexamethasone, Triamcinolone, Fludrocortisone), methyl group in 6th position (Methylprednisolone) and 16th (Dexamethasone), which effected in further intensification of anti-inflammatory activity, reduction of mineralocorticoid effect and simultaneously prolongation of action. Exception in the mentioned group is fludrocortisone, which to a great extent is characterized by the mineralocorticoid effect and it is not used for anti-inflammatory activity. Modification in chemical structure and activity of glucocorticoids, comparing to cortisol, are presented in table 1 and figure 1.
Table 1. The structure and examples of equivalent doses and duration of action of glucocorticosteroids (1).
| Name | Modification in chemical structure comparing to cortisol | Examples of equivalent doses of glucocorticosteroids | Anti-inflammatory activity | Mineralocorticoid activity | Duration of action
(T 1/2 in hours) |
| Cortisol | – | 20 mg | 1 | 1 | 8-12 h |
| Hydrocortisone | No modification | 25 mg | 0.8 | 0.8 | 8-12 h |
| Prednisone | Double bond between 1st and 2nd position, ketone group in 11th position | 5 mg | 4 | 0.8 | 12-36 h |
| Prednisolone | Double bond between 1st and 2nd position | 5 mg | 4 | 0.8 | 12-36 h |
| Methylprednisolone | Double bond between 1st and 2nd position, methyl group in 6th position | 4 mg | 5 | 0.5 | 12-36 h |
| Triamcinolone | Double bond between 1st and 2nd position, fluor group in 9th position | 4 mg | 5 | 0 | 12-36 h |
| Dexamethasone | Double bond between 1st and 2nd position, fluor group in 9th position methyl group in 16th position | 0.75 mg | 30 | 0 | 36-72 h |
| Fludrocortisone | Fluor group in 9th position | – | 10 | 125 | 12-36 h |
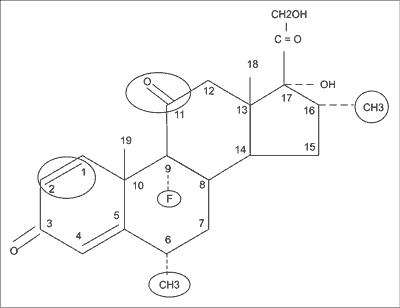
Fig. 1. Modification in chemical structure of cortisol (explanation in table 1).
The presence of hydroxyl group in 11th position is responsible for glucocorticoid and anti-inflammatory activity while substances with ketone group in the mentioned position (cortisone, prednisone) are biologically inactive and first have to be hydroxylated by 11β-hydroxysteroid dehydrogenase type 1. This enzyme is present in almost every tissue susceptible to glucocorticoids. The 11β-hydroxysteroid dehydrogenase type 2 is responsible for the reverse reaction of inactivation (oxidation). The medicines having in its structure a fluoro or methyl group are insensitive to the action of this enzyme. Cortisol, to a small extent, is also metabolized by 6β-hydroxylase (to biologically inactive 6β-hydrocortisone). This reaction is catalyzed by cytochrome P450 3A4. Therefore, interactions can occur between glucocorticosteroids (including ICS and intranasally admitted) and drugs inducing or inhibiting CYP3A4. The strong inhibitors of CYP 3A4 may slow the metabolism of GCS and in consequence cause its’ increased concentration and toxicity. The strong inducers of CYP 3A4 may cause the reverse reaction. The most important drugs that can influence the metabolism of glucocorticosteroids are listed in table 2.
Table 2. Drugs influencing the metabolism of glucocorticosteroids (including ICS) (2, 3).
| Strong inhibitors of CYP3A4 | Weak inhibitors of CYP3A4 | Strong inducers of CYP3A4 |
| Antiviral drugs (e.g. Atazanavir, Cobicistat, Darunavir) | Amiodaron, Dronedaron | Nevirapine, Efavirenz, Etravirine |
| Boceprevir, Telaprevir | Aprepitant, Fosaprepitant | Phenytoin, Fosphenytoin, Carbamazepine, Oxcarbazepine, Primidone |
| Itraconazole, Ketoconazole, Pasaconazole, Voriconazole | Fluconazole, Clotrimazole, Miconazole | Griseofulvin |
| Clarithromycin, Telithromycin | Erythromycin, Ciprofloxacin, Norfloxacina, Metronidazole, Tetracycline | Rifampin, Rifabutin, Rifapentine |
| Imatinib | Crizotinib, Lapatinib | Vemurafenib |
| Conivaptan | Cyklosporine | Barbiturates |
| Cimetidine* | Grapefruit juice | Nafcillin |
| Diltiazem*, Nicardipine | Verapamil | Aminoglutethimide, Mitotane |
| Fluoxetine | Sertraline, Haloperidol, Desipramine | Modafinil |
| Isoniazid | Abiraterone, Bicalutamide | Enzalutamide |
*These drugs are moderate inhibitors of CYP3A4 (2).
Regardless of the effect on cytochrome P450, glucocorticoids may increase the potency of other drugs for instance warfarin by not well known multiple mechanism, diuretics by enhancing their kaluretic action or NSAIDs by increasing the risk of peptic ulcer (4).
Endogenous glucocorticoids in blood are bound mainly (in 75%) to CBG protein (Corticosteroid Binding Globulin), in 15% are transported by albumins (in this form it is biologically inactive fraction) while 10% is a free fraction. CBG (transcortin) concentration increases under influence of medicaments such as mitotane, high estrogen levels (pregnancy, hormone replacement therapy, oral contraception) or in pathological conditions (diabetes, hyperthyroidism) (5). Increased CBG concentration may give falsely high values of serum total cortisol concentration. In such cases free cortisol in the blood or saliva should be measured.
Synthetic GCS (except prednisolone) are bound in 2/3 with albumins while the remaining 1/3 is present in serum in a free state (6). Protein binding also affects biological half-life, which for cortisol and cortisone is much shorter than that of the synthetic glucocorticoids. The results presented by Kozowera et al. shows that predisolone clearance in patients with side effects during corticotherapy was decreased comparing to those without adverse events (7). In the other study, elderly people had decreased corticoid clearance (8): for that reason it is necessary to lower GCS doses in this group of patients.
Comorbidities also affects the metabolism of GCS. The most important include:
Hyperthyroidism – the increased metabolism of prednisone and prednisolone, as well as the reduction of the biological effect, was reported (9). It may be necessary to use higher doses of GCS.
Inflammatory bowel disease – the investigation showed reduced protein binding with prednisolone in patients with active disease, probably caused by lower albumin concentration, which can accelerate the elimination of the drug from the body (10). Chronic kidney disease increases the half-life of GCS and thus it is necessary to reduce the dose. On the contrary, the patients on hemodialysis may need the increased dose of GCS.
Obesity – when determining the doses of glucocorticosteroids the ideal body weight should be taken into consideration instead the actual body weight.
Cirrhosis – together with deteriorating liver function, the concentration of 6β-hydroxylase may decrease, which leads to increased exposure to the biologically active form of corticosteroids (11). Hypoalbuminemia, increasing the free fraction of GCS is an additional factor causing that in this disease dose should be reduced.
General principles of initiating therapy with glucocorticoids
Before initiation of therapy with GCS the indications should be carefully considered. The determination whether the patient can receive other treatment for the given disease and if it has already been implemented, whether the dose is optimal. Therapeutic targets, assumed duration of therapy, the dose (the smallest dose for the shortest time period) and the preparation of GCS need to be identified. The most important is to define the criteria of effectiveness and ineffectiveness of treatment, when the treatment should be absolutely stopped. The next step is to identify the comorbidities whose course may deteriorate during treatment and may affect the metabolism of GCS (along with chronically taken medications) what in consequence may intensify action of GCS and lead to occurrence of side effects. Even very small dose of GCS (< 5 mg of prednisone equivalent) may cause adverse events thus it is important to follow the given recommendation at every case of chronic treatment with GCS. The typical doses of glucocorticoids are presented in table 3.
Table 3. The classification od glucocorticosteroid doses.
| The typical dose of glucocorticoids | The daily dose in prednisone equivalent |
| Small dose | ≤ 7.5 mg |
| Moderate dose | > 7.5 mg and ≤ 30 mg |
| High dose | > 30 mg and ≤ 100 mg |
| Very high dose | > 100 mg |
| Pulse dose | ≥ 250 mg |
The adverse events of chronic corticotherapy – prevention and monitoring
Cushingoid appearence and weight gain
Considering the number of patients treated with GCS annually, the fact that the main cause of Cushing’s syndrome is iatrogenic does not seem to be a surprise. The phenotype includes inter alia: weight gain, visceral obesity, plethora, dorsocervical fat pads („buffalo hump”), proximal myopathy, leg edema, ecchymoses, skin thinning and atrophy, red striae, hirsutism and acne (11). Not all of these symptoms must be present. The frequency of Cushingoid appearance is a „linear” dependent on dose and duration of treatment. It develops even while using less than 5 mg of prednisone/day and its occurrence is estimated from 4.3% after such doses to 24.6% when dosage increased over 7.5 mg prednisone/day during at least 6 months of glucocorticoid intake (12).
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Schimmer BP, Parker KL: Adrenocorticotropic hormone; adrenocortical steroids and their synthetic analogs; inhibitors of the synthesis and actions of adrenocortical hormones. [In:] Brunton LL, Lazo JS, Parker KL (eds.): The Pharmacological Basis of Therapeuthics. 11th ed., McGraw Hill, NY. P 2006: 1587.
2. Hansten PD, Horn JR: Top 100 drug interactions: guide to patient management. 12th ed., WA: H&H Publications, LPP 2012.
3. Czock D, Keller F, Rasche FM, Häussler U: Pharmacocinetics and pharmacodynamics of systemically administered glucocorticoids. Clin Pharmacokinet 2005; 44(1): 61-98.
4. Furst ED, Saag GK: Glucocorticoid withdrawal. Available at: http://www.uptodate.com/contents/glucocorticoid-withdrawal.
5. Zgliczyński W: Wielka Interna – Endokrynologia. Medical Tribune Polska, Warszawa 2012.
6. Ballard PL: Delivery and transport of glucocorticoids to target cells. [In:] Baxter JD, Rousseau GG (eds.): Glucocorticoid Hormone Action. Springer-Verlag, Berlin 1979: 25.
7. Kozower M, Veatch L, Kaplan MM: Decreased clearance of prednisolone, a factor in the development of corticosteroid side effects. J Clin Endocrinol Metab 1974; 38: 407.
8. Tornatore KM, Logue G, Venuto RC, Davis PJ: Cortisol pharmacodynamics after methylprednisolone administration in young and elderly males. J Clin Pharmacol 1997; 37: 304.
9. Frey FJ, Horber FF, Frey BM: Altered metabolism and decreased efficacy of prednisolone and prednisone in patients with hyperthyroidism. Clin Pharmacol Ther 1988; 44: 510.
10. Milsap RL, George DE, Szefler SJ et al.: Effect of inflammatory bowel disease on absorption and disposition of prednisolone. Dig Dis Sci 1983; 28: 161.
11. Renner E, Horber FF, Jost G et al.: Effect of liver function on the metabolism of prednisone and prednisolone in humans. Gastroenterology 1986; 90: 819.
12. Huscher D, Thiele K, Gromnica-Ihle E et al.: Dose-related patterns of glucocorticoid-induced side effects. Ann Rheum Dis 2009; 68: 1119.
13. Curtis JR, Westfall AO, Allison J et al.: Population-based assessment of adverse events associated with long-term glucocorticoid use. Arthritis Rheum 2006; 55: 420.
14. Hoes JN, Jacobs JW, Boers M et al.: EULAR evidence-based recommendations on the management of systemic glucocorticoid therapy in rheumatic diseases. Ann Rheum Dis 2007; 66: 1560.
15. Gurwitz JH, Bohn RL, Glynn RJ et al.: Glucocorticoids and the risk for initiation of hypoglycemic therapy. Arch Intern Med 1994; 154: 97.
16. Da Silva JA, Jacobs JW, Kirwan JR et al.: Safety of low dose glucocorticoid treatment in rheumatoid arthritis: published evidence and prospective trial data. Ann Rheum Dis 2006; 65: 285.
17. Angelopoulos TP, Tentolouris NK, Bertsias GK, Boumpas DT: Steroid-induced diabetes in rheumatologic patients. Clin Exp Rheumatol 2014 Jan-Feb; 32(1): 126-130.
18. Hirsch IB, Paauw DS: Diabetes management in special situations. Endocrinol Metab Clin North Am 1997; 26: 631.
19. Simmons LR1, Molyneaux L, Yue DK, Chua EL: Steroid-induced diabetes: is it just unmasking of type 2 diabetes? ISRN Endocrinol 2012; 2012: 910905.
20. Van der Goes MC, Jacobs JWG, Boers M et al.: Monitoring adverse events of low-dose glucocorticoid therapy: EULAR recommendations for clinical trials and daily practice. Ann Rheum Dis 2010 Nov; 69(11): 1913-1919.
21. Polish Diabetes Association: Zalecenia kliniczne dotyczące postępowania u chorych na cukrzycę. (Clinical Recommendations for the management of patients with diabetes). Clinical Diabetology 2013; 2 (suppl. A).
22. Walker BR: Glucocorticoids and cardiovascular disease. Eur J Endocrinol 2007 Nov; 157(5): 545-559.
23. Souverein PC, Berard A, Van Staa TP et al.: Use of oral glucocorticoids and risk of cardiovascular and cerebrovascular disease in a population based case-control study. Heart 2004; 90: 859.
24. Christiansen CF, Christensen S, Mehnert F et al.: Glucocorticoid use and risk of atrial fibrillation or flutter: a population-based, case-control study. Arch Intern Med 2009; 169: 1677.
25. White KP, Driscoll MS, Rothe MJ, Grant-Kels JM: Severe adverse cardiovascular effects of pulse steroid therapy: is continuous cardiac monitoring necessary? J Am Acad Dermatol 1994; 30: 768.
26. Piper JM, Ray WA, Daugherty JR, Griffin MR: Corticosteroid use and peptic ulcer disease: role of nonsteroidal anti-inflammatory drugs. Ann Intern Med 1991; 114: 735.
27. Carone FA, Liebow AA: Acute pancreatic lesions in patients treated with ACTH and adrenal corticoids. N Engl J Med 1957; 257: 690.
28. Sadr-Azodi O, Mattsson F, Bexlius TS et al.: Association of oral glucocorticoid use with an increased risk of acute pancreatitis: a population-based nested case-control study. JAMA Intern Med 2013; 173: 444.
29. Canalis E, Mazziotti G, Giustina A, Bilezikian JP: Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 2007; 18: 1319-1328.
30. Natsui K, Tanaka K, Suda M et al.: High-dose glucocorticoid treatment induces rapid loss of trabecular bone mineral density and lean body mass. Osteoporos Int 2006; 17: 105-108.
31. Sewerynek E: Current indications for prevention and therapy of steroid-induced osteoporosis in men and women. Endokrynologia Polska 2011; 62(1); suppl. II: 9.
32. Steinbuch M, Youket TE, Cohen S: Oral glucocorticoid use is associated with an increased risk of fracture. OsteoporosInt 2004; 15: 323-328.
33. The Royal College of Physicians: Glucocorticoid-induced osteoporosis. A concise guide to prevention and treatment. 2002. Available at: http://bookshop.rcplondon.ac.uk/contents/pub89-64206b70-b147-4976-9ee1-bf4948458468.pdf.
34. Grossman JM, Gordon R, Ranganath VK et al.: American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken) 2014 Apr 9.
35. Głuszko P, Lorenc R: Primary and secondary osteoporosis. Guidelines for rheumatologists. Reumatologia 2012; 50(5): 370-377.
36. Goncerz G: Polskie zalecenia postępowania diagnostycznego i leczniczego w osteoporozie – podsumowanie aktualizacji 2013. Medycyna Praktyczna 2013; 6: 33-46.
37. Papierska L, Rabijewski M, Kasperlik-Załuska A, Zgliczyński W: Effect of DHEA supplementation on serum IGF-1, osteocalcin, and bone mineral density in postmenopausal, glucocorticoid-treated women. Advances in Medical Sciences/Annales Academiae Medicae Bialostocensis 2012; 57(1): 51-57.
38. Shigemura T, Nakamura J, Kishida S et al.: Incidence of osteonecrosis associated with corticosteroid therapy among different underlying diseases: prospective MRI study. Rheumatology (Oxford) 2011 Nov; 50(11): 2023-2028.
39. Zizic TM, Marcoux C, Hungerford DS et al.: Corticosteroid therapy associated with ischemic necrosis of bone in systemic lupus erythematosus. Am J Med 1985 Nov; 79(5): 596-604.
40. Saag KG, Koehnke R, Caldwell JR et al.: Low dose long-term corticosteroid therapy in rheumatoid arthritis: an analysis of serious adverse events. Am J Med 1994; 96: 115.
41. Tripathi RC, Parapuram SK, Tripathi BJ et al.: Corticosteroids and glaucoma risk. Drugs Aging 1999; 15: 439.
42. Stuck AE, Minder CE, Frey FJ: Risk of infectious complications in patients taking glucocorticosteroids. Rev Infect Dis 1989; 11(6): 954-963.
43. Herron A, Dettleff G, Hixon B et al.: Influenza vaccination in patients with rheumatic diseases. Safety and efficacy. JAMA 1979; 242: 53.
44. Spika JS, Halsey NA, Fish AJ et al.: Serum antibody response to pneumococcal vaccine in children with nephrotic syndrome. Pediatrics 1982; 69: 219.
45. Fardet L, Petersen I, Nazareth I: Suicidal Behavior and Severe Neuropsychiatric Disorders Following Glucocorticoid Therapy in Primary Care. Am J Psychiatry 2012 May; 169(5): 447-449.
46. Minden SL, Orav J, Schildkraut JJ: Hypomanic reactions to ACTH and prednisone treatment for multiple sclerosis. Neurology 1988; 38: 1631.
47. Naber D, Sand P, Heigl B: Psychopathological and neuropsychological effects of 8-days’ corticosteroid treatment. A prospective study. Psychoneuroendocrinology 1996; 21: 25.
48. Schlaghecke R, Kornely E, Santen RT, Ridderskamp P: The effect of long-term glucocorticoid therapy on pituitary-adrenal responses to exogenous corticotropin-releasing hormone. N Engl J Med 1992 Jan 23; 326(4): 226-230.
49. Nieman KL: Pharmacologic use of glucocorticoids. Available at: http://www.uptodate.com.
50. Stewart PM, Krone NP: The adrenal cortex. [In:] Melmed S, Polonsky K, Larsen PR, Kronenberg H: Williams Textbook of endocrinology. 12th edition. Philadelphia 2011: 496-497.

