*Jacek Wadełek
Colon perforation during colonoscopy – the role of an anaesthesiologist in patient management. A case report
Perforacja jelita grubego podczas wykonywania kolonoskopii – rola anestezjologa w prowadzeniu pacjenta. Opis przypadku
Department of Anaesthesiology and Intensive Care, St. Anne’s Provincial Hospital of Trauma Surgery, Mazowieckie Rehabilitation Centre STOCER, LLC, Warsaw
Streszczenie
Perforacja jelita podczas kolonoskopii jelita grubego jest poważnym powikłaniem. Objawy kliniczne zależą od wielkości przedziurawienia jelita grubego, stopnia wypełnienia jelita masami kałowymi oraz od miejscowych i ogólnych właściwości obronnych.
W pracy przedstawiono przypadek 81-letniego pacjenta, u którego doszło do perforacji w trakcie endoskopowej resekcji uszypułowanego polipa esicy. Przedziurawienia przewodu pokarmowego nie udało się zaopatrzyć endoskopowym klipsowaniem. Wykonano laparotomię, podczas której stwierdzono przedziurawienie esicy, zeszyto je. W okresie pooperacyjnym pacjent był leczony na oddziale anestezjologii i intensywnej terapii. W 3. dobie w stanie poprawy stanu ogólnego został wypisany na oddział chirurgii ogólnej.
Perforacja jelita w trakcie kolonoskopii jest poważnym powikłaniem, jednak wcześnie rozpoznana i zaopatrzona chirurgicznie jest kluczem do pomyślnego przebiegu choroby. Właściwa opieka anestezjologiczna, wyrównywanie zaburzeń metabolicznych i krążeniowo-oddechowych jest niezbędnym elementem w wyprowadzeniu pacjenta z tego powikłania.
Summary
Colon perforation is a serious complication of colonoscopy. Clinical signs and symptoms depend on the specific characteristics of the perforation (e.g., size, location, and aetiology) and patient’s general status.
The paper presents a case of an 81-year-old man who underwent diagnostic colonoscopy with perforation of the sigmoid diverticulum. The endoscopist was unsuccessful in ceiling the sigmoid perforation by clipping. Therefore, emergency laparotomy was performed, during which the perforation was repaired. Postoperatively, the patient was cared for in an intensive care unit. He was discharged from the intensive care unit to general surgery on day 3 postoperatively in a good general condition.
Colon perforation at colonoscopy is one of the most serious complications, with early diagnosis and surgical repair being the key to successful outcome. Proper anaesthetic management is centred around correction of metabolic, cardiovascular and respiratory derangements, which is also crucial for patient outcomes.

Introduction
Perforation of the colon is one of the most serious complications of colonoscopy (1). Although still rare, its incidence has increased with the dynamic development of modern techniques in the field of surgical endoscopy. Despite many years of observations, it is still disputable which factors are the main contributors to the risk of endoscopic perforation of the colon. The incidence of iatrogenic perforation of the colon is only 0.12% (2). Since the pressure of the intraluminal gas used for insufflation is increased during the procedure, free peritoneal gas is detected in almost all patients with colon wall damage (3). The paper presents a case of a patient who underwent emergency laparotomy due to endoscopic perforation of the colon, which was unsuccessfully managed by clipping during colonoscopy.
Case report
An 81-year-old man (height: 185 cm, body weight 75 kg, ASA class 3) after colonoscopy and endoscopic resection of a pedunculated sigmoid polyp was transferred from endoscopic laboratory to a general surgery suite for emergency laparotomy due to sigmoid perforation. Colonoscopy revealed multiple sigmoid diverticula and a 20 mm polyp with a thick pedicle located in between them. The polyp was removed with a diathermic loop. The resected polyp fell deeper into the sigmoid colon. During attempts at capturing the polyp, it migrated further into the sigmoid diverticulum, resulting in perforation. The perforation was secured with several clips; however, the closure was unsuccessful. The gas used for colonoscopy escaped through sigmoid perforation, causing pneumoperitoneum with significant enlargement of the abdominal circumference and a large subcutaneous emphysema of the hypogastrium and the upper part of lower limbs, with peripheral cyanosis. Chest radiography revealed free gas under the diaphragm dome (fig. 1). Emergency laparotomy was planned due to the diagnosed perforation of the sigmoid diverticulum and the lack of possibility to perform endoscopic repair (fig. 2). Electrocardiography showed no ischaemic changes or arrhythmias. Laboratory findings were unremarkable. Patient’s medical history of chronic diseases revealed arterial hypertension. The patient was on chronic therapy for hypertension: ramipril (Tritace) 5 mg daily, metoprolol (Metocard ZK) 12.5 mg daily, amlodipine (Amlodipine) 5 mg in the evening. Premedication involved an intravenous administration of atropine at 0.25 mg and midazolam at 2 mg. The patient also received preoperative infusion of an antibiotic (Tazocin 4.5 g). Basic monitoring was set up in the operating room: ECG monitor was connected to the electrodes; periodic non-invasive BP monitoring, pulse oximeter and capnograph were set up. Vital signs monitored before induction for general anaesthesia were as follows: blood pressure 150/80 mmHg, heart rate 105/min, percutaneous saturation 100%, body temperature 36.7°C. The patient was explained about the method of and the need for applying pressure on the cricoid cartilage before endotracheal intubation. In order to monitor the depth of anaesthesia, electrodes were applied to the patient’s forehead for the analysis of the bioelectrical activity of the brain (the bispectral index – BIS). In order to monitor muscle relaxation, neuromuscular blockade was monitored with a peripheral nerve stimulator with the use of two electrodes placed in the ulnar nerve area, using a series of four pulses sent at a frequency of 2 Hz (Train of Four – TOF), and with the motor responses of the adductor pollicis (thumb) muscle recorded visually. Before induction of anaesthesia, the patient, already placed on the operating table, received 5-minute passive oxygenation with oxygen 100%, as well as IV lidocaine 1% at 50 mg and titrated propofol at 80 mg. IV fentanyl was administered at a dose of 3 μg/kg body weight. The patient was appropriately positioned for intubation. Non-invasive monitoring of the patient (SpO2, HR, 3-lead ECG, NIBP) was initiated, and pressure on the cricoid cartilage was applied. Muscle relaxation was achieved after administering IV succinylcholine (1.5 mg/kg). The trachea was intubated using direct laryngoscopy (glottic view). Under visual control, an 8 mm endotracheal tube was introduced into the trachea between the vocal cords. Correct placement of the endotracheal tube was checked in accordance with the procedure, by observing symmetrical chest movements during mechanical ventilation; bilateral auscultation of the chest was performed, checking the bilateral respiratory sound; and capnography/capnometry monitoring was initiated. BIS value of 30 was observed. Sevoflurane inhalation (1-2 vol%) in an air/oxygen (inspired oxygen concentration of 40%) mixture was used to maintain anaesthesia. Fentanyl at a dose of 1 μg/kg body weight was used for intraoperative analgesia. Mechanical ventilation with an anaesthetic system ventilator was monitored with end-expiratory levels of carbon dioxide within the range of 35-40 mmHg. Intraoperatively, the cardiovascular system remained stable, the intraoperative body temperature was 36.5-36.7°C, and BIS was 40-50. TOF was 0.4 after a dose of muscle relaxant administered before endotracheal intubation and 0.6 during the remaining period of general anaesthesia. Surgical management involved emergency laparotomy, during which the peritoneal cavity was opened with left pararectus incision. Sigmoid rupture measuring 1 cm was located about 5 cm above the peritoneal fold. Perforation was closed with single absorbable sutures; continuous suture was used for the second layer. The peritoneal cavity was checked. Thorough irrigation of the peritoneal cavity with Betadine was performed. A drain was placed in the area of the damaged sigmoid through another incision. Haemostasis was performed and the abdominal wall was closed layer-by-layer. After suturing of the skin, the anaesthetic management involved discontinuation of sevoflurane, and ventilation was supported manually with oxygen 100% at 5 L/min. Before the return of efficient spontaneous breathing, BIS value was 85 and TOF was 0.9 after 4 minutes. During clinical examination, when asked to lift his head, the patient was able to keep his head raised for 4 seconds and to follow instructions. Once efficient spontaneous breathing was confirmed and consciousness returned, the endotracheal tube was removed from the airways. Due to the patient’s age and the risk of complications, he was postoperatively transferred from the general surgery suite to the Department of Anaesthesiology and Intensive Care. Postoperative analgesia involved continuous intravenous infusion of fentanyl at 1-2 μg/kg/hour and IV paracetamol at a dose of 4 g/day. Due to increased body temperature up to 38.5°C, the patient required physical cooling and additional administration of intravenous metamizole at 2.5 g. Preoperative antibiotic therapy was continued. On day 4, the patient was transferred to the Department of Surgery. On day 5, the patient was discharged home in good overall condition.
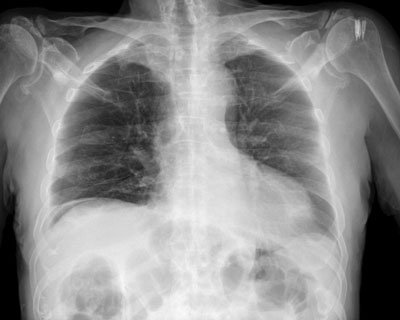
Fig. 1. Chest radiography. Free gas under the diaphragm dome on the right side
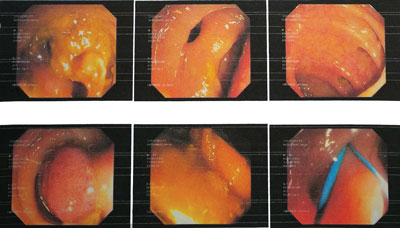
Fig. 2. Colonoscopy. Multiple sigmoid diverticula and a polyp with a wide pedicle
Discussion
Powyżej zamieściliśmy fragment artykułu, do którego możesz uzyskać pełny dostęp.
Mam kod dostępu
- Aby uzyskać płatny dostęp do pełnej treści powyższego artykułu albo wszystkich artykułów (w zależności od wybranej opcji), należy wprowadzić kod.
- Wprowadzając kod, akceptują Państwo treść Regulaminu oraz potwierdzają zapoznanie się z nim.
- Aby kupić kod proszę skorzystać z jednej z poniższych opcji.
Opcja #1
29 zł
Wybieram
- dostęp do tego artykułu
- dostęp na 7 dni
uzyskany kod musi być wprowadzony na stronie artykułu, do którego został wykupiony
Opcja #2
69 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 30 dni
- najpopularniejsza opcja
Opcja #3
129 zł
Wybieram
- dostęp do tego i pozostałych ponad 7000 artykułów
- dostęp na 90 dni
- oszczędzasz 78 zł
Piśmiennictwo
1. Wadełek J: Wybrane powikłania kolonoskopii – punkt widzenia anestezjologa. Nowa Med 2018; 25(3): 164-175.
2. Luning TH, Keemers-Gels ME, Barendregt WB: Colonoscopic perforations: a review of 30,366 patients. Surg Endosc 2007; 21: 994-999.
3. Cobb WS, Heniford BT, Sigmon LB et al.: Colonoscopic perforations: incidence, management, and outcomes. Am Surg 2004; 70: 750-757.
4. Azer S: Intestinal perforation treatment and management; http://emedicine.medscape.com/article/195537-treatment#d10 (data dostępu 01.2018).
5. National Emergency Laparotomy Audit 2016 Reports: Third NELA patient audit report; http://www.nela. org.uk/reports (data dostępu 01.2018).
6. Andeweg CS, Mulder IM, Felt-Bersma RJ et al.: Guidelines of diagnostics and treatment of acute left-sided colonic diverticulitis. Dig Surg 2013; 30(4-6): 278-292.
7. Wadełek J: Anestezjologiczna ocena i przygotowanie dorosłego pacjenta do dużej operacji urologicznej. Geriatria 2014; 8: 248-257.
8. Gray LD, Morris C: The principles and conduct of anaesthesia for emergency surgery. Anaesthesia 2013; 68 (suppl. 1): 14-29.
9. Tursi A, Papa A, Danese S: Review article: the pathophysiology and medical management of diverticulosis and diverticular disease of the colon. Aliment Pharmacol Ther 2015; 42(6): 664- 684.
10. Gatto NM, Frucht H, Sundararajan V et al.: Risk of perforation after colonoscopy and sigmoidoscopy: a population-based study. J Natl Cancer Inst 2003; 95: 230-236.
11. Cho SB, Lee WS, Joo YE et al.: Therapeutic options for iatrogenic colon perforation: feasibility of endoscopic clip closure and predictors of the need for early surgery. Surg Endosc 2012; 26: 473-479.
12. Hawkins AT, Sharp KW, Ford MM et al.: Management of colonoscopic perforations: A systematic review. Am J Surg 2018; 215(4): 712-718.
13. Priebe HJ: Cricoid pressure: an expert’s opinion. Minerva Anestesiol 2009; 75(12): 710-714.
14. Johnson RL, Cannon EK, Mantilla CB, Cook DA: Cricoid pressure training using simulation: a systematic review and meta-analysis. Br J Anaesth 2013; 111(3): 338-346.
15. Son YG, Shin J, Ryu HG: Pneumonitis and pneumonia after aspiration. J Dent Anesth Pain Med 2017; 17(1): 1-12.
16. Putzu A, Tramèr MR, Giffa M, Czarnetzki C: The optimal dose of succinylcholine for rapid sequence induction: a systematic review and meta-analysis of randomized trials. BMC Anesthesiol 2020; 20(1): 54.
17. El-Orbany M, Connolly LA: Rapid sequence induction and intubation: current controversy. Anesth Analg 2010; 110(5): 1318-1325.
18. Malbouisson LMS, Oliveira RAG: Intraoperative protective mechanical ventilation: what is new? Rev Bras Ter Intensiva 2017; 29(4): 404-407.
19. Brienza N, Biancofiore G, Cavaliere F et al.: Clinical guidelines for perioperative hemodynamic management of non cardiac surgical adult patients. Minerva Anestesiol 2019; 85(12): 1315-1333.
20. Dhesi JK, Lees NP, Partridge JS: Frailty in the perioperative setting. Clin Med (Lond) 2019; 19(6): 485-489.
21. Ghaffar S, Pearse RM, Gillies MA: ICU admission after surgery: who benefits? Curr Opin Crit Care 2017; 23(5): 424-429.
22. Marik PE: Perioperative hemodynamic optimization: a revised approach. J Clin Anesth 2014; 26(6): 500-505.



