© Borgis - Postępy Nauk Medycznych 6/2015, s. 433-438
*Michał Mączewski, Monika Duda, Mariusz Marciszek, Joanna Kołodziejczyk, Urszula Mackiewicz
Wpływ diety wzbogaconej w wielonienasycone kwasy tłuszczowe omega-3 na funkcję skurczową i morfologię lewej komory oraz na wewnątrzkomórkowy obieg Ca2+ i elektrofizjologię w sercu szczura
The effect of omega-3 polyunsaturated fatty acids enriched diet on the contractile function, structure, Ca2+ handling and electrophysiology in the rat heart**
Department of Clinical Physiology, Medical Center of Postgraduate Education, Warszawa
Head of Department: Michał Mączewski, MD, PhD
Streszczenie
Wstęp. Wielonienasycone kwasy omega-3 (ω-3 WNKT) należą do grupy tzw. niezbędnych kwasów tłuszczowych, których organizm ssaków nie może syntetyzować de novo i muszą być one dostarczane z pokarmem. Badania epidemiologiczne sugerują, że suplementacja ω-3 WNKT zmniejsza śmiertelność z przyczyn sercowo-naczyniowych. Jednakże przeprowadzone dotychczas badania kliniczne i podstawowe dostarczyły różnorodnych i często sprzecznych wyników.
Cel pracy. Celem tej pracy jest zbadanie wpływu suplementacji dwóch głównych przedstawicieli ω-3 WNKT: kwasu eikozapentaenowego (EPA) i dokozaheksaenowego (DHA), na układ sercowo-naczyniowy.
Materiał i metody. Szczurom podawano karmę wzbogaconą w EPA, DHA lub standardową dietę kontrolną. Po 4 tygodniach rejestrowano potencjały czynnościowe z powierzchni serca, badano wewnątrzkomórkowy obieg Ca2+ w izolowanych kardiomiocytach oraz echokardiograficznie oszacowano funkcję i wymiary lewej komory serca.
Wyniki. Dieta wzbogacona w EPA i DHA nie miała wpływu na frakcję wyrzucania, grubość ściany i średnicę lewej komory. Dieta wzbogacona w EPA nie miała wpływu na parametry wewnątrzkomórkowego obiegu Ca2+, a dieta bogata w DHA zwiększała amplitudę potencjału czynnościowego i zawartość Ca2+ w siateczce sarkoplazmatycznej. Obie diety nie wywierały wpływu na czas trwania potencjału czynnościowego, podczas gdy dieta wzbogacona w EPA nieznacznie zwiększała jego amplitudę.
Wnioski. Uzyskane wyniki wskazują, że diety wzbogacone w ω-3 WNKT są bezpieczne dla układu sercowo-naczyniowego, ale wpływ DHA na wewnątrzkomórkowy obieg Ca2+ może przyczynić się w sprzyjających okolicznościach do powstania wapniozależnych zaburzeń rytmu serca.
Summary
Introduction. Omega-3 polyunsaturated fatty acids (ω-3 PUFA) are essential fatty acids that cannot be synthesized by mammals and must be delivered with food. Epidemiological observations suggest that supplementation of ω-3 PUFA reduces cardiovascular mortality, although results of clinical trials and animal studies are highly variable and inconsistent.
Aim. The aim of the study was to examine cardiovascular effects of ω-3 PUFA supplementation in healthy rats. Eicosapentanoic acid (EPA) and docosahexanoic acid (DHA), the two most commonly used ω-3 PUFAs, were used.
Material and methods. Rats were fed with normal chow or EPA- or DHA-enriched diet for 4 weeks. Epicardial monophasic action potentials (MAPs) were recorded, cardiomyocyte Ca2+ handling was assessed and eachocardiographic imaging was performed before and after 4 weeks of feeding.
Results. Neither EPA nor DHA affected left ventricular (LV) ejection fraction, wall thickness or diameters, indicating that 4 weeks of feeding had no effect on LV structure and function. While EPA enriched diet had no impact on intracellular Ca2+ handling, DHA-rich diet significantly increased amplitude of Ca2+ transient and SR Ca2+ content. Neither EPA nor DHA affected action potential duration, however EPA mildly increased action potential amplitude.
Conclusions. This indicates that ω-3 PUFAs are safe for the cardiovascular system, but the above mentioned cellular effects may be considered arrhythmogenic under specific conditions.

INTRODUCTION
Omega-3 polyunsaturated fatty acids (ω-3 PUFA) and omega-6 polyunsaturated fatty acids (ω-6 PUFA) belong to a group of so called essential fatty acids that cannot be synthesized by mammals due to lack of enzymes required to synthesize a double bond in a respective ω-3 or ω-6 position. Therefore ω-3 and ω-6 PUFA must be delivered with food.
Epidemiological observations suggest that diet rich in fatty marine fish (that are a rich source of eicosapentanoic acid (EPA) and docosahexanoic acid (DHA), two most common ω-3 PUFA) reduce cardiovascular mortality (Inuits from Grenland, Japanese from Okinawa) (1). Physicians Health Study demonstrated that males who consumed fish at least once weekly had lower risk of sudden cardiac death despite unchanged incidence of myocardial infarction (2). These observations were supported by clinical trials: in DART study increased fish consumption led to a 29% reduction of mortality of myocardial infarction patients (3). GISSI-Prevezione Trials demonstrated that ω-3 PUFA supplementation reduced sudden cardiac death by 45% (4). This and other evidence led to adoption of ω-3 PUFA supplementation by many organizations, such as American Heart Association, as a recommended measure to reduce cardiovascular risk.
However, there are emerging data on potentially detrimental effects of ω-3 PUFA supplementation. PUFAs also undergo random oxidation (also referred to as peroxidation or auto-oxidation) rapidly under the conditions of the human body and are a source of tissue-damaging free radicals. Omega-3 fatty acids can alter immune function sometimes in ways that may lead to a dysfunctional immune response to a viral or bacterial infection (5). Furthermore, experimental studies demonstrated that ω-3 PUFA can affect variety of cardiomyocyte-specific ion channels and transporters engaged in intracellular Ca2+ handling potentially affecting both contractility and susceptibility to arrhythmias (6).
AIM
The aim of the study was to examine cardiac safety of ω-3 PUFA supplementation, i.e. effects of 4-weeks of DHA and EPA feeding on left ventricular function, structure, action potential duration and parameters of cardiomyocyte Ca2+ handing to verify effects of ω-3 PUFA supplementation on both cardiac hemodynamic performance and pro/antiarrhythmic predisposition.
MATERIAL AND METHODS
Twenty four male Wistar-Kyoto rats, weighing 210-230 g, were used. All study animals were used in compliance with local and institutional regulations. The study conformed to the Guide for the Care and Use of Laboratory Animals, US National Institutes of Health (NIH Publication No. 85-23, revised 1996) and was approved by the local ethics committee.
Study protocol
The rats were randomly assigned to 3 groups (8 rats per group) that received control chow, chow enriched with EPA or DHA in amount 3% of total energy intake for 4 weeks. EPA and DHA were a gift from KD Pharma, Germany.
At the end of the feeding period, all animals underwent echocardiography, the epicardial monophasic action potentials (MAPs) were recorded and hearts were processed for cardiomyocyte isolation and Ca2+ handling measurements.
Echocardiography
Echocardiography was performed using MyLab25 (Esaote, Italy) with 13 MHz linear array transducer. Under light anesthesia (ketamine HCl and xylazine, 75 mg and 3.5 mg/kg body weight, IP) wall thickness were determined from the short-axis view at the midpapillary level. LV end-diastolic and end-systolic areas were planimetered from the parasternal long-axis view. LV ejection fraction (LVEF) was calculated as (LV diastolic area – LV systolic area)/LV diastolic area.
Myocyte isolation and Ca2+ transient recording
The LV myocytes were isolated by enzymatic digestion, as described previously (7) and superfused at 37°C with Tyrode’s solution containing 1.8 mmol/l Ca2+. Ca2+ transient was recorded using indo-1 fluorescence (excited at 365 and measured at 405 and 495 nm) (7).
Rate of Ca2+ transport by SERCA, NCX, PMCA and sarcoplasmic reticulum Ca2+ content
The rate of Ca2+ transport by sarcoplasmic reticulum (SR) Ca2+-ATPase (SERCA), Na+/Ca2+ exchanger (NCX) and plasma membrane Ca2+-ATPase (PMCA) was estimated from the rate constants (r1, r2, r3) of the single exponential curves fitted to electrically- and caffeine-evoked Ca2+ transients, as presented in figure 2A. The rate constants of the Ca2+ transient decay for SERCA and NCX was calculated according to formulas: rSERCA = r1-r2 and rNCX = r2-r3, respectively. The r3 was taken as an index of the rate of Ca2+ transport by PMCA (rPMCA = r3). rSERCA, rNCX and rPMCA describe average velocity of Ca2+ transport by SERCA and NCX and PMCA, respectively. SR Ca2+ content was estimated from the amplitude of caffeine-evoked Ca2+ transients in myocytes superfused with Na+, Ca2+-free (0Na0Ca) solution (fig. 2A) (7).
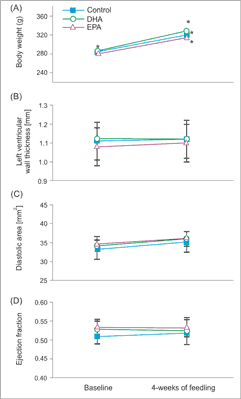
Fig. 1. Body mass, LV function and morphology in rats feeding with chaw and EPA or DHA enriched diet. Mean body mass (A), left ventricular wall thickness (B) left ventricular end-diastolic area (C) and left ventricular ejection fraction (D) in rats before and after 4 weeks of feeding with chaw and EPA or DHA enriched diets. Means ± SEM, n = 8 rat in each group.
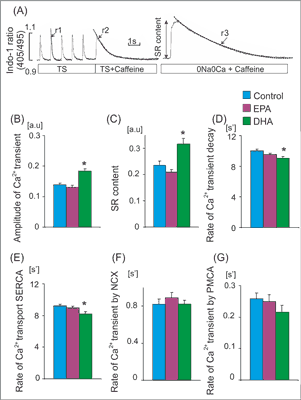
Fig. 2. Ca2+ handling parameters in rats receiving standard and EPA or DHA enriched diet. (A) Experimental protocols: cardiomyocytes were stimulated at 1Hz. Caffeine was applied in cardiomyocytes superfused with Tyrode solution (TS) or Na+/Ca2+ free solution (0Na0Ca). Single exponential curves were fitted to decaying part of electrically- or caffeine-evoked Ca2+ transients and rate constants of their decay (r1, r2 and r3) were calculated. The rate of Ca2+ transport by sarcoplasmic recticulum Ca2+-ATPase (SERCA) and by sarcolemmal transporters: Na+/Ca2+ exchanger (NCX) and sarcolemmal Ca2+-ATPase (PMCA) was calculated according to formulas: rSERCA = r1-r2 and rNCX = r2-r3, rPMCA=r3, respectively. Amplitude of Ca2+ transients evoked by caffeine in myocytes superfused with 0Na0Ca solution was taken as an index of sarcoplasmic reticulum (SR) Ca2+ content. Amplitude of Ca2+ transient (B), SR Ca2+ content (C), the rate of Ca2+ transient decay (D) and the rate of Ca transport by SERCA (E) NCX (F) and PMCA (G) in cardiomyocytes from rats receiving standard diet (Control) and EPA or DHA enriched diet. Means ± SEM, n = 32-120 measurements in myocytes isolated from 8 rat in each group.
*p < 0.05 EPA or DHA enriched diet vs. control diet
Monophasic action potentials recording
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. London B, Albert C, Anderson ME et al.: Omega-3 Fatty Acids and Cardiac Arrhythmias: Prior Studies and Recommendations for Future Research: A Report from the National Heart, Lung, and Blood Institute and Office of Dietary Supplements Omega-3 Fatty Acids and Their Role in Cardiac Arrhythmogenesis Workshop. Circulation 2007; 116(10): e320-e335.
2. Albert CM, Hennekens CH, O’Donnell CJ et al.: Fish Consumption and Risk of Sudden Cardiac Death. JAMA 1998; 279(1): 23-28.
3. Burr ML, Fehily AM, Gilbert JF et al.: Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: diet and reinfarction trial (DART). Lancet 1989; 2(8666): 757-761.
4. GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: results of the GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Lancet 1999; 354(9177): 447-455.
5. Fenton JI, Hord NG, Ghosh S, Gurzell EA: Immunomodulation by dietary long chain omega-3 fatty acids and the potential for adverse health outcomes. Prostaglandins Leukot Essent Fatty Acids. 2013; 89(6): 379-390.
6. Sankaranarayanan R, Venetucci L: Are the anti-arrhythmic effects of omega-3 fatty acids due to modulation of myocardial calcium handling? Front Physiol 2012. doi:10.3389/fphys.2012.00373.
7. Mączewski M, Mackiewicz U: Effect of metoprolol and ivabradine on left ventricular remodelling and Ca2+ handling in the post-infarction rat heart. Cardiovasc Res 2008; 79: 42-51.
8. Macleod JC, Macknight AD, Rodrigo GC: The electrical and mechanical response of adult guinea pig and rat ventricular myocytes to omega3 polyunsaturatedfatty acids. Eur J Pharmacol 1998; 356: 261-270.
9. Rodrigo GC, Dhanapala S, Macknight AD: Effects of eicosapentaenoic acid on the contraction of intact, and spontaneous contraction of chemically permeabilized mammalian ventricular myocytes. J Mol Cell Cardiol 1999; 31: 733-743.
10. Negretti N, Perez MR, Walker D, O’Neill SC: Inhibition of sarcoplasmic reticulum function by polyunsaturatedfatty acids in intact, isolated myocytes from rat ventricular muscle. J Physiol 2000; 523: 367-375.
11. O’Neill SC, Perez MR, Hammond KE et al.: Direct and indirect mod- ulation of rat cardiac sarcoplasmic reticulum function by n-3 polyunsaturatedfatty acids. J Physiol 2002; 538: 179-184.
12. Leifert WR, Dorian CL, Jahangiri A, McMurchie EJ: Dietary fish oil prevents asynchronous contractility and alters Ca(2+) handling in adult rat cardiomyocytes. J Nutr Biochem 2001; 12: 365-376.
13. Verkerk AO, vanGinneken AC, Berecki G et al.: Incorporated sarcolemmal fish oil fatty acids shorten pig ventricular action potentials. Cardiovasc Res 2006; 70: 509-520.
14. Billman GE, Harris WS, Carnes CA et al.: Dietary omega-3 fatty acids and susceptibility to ventricular fibrillation: lack of protection and a pro-arrhythmic effect. Circ Arrhythm Electrophysiol 2012; 5: 553-560.
15. McLennan PL, Barnden LR, Bridle TM et al.: Dietary fat modulation of left ventricular ejection fraction in the marmoset due to enhanced filling. Cardiovasc Res 1992; 26: 871-877.
16. Billman GE, Nishijima Y, Belevych AE et al.: Effects of dietary omega-3 fatty acids on ventricular function in dogs with healed myocardial infarctions: in vivo and in vitro studies. Am J Physiol Heart Circ Physiol 2010; 298: H1219-H1228.
17. Nattel S, Maguy A, Le Bouter S, Yeh YH: Arrhythmogenic Ion-Channel Remodeling in the Heart: Heart Failure, Myocardial Infarction, and Atrial Fibrillation. Physiol Rev 2007; 87: 425-456.
18. DenRuijter HM, Berecki G, Verkerk AO et al.: Acute administration of fish oil inhibits triggered activity in isolated myocytes from rabbits and patients with heart failure. Circulation 2008; 117: 536-544.
19. Berecki G, DenRuijter HM, Verkerk AO et al.: Dietary fish oil reduces the incidence of triggered arrhythmias in pig ventricular myocytes. Heart Rhythm 2007; 4: 1452-1460.


